Make Ammonia: N₂ + H₂NH3 Separate Water: H₂O H₂+0₂ Combust Methane: CH₂ + O₂ CO₂ + H₂O Determine for which elements the specified atoms are balanced or unbalanced in each of these chemical equations when every substance in the reaction is given a coefficient of one (1) Use the PbET cimula this meables
Make Ammonia: N₂ + H₂NH3 Separate Water: H₂O H₂+0₂ Combust Methane: CH₂ + O₂ CO₂ + H₂O Determine for which elements the specified atoms are balanced or unbalanced in each of these chemical equations when every substance in the reaction is given a coefficient of one (1) Use the PbET cimula this meables
Introductory Chemistry: A Foundation
8th Edition
ISBN:9781285199030
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter6: Chemical Reactions: An Introduction
Section: Chapter Questions
Problem 48AP
Related questions
Question
One of them is sorted incorrectly
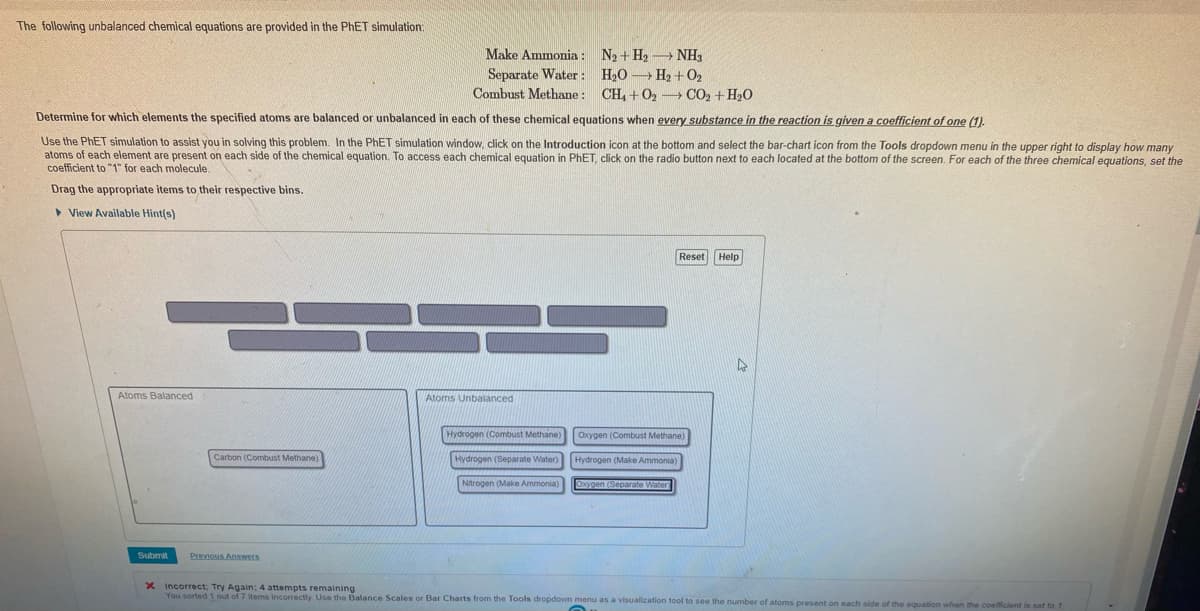
Transcribed Image Text:The following unbalanced chemical equations are provided in the PhET simulation:
Determine for which elements the specified atoms are balanced or unbalanced in each of these chemical equations when every substance in the reaction is given a coefficient of one (1).
Use the PhET simulation to assist you in solving this problem. In the PhET simulation window, click on the Introduction icon at the bottom and select the bar-chart icon from the Tools dropdown menu in the upper right to display how many
atoms of each element are present on each side of the chemical equation. To access each chemical equation in PhET, click on the radio button next to each located at the bottom of the screen. For each of the three chemical equations, set the
coefficient to "1" for each molecule.
Drag the appropriate items to their respective bins.
View Available Hint(s)
Atoms Balanced
Submit
Carbon (Combust Methane)
Make Ammonia: N₂ + H₂NH3
Separate Water: H₂O H₂+0₂
Combust Methane: CH₂ + O₂ CO2 + H₂O
Previous Answers
Atoms Unbalanced
Hydrogen (Combust Methane)
Hydrogen (Separate Water)
Nitrogen (Make Ammonia)
Reset Help
Oxygen (Combust Methane)
Hydrogen (Make Ammonia)
Oxygen (Separate Water
4
X Incorrect; Try Again: 4 attempts remaining
You sorted 1 out of 7 items incorrectly. Use the Balance Scales or Bar Charts from the Tools dropdown menu as a visualization tool to see the number of atoms present on each side of the equation when the coefficient is set to 1.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning