Q: An aqueous solution contains 0.31 M hypochlorous acid. One Liter of this solution could be converted…
A: A buffer is the mixture of weak acid and its conjugate base or mixture of weak base with its…
Q: What major product is expected from the following reaction? I OH 01 OIV V Oll ||| Br 11 Br OH III…
A:
Q: The pH of 0.10 M hydrazine is 10.47 at 25 °C. What is the concentration of hydroxide in the…
A:
Q: Which does not belong to the group and why? Hg₂ + 2Cl⁻¹ ⇌ HgCl₂ Ag⁺¹ + Cl⁻¹ ⇌ AgCl₂ Pb⁺² + 2Cl⁻¹ ⇌…
A:
Q: A group of students is asked to predict which compounds could be added to increase the concentration…
A: 12. Le Chatelier's principle states that when a system under equilibrium is subjected to any kind…
Q: Give the starting materials. (a) (b) (c) (d) a O b O c Od Br Br + NaNH,
A:
Q: A solution has an [H3O+] of 3.35 x 10-4. What is the pOH of the solution? pOH = [?] Hint: Remember…
A:
Q: 10.1 Draw the structure of (E)-6-chloro-2-hexene
A:
Q: Identify the sites of steric strain in this ring. A H H H H H C Br B H -H H I E D
A:
Q: An experimental catalyst used to make polymers has the following composition: Co, 23.3%; Mo, 25.3%;…
A: Determination of empirical formula The percentage composition of elements present in given…
Q: Explain melting point determination as a characterization technique.
A:
Q: Choose the structure that best fits the following data. 200 180 160 140 120 100 ppm CDS-00-825 80 60…
A: The IR and HNMR tells us about the structure of the unknown compound .
Q: What is the expected major product of the following reaction? O II O I O IV I V OH 11 OH III H₂O*…
A:
Q: What is the hybridization of PS2Br2-
A:
Q: What is the 1, 4 - addition product in the reaction equation below? A.D B.A OC.E OD.C + HC1- B C D E
A: For the formation 1,4-addition product the carbocation has to undergo resonance stability to form…
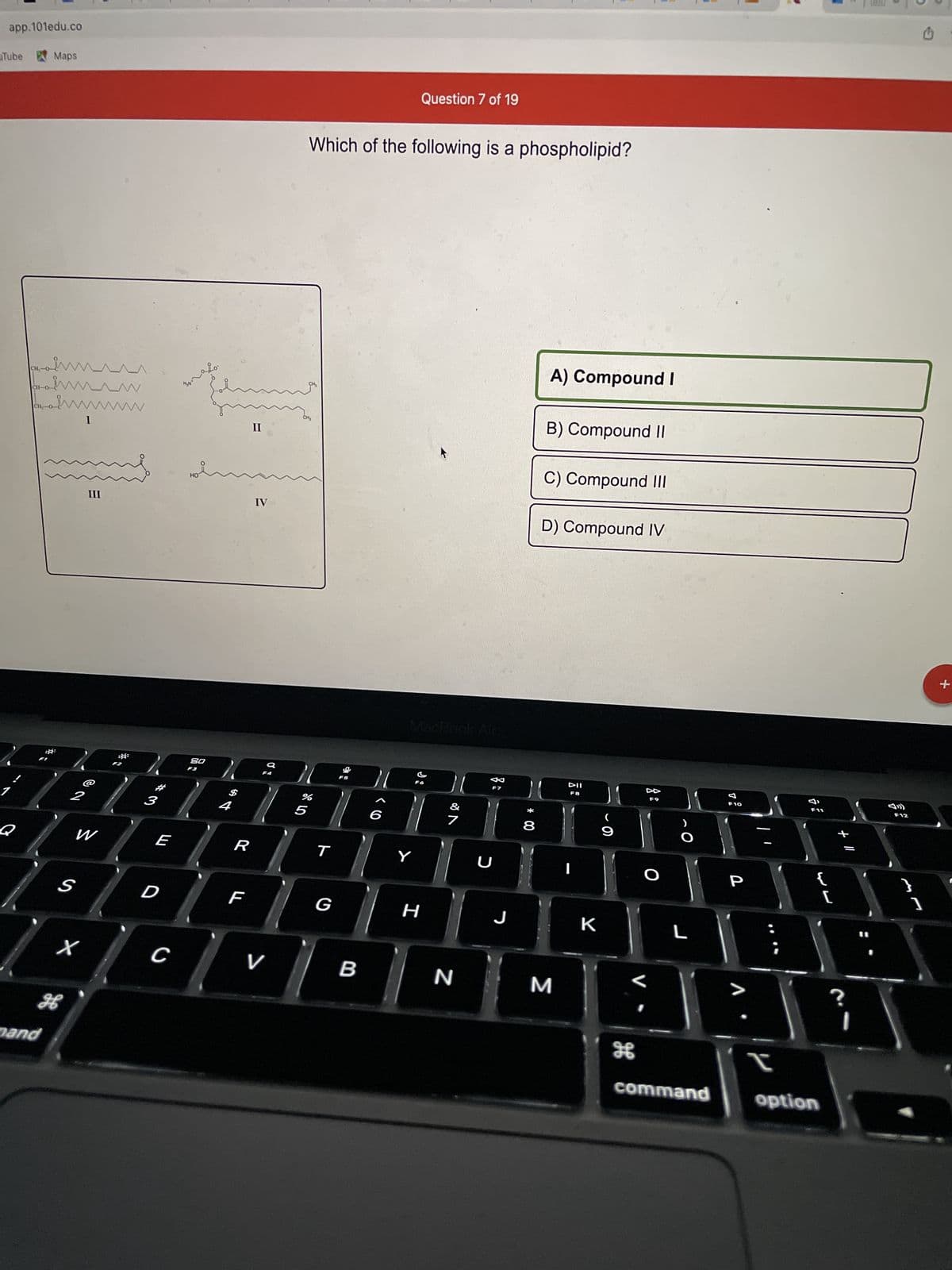
Q: CH2OH CIO HO—CH HC-OH HO—CH CH2OH MacBook Air A) ketohexose B) ketopentose C) aldohexose D)…
A: Thus is the example compound of ketohexose. As we can see here there are six no. Of carbon atoms…
Q: Select the major product of the following reaction. 0 Br HBr, peroxides Br Br -Br to save
A:
Q: In a laboratory experiment, a student set up an electrochemical cell that contained sodium bromide,…
A:
Q: 8. Given the molecule BrSeSF₂¹where Br breaks the octet, what would be the Geometry? What is the…
A: Given that Br breaks the octet rule. This signifies that in this molecule bromine will act as the…
Q: Which substituent would be assigned the highest priority when naming an alkene E or Z? 0 0 O 0=0 CH₂…
A: The higher the atomic number of the immediate substituent atom, the higher the priority. For…
Q: 0 +
A:
Q: 2. Show the Atomic and molecular orbitals of acetic acid. Draw the energy diagram for the AO and MO.…
A: The atomic and molecular orbital of acetic acid
Q: Which reaction is characteristic of alkenes and alkynes? a) Nucleophilic addition reaction b)…
A:
Q: Select the option that is a propagation step for the following action. 0 O Br + Br Br +HBr + HBr…
A: 1. Addition of HBr to alkene in presence of peroxide occure through free radical mechanism. 2. It…
Q: Explain why the following reaction doesn't work and provide alternative synthesis (1) LDA Br одан ж…
A:
Q: Which of the following is an energy diagram for a three-step reaction? O O A C B reaction coordinate…
A: Energy diagram is a graph which plots reaction coordinate against energy of a chemical reaction.
Q: Choose the best reagent for this reaction. Assume an acidic workup. Br (CH₂CH=CHCH₂CH₂)₂CuLi…
A:
Q: What is the correct name of this compound? CI R-2-chloropentane S-2-chloropentane R-2-chlorobutane…
A: Rules to assign R and S nomenclature The four atoms or group attached to chiral centre are given…
Q: Select the most stable conformation of this molecule. . НО. H3CH₂C H н CH3 н IV н II н НО. H3C…
A:
Q: Identify the configuration of the alkene: (E) or (Z). Br. OZ Not a case of E vs. Zisomerism. OE
A:
Q: Which is the correct sequence of reaction steps necessary to complete the following transformation?…
A: we have to find the correct sequence of reaction steps
Q: HA+KOH → NaA + H₂O Nia titrates half of a weak acid solution with KOH until she observes the…
A:
Q: Example 18-11: Calculate the concentrations of the various species in 0.15 M acetic acid, CH3COOH,…
A: Given, [CH3COOH] = 0.15 M Calculate the concentrations of the following species at equilibrium ?…
Q: Determine whether the two structures in each of the following pairs represent enantiomers,…
A:
Q: (SHOW CALCULATIONS) A sample of argon gas occupies 33.0 L at 85.0°C. What volume will the gas occupy…
A:
Q: Consider the reaction H₂(g) + C₂H4(g)→→→→→C₂H6(g) Using the standard thermodynamic data in the…
A: Solutions Using the thermodynamic data are given table
Q: Answer it completely. Dont answer partially. Two tanks with constant temperature and volume. One…
A: Given that , There are two tanks . Tank -1 have 10 moles of Ar gas. Tank-2 have 10 moles of He…
Q: What is the IUPAC n
A: According to IUPAC 1st give numbering to longest chain according to priority order i.e ethyl will be…
Q: 4 Nitrogen is less reactive than flourine. Why could the effective nuclear charge be used to…
A: Effective nuclear charge is basically the nuclear charged faced by the valance electron. The valance…
Q: Br Br CH₂ONa CH₂OH 1 (CH₂) CONa (CH₂)COH CH₂ONa CH₂OH This reaction gives two products. Show both.…
A: Requirement from question Six organic reactions and products
Q: A 1.00 liter solution contains 0.43 M potassium cyanide and 0.56 M hydrocyanic acid. If 0.14 moles…
A: Here 0.14 moles of Ca(OH)2 is added to 1L solution containing 0.43 M KCN and 0.56 M HCN. We have to…
Q: What is the percent ionization of a 2.50 M solution of benzoic acid? C6H5COOH K₂ = 6.46 x 10-5 [?] %
A:
Q: 0 F CH₂1 Br Br
A: Here we have to write predict E- configuration of the following given alkenes.
Q: Q/ differentiate in summary between
A: There is an equal free water level which is altitude in which pressure of water and hydrocarbon…
Q: Give the product. O a.. O b.. Oc.. O d.. O e.. Of.. Br₂ (2 eq) (a) (b) A Moving to another question…
A:
Q: 4 3 2 1 Example: An equimolar liquid mixture of benzene (B) and toluene (T) at 10 °C and 1 atm is…
A: Solutions An equimolar liquid mixture of benzene and toluene..
Q: HA + NaOH → → NaA + H₂O Kelly split a 50.0 mL solution of weak acid into two separate 25.0 mL parts.…
A: In this question a monoprotic acid is being titrated to NaOH base. The relation between pH and pKa…
Q: 26. The compound that would react immediately with aqueous bromine, resulting in a colourless…
A: Alkene reacts with liquid bromine and the double bond breaks, Br atom gets attached to each carbon.…
Q: Choose the reagent necessary to carry the following reaction: 8-8 O H₂S in water NaSH in Water NaSH…
A:
Q: What is the pH of the combined solution? [HBr] = 0.083 M [HBrO] = 0.17 M Ka (HBrO) = 2 x 10-9 pH =…
A:
The answer is not a but I'm not sure what the answer is. Can you solve it please?
Step by step
Solved in 2 steps

- The separation and purification processes given below and the method used against them are given. Which or which of these pairings are correct? I. Brewing of tea - ExtractionII. Purification of water impurities - ChromatographyIII. Separation of olive pomace from olive oil while producing olive oil-DecantationIV.Petroleum fractions obtaining - Ordinary distillationV. Obtaining essential oils and using them in perfume making - Water-steam distillationQ2. AnursegavelVtoapatientwith0.10MCaCl2 solution.Whatwillhappentothepatientsbloodcells?(3) (A) BehaveNormal (B) Dieoffduetohemolysis (C)Dieoffduetocrenation(D)CryingforhelpPlease tell me how to get this answer (step-by-step) including equations used. Thank you! 40 mL of a 35% v/v acetic acid solution is needed. In the stock room is a bottle of 50% v/v aceticacid. How much water should be used as the diluent?ANS: 12 mL of water is needed
- Please label the parts of the two cleavengers below:A 0.9000-g of KIO3 is dissolved in 500-mL distilled water. A 50.0-mL aliquot portion was run down into an Erlenmeyer flask and 2-g of KI and 2-mL of 6M HCl were added to it. Directly after, the solution was titrated with a 24.80-mL Na2S2O3 solution to a faint yellow solution. Starch TS was added to the resulting solution and the titration with the Na2S2O3 solution continued wherein an additional 1.33-mL was required to bring the blue color of the solution to disappear completely. MW: KIO3 = 214.0 g/mol ; Na2S2O3 = 158.1 g/mol Compute the N of the Na2S2O3 solution. None of the choices 0.09657 N 0.1075 N 0.1017 NPlease don't provide hnadwrritten solutrion .....
- Given the Ksp = 1.4 x 10-13, what is the solubility of CoCO3 (i.e., what is the [Co2+] concentration) in water?There are two pH values in which Glutamic acid will have an a complete net charge of 2.17? what is the lowest PH1 and highest of pH2 round your amswers to the nearest 0.1 no orher information was providedKsp for BaCrO4 is 1.17x10^-10
- Calculate the volume of hydrogen at 0°C and 1.00 atmthat is required to convert 500.0 g linoleic acid (C18H32O2)to stearic acid (C18H36O2). (See Sec. 9.1.)Determine the branch points and reducing ends of amylopectin. A 0.5g sample of amylopectin was analyzed to determine the fraction of the total glucose residues that are branch points in the structure. The sample was methylated and then digested, yielding 50 μmol of 2,3-methylglucose and 0.4 μmol of 1,2,3,6-tetramethyl glucose. (MWglucose = 162 g/mol, minus H2O) What fraction of the total residues are branch points? How many reducing ends does the amylopectin have?https://www.youtube.com/watch?v=2EQznGPZY5A

