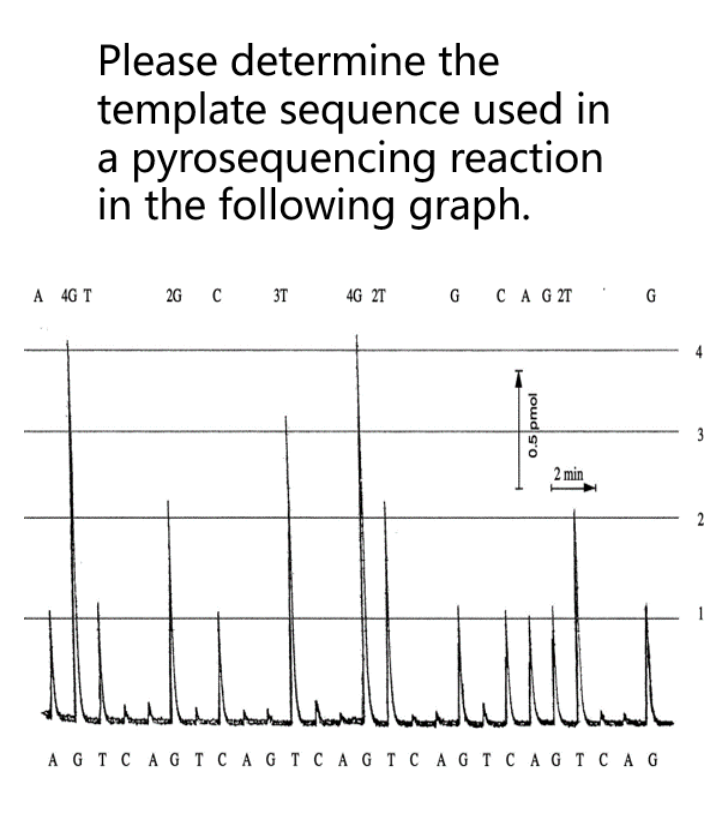
Please determine the template sequence used in a pyrosequencing reaction in the following graph. A 4G T 2G C 3T 4G 2T G CA G 2T 3 2 min 2 AG T CAG T C A G T C A G T C A G T C A G T C A G joud s'o
Q: Use arrow-pushing to show how the glycerol phosphate reacts with the base :8 and NAD* to form DHAP.…
A: Cellular respiration is a catabolic pathway of the process of metabolism, where a series of chemical…
Q: complete the following mechanism showing the cleavge of the peptide bond for the following cysteine…
A: ghhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhf vcxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
Q: abbreviation, CoA, stands for
A: Coenzyme A is synthesized from Vitamin B5, that is rich in foods, such as meat, vegetables, cereal…
Q: Describe the pyrimidine biosynthesis pathway
A: Pyrimidines are the nitrogenous base of DNA & RNA. For DNA , pyrimidines are Thymine , Cytosine…
Q: The role of Zn2+ in catalysis is usually to: a. Stabilize a (-) charge in the transition state Ob.…
A: Proteins include zinc, which is either essential for preserving protein stability and structure or…
Q: Circle and label one place on the mechanism where covalent catalysis is occurring. Suggest an amino…
A: Glycolysis is a catabolic breakdown of Glucose into pyruvate which consist of 10 metabolic steps. so…
Q: The amino acid histidine has been found to participate in both acid and base catalysis in many…
A: Histidine is a basic amino acid and it contains an imidazole ring in its side-chain. Histidine is…
Q: What type of reaction does Ribulose-5-phosphate isomerase catalyze? A) isomerization B) not enough…
A: Ribulose 5-phosphate is a substrate for two enzymes includes 1) ribulose 5 phosphate epimerase…
Q: Predict the locations of 14C in Aspartate synthesis using the following labeled 14C succinate.
A: Given, Structure of radio label succinate: -OO14C-CH2-14CH2-14COO- Succinate is an intermediate of…
Q: Considering the general structure of penicillins, how are they rendered ineffective? Select…
A: Penicillin is an antibiotic that inhibits the formation of peptidoglycan cross-links of the…
Q: Consider the metabolic pathway show below that converts substrate A to B with the enzyme A-ase, B to…
A: For regulation of A-ase: Feedback inhibition refers to the inhibition of the enzymes's activity by…
Q: Is It Possible to Design an Enzyme to CatalyzeAny Desired Reaction?
A: Enzymes catalyze thermodynamically favorable reactions causing them to proceed at extraordinarily…
Q: Methyl iodide (CH31) can be used to modify DNA in solution. It takes the following mechanism for…
A: DNA DNA is a heritable molecule that carry all necessary information of the organism.
Q: Using Fischer projections, complete the reaction equation for this transformation. racemase HN-H ?…
A: Introduction: L-alanine is converted to D-alanine by the action of the enzyme alanine racemase.
Q: Show how you would accomplish the following synthetic transformations. Show all intermediates.(a)…
A: Synthetic Reagents have a broad range of chemicals with multiple uses in chemical synthesis. They…
Q: help fill in the word between brackets Serine proteases are an example of…
A: Serine protease are the enzyme which causes cleavage of peptide bonds in protein . They are called…
Q: The rate constant for the uncatalyzed reaction of two molecules of glycine ethyl ester to form…
A: Given values: Rate constant in the uncatalyzed reaction = 0.6 M-1s-1 In the presence of catalyst…
Q: explain the biochemical processes involved in each of the following, in named cells. Start by…
A: 1. Homolactic fermentation-(production of lactic acid) Purpose- Any form of fermentation that…
Q: If photosynthesizing green algae are provided with CO2 containing heavy oxygen (180), which of the…
A: The Calvin cycle is the light-independent chemical reaction in which carbon dioxide is fixed to…
Q: In addition to the reactions mentioned in Section 23.5, PLP can catalyze b-substitution reactions.…
A: Pyridoxal phosphate (PLP) is a coenzyme for a variety of enzymatic biochemical reaction. PLP is the…
Q: Which of the following statements about ribozyme catalytic residues is INCORRECT? General acid…
A: Solution :- the ribozyme Catalytic residues which are RNA molecules and it's fuction as chemical…
Q: Consider the metabolic pathway show below that converts substrate A to B with the enzyme A-ase, B to…
A: Mechanism of Regulation of enzymes is tej process by which enzyme is controlled to catalyse the…
Q: Define the following terms:a. amphibolic pathwayb. anaplerotic reactionc. glyoxylate cycled.…
A: Since you have posted a question with multiple subparts, we will solve the first three subparts for…
Q: . The first two steps of ovothiol C biosynthesis are shown below. Propose the rest of the pathway,…
A: Ovothiol C is an antioxidant and a marine metabolite. It is synthesized from histidine and cysteine…
Q: Determine the type of biochemical reaction that occurs in the reaction shown below. O redox…
A: In the given question, a cyclic pyranose structure is given. It is a pyranose because it has 6…
Q: Identify each reaction catalyzed by (a) a nucleotidase; (b) a phosphorylase; (c) a…
A: Nitrogenous bases (adenine, guanine, thymine, cytosine) are used to form nucleotide phosphates by…
Q: Describe the mechanism of catalysis Thiamine Pyrophosphate (TPP), (Vit. B1) Pyridoxal Phosphate…
A: Introduction: The substance that increases the rate of the chemical reaction without changing itself…
Q: Help me, please
A: Proteases are enzymes that catalyze the breakdown of the peptide bonds in the protein. Serine…
Q: enzyme A enzyme B enzyme C compound 1 compound 2 compound 3 compound 4 Consider the biochemical…
A: The one gene one enzyme hypothesis is proposed by the Beadle and Tatum. George Beadle and Edward…
Q: One of the important features of enzymes is that they stabilize transition state of the reaction…
A: An enzyme is a type of biological catalyst that aids in the acceleration of chemical reactions.…
Q: Draw a stepwise mechanism for the following reaction. The fourmemberedring in the starting material…
A: DBN (C7H12N2) is used the synthesis f organic compounds. It is an amidine. It is a strong non…
Q: Given the active site diagram below, indicate the mechanism(s) of catalysis.
