Only five general types of reactions are commonly observed in biochemical transformations. Given the proposed mechanism (illustrated above) for Class A β-lactamases, what is the biochemical reaction type represented by steps 1 and 2?
Only five general types of reactions are commonly observed in biochemical transformations. Given the proposed mechanism (illustrated above) for Class A β-lactamases, what is the biochemical reaction type represented by steps 1 and 2?
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter18: Glycolysis
Section: Chapter Questions
Problem 18P: Distinguishing the Mechanisms of Class I and Class I Aldolases Fructose bisphosphate aldolase in...
Related questions
Question
Only five general types of reactions are commonly observed in biochemical transformations. Given the proposed mechanism (illustrated above) for Class A β-lactamases, what is the biochemical reaction type represented by steps 1 and 2?

Transcribed Image Text:O oxidation/reduction
O carbonyl condensation
O a, B-elimination
nucleophilic acyl substitution
nucleophilic addition
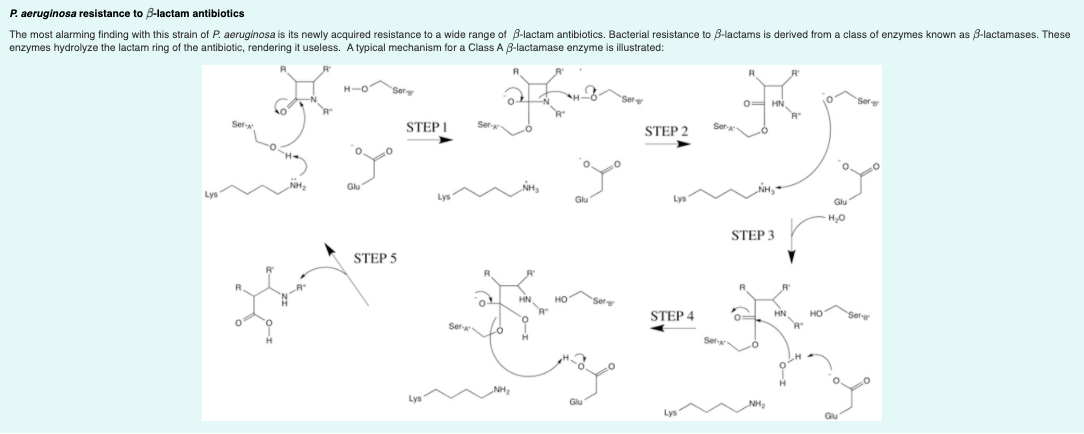
Transcribed Image Text:P. aeruginosa resistance to B-lactam antibiotics
The most alarming finding with this strain of P. aeruginosa is its newly acquired resistance to a wide range of B-lactam antibiotics. Bacterial resistance to 8-lactams is derived from
enzymes hydrolyze the lactam ring of the antibiotic, rendering it useless. A typical mechanism for
class of enzymes known as 8-lactamases. These
Class A 3-lactamase enzyme is illustrated:
R
H-o Ser
0= HN
STEP I
STEP 2
Ser
NH,
NH,
Glu
Lys
Glu
H20
STEP 3
STEP 5
R
но
Ser
STEP 4
HN
но
Ser
Ser
Glu
NH,
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning