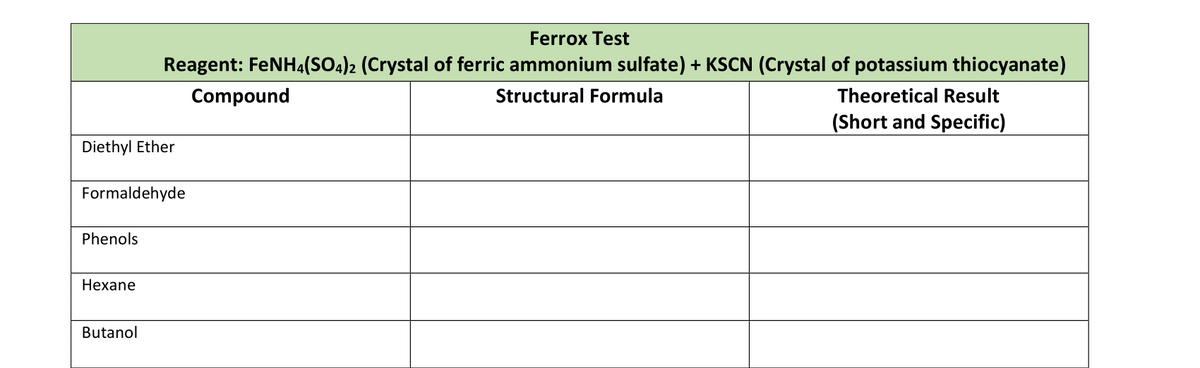
Ferrox Test Reagent: FENH4(SO4)2 (Crystal of ferric ammonium sulfate) + KSCN (Crystal of potassium thiocyanate) Compound Structural Formula Theoretical Result (Short and Specific) hyl Ether naldehyde nols
Q: de Sn2 attack?
A:
Q: 10. Which of the following substances has the highest concentration of hydrogen ions in solution? F)…
A:
Q: What are reactants will form the enamine shown? For the following molecules classify them as…
A:
Q: 37 The average pH of normal arterial blood is 7.40. At normal body temperature (37°C), K, = 2.4 ×…
A:
Q: asuodsais aAes atropy incre ase or decrease for each of the following proceases? ng coal In burning…
A: Entropy of a system can be defined as the degree of disorder of a system.Entropy may be positive ,…
Q: The enthalpy of vaporization of water is 43.21kJ/mol and normal boiling point temperature is…
A:
Q: What is the indicator of the end point for the Fajans method? A green fluorescence (B white…
A: Fajans Method as,the precipitation titration in which silver ion is titrated with Halide or…
Q: CH3COOH + NaOH --> CH3COONa + H2O Write an equation showing the reaction of each carboxylic acid…
A: Given that - Carboxylic acid, ethanoic acid is treated with sodium hydroxide to give sodium acetate…
Q: drug passing throughn a 1-mm-thick membrane has a diffusion coefficient f 4.23 10-7 cm? per second.…
A: The answer is attached below-
Q: 4 Would you expect the entropy to increase in each of the following reactions? Explain your answer.…
A: Entropy: Entropy can be defined as the measurement of randomness or disorderness in the…
Q: Calculate the following values and indicate what type of electromagnetic radiation is present. (a)…
A:
Q: Quercetin, a flavonoid found in fruit juice, was quantified by UV-Vis using external calibration.…
A:
Q: Below is a selection of reactions; you may not need all of them. Write the letters of the needed…
A:
Q: An instrument, maintained at 165.2 °C, has two separate flasks that contain different gases. If the…
A: Here two different gases enclosed in two different vessel are connected by a narrow tube are mixed…
Q: Describe the difference between an aldehyde and a ketone, and indicate how each differs from an…
A: Both aldehydes and ketones are carbonic chemicals containing a carbonyl group. A carbonyl group…
Q: PPh + R. +R. PPh RBr oc- Br Ph,P PhP čo explain how's the hybridization? prove your answer
A: To solve this question, it is quite important to determine the oxidation state and the proper…
Q: The mole percent of methane to water in clathrate compounds that contain methane hydrate is 1:5.75.…
A:
Q: A buffer solution contains 0.150 moles of HSO4° and 0.150 moles of SO,²² in a total volume of 0.250…
A: Given: HSO4- = 0.150 moles SO42- = 0.150 moles volume of solution = 0.250 L pKa = 1.90 To…
Q: What concentration of NaF will precipitate BaF2 from a solution that is 0.1045M Ba2+?
A: We would use Ksp value of BaF2 and given Concentration of Ba2+ to calculate Concentration of F- ion…
Q: Balance the following Redox reaction in acidic media. VO2" (aq) + Sn(s) vo²+ (aq) + Sn²+, (aq)
A: Here we are required to balance the redox reaction
Q: Question 10 will be released by the reaction of erergy the following reaction: How much of 235U by…
A:
Q: What is the normal concentration of a 12.0M phosphoric acid solution? 2. How would you prepare a…
A: The relationship between Normality (N) and Molarity (M) for an acid solution is N = basicity×M…
Q: calculate iodine value . Fatty acid Molecular Formula % 16:0 C16H32O2 13.2 16:1 C16H3002 0.2 18:0…
A: The iodine value of an oil is a measure of the degree of unsaturation which is defined as the number…
Q: Balancing Equations Practice Balance each of the following chemical equations. Directions: You are…
A:
Q: Name this compound according to the system we went over in lecture: H2C Na OH
A: Namming of the organic compound can be done according to IUPAC nomenclature. First number the…
Q: "When 0.774g sample of C7H 5N 30 6 is burned in a bomb calorimeter, the temperature increases from…
A: Given ,that the sample=0.774g of C7H5N3O6 is burned in a bomb calorimeter Temperature increases from…
Q: Titanium(IV) and vanadium(V) form colored complexes when treated with hydrogen peroxide in 1 M…
A:
Q: 16.73 Ephedrine, a central nervous system stimulant, is used in nasal sprays as a decongestant. This…
A:
Q: The triphenylmethyl radical is highly delocalized by resonance. In the resonance hybrid, how many…
A: There are ten different positions have radical character
Q: 5 ml of a 0.2 M NH3 solution are added to 5 ml of a 0.1 M NHẠCI solution (Kb for NH3 is = 1.8 ×…
A: Dear student, As yoy have asked multiple part questions but according to guidelines we will solve…
Q: Aspirin is a weakly acidic drug with a pka of= 3.4 What will be the ionisation state of the drug in…
A: Aspirin is an acidic drug with acidic, -COOH group whose pKa is 3.4. Deprotonation of an acidic…
Q: Which is NOT belong to the group
A: There are various types of intermolecular forces present in the molecules such as hydrogen bonding,…
Q: For a certain chemical reaction, the standard Gibbs free energy of reaction at 30.0 °C is – 147. kJ.…
A:
Q: 26 1 3. 12M9+On → le+
A:
Q: Question I1 For the reaction at 25°C:Cd)C Determine E° De ter mine AG° De termine K
A: Equilibrium constant of a reaction can be defined as the ratio of concentration of products to…
Q: A 2.25 mol sample of an element has a mass of 51.8 g. What is the element?
A:
Q: Na20 CO SiC SO2
A: An ionic compound is formed when a cation and ion combine via electrostatic force of…
Q: Mass of Inner cup with stirrer on the digital balance=36.7 Mass of Inner cup with stirrer and warm…
A: Given :- Mass of Inner cup + stirrer(M') = 36.7g Mass of Inner cup + stirrer + warm water(M") =…
Q: Question 17 For the following reaction at 25°C: 2C2H2+ 502-4C02+ 2H20 www Calculate AH -2600.4KJ…
A: here we are required to find the enthalpy of reaction ,entropy of reaction and Gibbs free energy of…
Q: 9. The table below shows the pH values of several unknown substances. Substance pH W 5.8 14.0 Y 7.0…
A:
Q: Calculate the following values and indicate what type of electromagnetic radiation is present. (a)…
A: Here we are required to find the following value and the type of electromagnetic radiation
Q: Calculate both saponification and iodine value? Fatty acid Molecular Formula 16:0 C16H32O2 13.2 16:1…
A: The saponification value is the number of milligrams of potassium hydroxide required to neutralize…
Q: Calculate the equilibrium constant for the overall reaction at this temperature. 2) Calculate…
A:
Q: Rank the following dienes in order of increasing reactivity in a Diels-Alder reaction.
A: In this question we have to tell the reactivity of diene in Diels Alder reaction.
Q: Show the structure of all alcohols with the molecular formula C5H11OH. Identify the ones that are…
A:
Q: The name of the product of this reaction is:
A: In this question we have to tell the name of the reaction.
Q: g)A chemical reaction has ΔH° = +250kJ/mol. Which of the following statements is/are true concerning…
A: ∆H° sign is positive that means Reaction is Endothermic because energy is absorbed by the system.…
Q: A chemist dissolves 601. mg of pure barium hydroxide in enough water to make up 190. mL of solution.…
A:
Q: 3
A: Starting with optically pure R or S-2-butanol which method below would give the best yield of the…
Q: В. CH;-0-C-(CH,)|6–CH3
A: A fat molecule contain two main groups one is Glycerol and other is fatty acid .In Triacylglycerol…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- 1. Which of the following compounds can form hydrogen bonds with water?a. octaneb. HClc. NaCld. acetic acid2. Maria wants to determine the percent water content of an unknown hydrate of AB. Given the following data:mass of empty crucible - 12. 2784gmass of AB · xH20 + crucible - 17.4960gmass of AB + crucible - 15.1314gCalculate the % water content of the hydrate3. From the above data, give the correct formula for the hydrate. (AB₂ Molar mass = 129.84 g/mol)1-A bottle of Na2EDTA.2H2O is labeled with an assay of 100.5%, what is this implied?What is the result of heating to 100 ºC?CH.8 #8 - Please fill in the blank, cirlce your answer for thumbs up!
- 4g 5 g 2 g 100 g 49." R Lactic acid Salicylic acid Trichloroacetic acid Flexible collodion qs ad Sig: wart remover. Use as directed. (a) Flexible collodion contains 20% w/w camphor and 30% w/w castor oil. How many grams of each would be contained in 30 g of the mixture? (b) The specific gravity of castor oil is 0.955. How many milliliters of the oil is contained in 30 g of the mixture? (c) If the specific gravity of the mixture is 0.781, what are the percent w/v con- centrations of lactic acid, salicylic acid, and trichloroacetic acid in the mixture?2-Methylcyclohexanol (5 mL, d=0.929g g/ml, MW= 114.19) was heated under reflux with 5mL of 85% phosphoric acid. What is the theoretical yield of the product for this reaction? a) 4.5 g b) 3.9g c) 3.2g d) 2.9g e) 2.5gA student runs a TLC of a sample using a nonpolar development solvent. The TLC shows one spot. The MP of the same sample is 115.5 - 118.2 oC. What can we conclude from the experimental results. Group of answer choices The incorrect development solvent was used. The sample is pure. The sample is non polar. The sample is polar.
- At -28 C sodium bromide and water from a eutectic mixture which contains 48 % NaBr by weight. What is the maximum weight of pure NaBr that can be recovered from a mixture containing 80 g NaBr and 20 g water? a. 33.3 g b. 28.6 g c. 66.6 g d. 40 gWhat is the simplest CGS unit for "poise"?Given the information listed below, identify the compound. Physical description: This substance is a colorless but fragrant liquid that is oily in appearance. Chemical information: Observed Melting point: -78.5 °C Observed Boiling point: 138-142 °C Calculated MW: 130 g/mol Density: 0.876 g/cm3 Solubility in water: Minimally soluble, Other: Flash Point: 77 °F Spectroscopic information: IR bands indicate this compound contains C-H functionalities and the appearance of a strong stretch near 1700 cm-1 as well as several strong bands between 1500 and 1000 cm-1. The C-13 NMR spectrum for this compound indicates 6 unique carbon atoms and no cyclic structure. The proton NMR data indicate 5 sets of unique protons. C-13 NMR data of : PPM: 171.08, 63.13, 37.48, 25.18, 22.51, 20.96 Int: 222, 887, 645, 635, 1000, 473 Assign: 1, 2, 3, 4, 5, 6 H-1 NMR Assign: A, B, C, D, E Shift: 4.095, 2.037, 1.693, 1.521, 0.926
- In what order should the following methods be used to separate a mixture containing sand-sugar-alcohol and water into its components? 1) Adi distillation II) Fractional distillation HII) Filtration under vacuum B. II, I, II C. III, I, II D. 1, III, II E III, II, IPlant pigments in spinach were separated with TLC with cellulose as thestationary phase and 80/20 diethyl ether/acetone mixture as themobile phase. If needed, the solvent structures in the lab Appendix.1. Determine the Rf values of each plant component on this TLC plate.2. Arrange the plant components from most polar to most nonpolar. Please answer both 1 and 2 (picture below is the same question attached )STARTING WITH 18ML OF 2-methyl-2-butanol