A: The given enzyme substrate complex diagram indicates that the enzyme is phosphatases and substrate…
Q: The key regulatory enzyme in pyrimidine synthesis is: a. Aspartate transcarbamoylase. b. Glutamine…
A: Pyrimidine synthesis takes place in cytoplasm. Pyrimidine is synthesized as a free ring and then a…
Q: Describe the function implied by the name of each of the following enzymes Citrate decarboxylase…
A: The enzymes are essentially the biocatalysts present in all living systems. Each enzyme catalyzes a…
Q: Give the name and abbreviation for the following molecule and list its 5' "0-P-0-CH2 sugar: `NH2 H H…
A: Answer. 1 The monomeric units of nucleic acids are called nucleotides. Nucleic acids, therefore,…
Q: For production of penicillin (C16H18O4N2S) using Penicillium mold, glucose (C6H12O6) is used as a…
A: Introduction :- The penicillin is an important antibiotic produced by the microorganism which helps…
Q: Enolase has a glutamic acid residue in its active site. Describe the experiment that was done that…
A: Enolase is a metalloenzyme used in the 9th step of glycolysis it other name is PG i.e…
Q: The following complex natural product has been biogenetically produced via a pericyclic reaction(s)…
A: Pericyclic reactions are a set of reactions involved during the transition to and from the…
Q: Define the following terms:a. oxidoreductaseb. lyasec. ligased. transferasee. hydrolasef. isomerase
A: These all that are mentioned or asked in the question are Enzymes. we can also identify it by just…
Q: The isoelectric point, pl, of the protein deoxyribonuclease I is 10.2 , while that of pepsin is 1.…
A: Amino acids contain at least two ionizable groups-alpha the carboxylic acid group and the…
Q: Table 4. Rates of saponification at 35°C. Ester k(M-' sec=!) k, relative* (CH,O),PO, (CH,O),P=O…
A: Introduction A phosphodiester bond is formed when two of the hydroxyl groups in phosphoric acid…
Q: The synthetic antiviral drug Tamifl u, currently the most effective agent against avian infl uenza,…
A: Tamiflu is an antiviral drug produces from the shikimic acid conversion to oseltamivir, obtained…
Q: Given the active site diagram below, indicate the mechanism(s) of catalysis. 5 HO OH HN OH *HN NH…
A: The figure in question shows the mechanism of formation of transition state. By analyzing the active…
Q: For production of penicillin (C16H18O4N2S) using Penicillium mold, glucose (C6H1206) is used as a…
A: The penicillin is an important antibiotic produced by the microorganism which helps in inhibiting…
Step by step
Solved in 2 steps

- Define the following terms:a. amphibolic pathwayb. anaplerotic reactionc. glyoxylate cycled. reduction potentiale. conjugate redox pairCalculate the net charge on the following tripeptides at pH 5.0: (a) Leu-His-Asp [0] (b) Ala-Ile-Val [0] (c) Met-Lys-Arg [+2] (d) Which tripeptide will be retained the shortest on a cation-exchange chromatographic column in a pH 5.0 buffer? Why?Consider the following equilibrium at 25ºC :Glucose-1-Phosphate Glucose-6-PhophateUsing the equilibrium concentrations of [Glucose-1-Phosphate] = 0.35 M and [Glucose-6-Phosphate] = 1.65 M, calculate BOTH K′eqand Gº′ for this reaction. Is this reaction exergonicor endergonic? R = 8.314 J/K·mol
- Utilising the provided class data generate the following graphs: I) Michaelis Menten; II) Lineweaver-Burk; and III) Hanes-Woolf. Ensure that you clearly label each graph,and add the relevant trendlines with equations. Table 1: Class data demonstrating the Absorbance at 700nm obtained for the alkaline phosphatase enzyme reaction Table 1 tube Abs700mm 1 0.000 2 0.060 2 0.090 4 0.140 5 0.190 6 0.250 7 0.290 The equipment we used are • 20mM Tris Buffer pH 8.5 • 33mM MgCl2 • Alkaline Phosphatase (2mg/ml) in 20mM Tris Buffer pH 8.5 • 4mM Glucose-1-phosphate • Acid Molybdate pH 5.0 • Reducing Agent • Distilled Water • Glass Test tubes • Tube Rack • Cuvette • Pipettes and Tips • Water bath set to 37oC The method we used is Method/Protocol: 1. Read the protocol in its entirety before starting. Take note of any additional information that appears in subsequent steps that may influence how previous steps are performed. 2. Using glass tubes, generate the reactions mixtures…Calculate the actual, physiological ΔG for the reaction at 37 °C, as it occurs in the cytosol of neurons, with phosphocreatine at 4.7 mM, creatine at 1.0 mM, ADP at 0.73 mM, and ATP at 2.6 mM.A) Is this reaction ( in picture provided) in equilibrium? B) If it is not then ,what is ∆G' at 25°C if the concentration of Glucose-1-phosphate is 15.04µM and the concentration of Glucose-6-phosphate is 1.62 mM? Answer in Joules. Round to the correct number of significant figures. (There are 103 µM in 1mM.) Thank you so Much!!!
- 1. a. Calculate the physiological DG of the reaction shown below at 37°C, as it occurs in the cytosol ofneurons, with phosphocreatine at 4.7 mM, creatine at 1.0 mM, ADP at 0.73 mM, and ATP at 2.6mM. The standard free energy change for the overall reaction is –12.5 kJ/mol. Phosphocreatine + ADP ® creatine + ATP b. The enzyme phosphoglucomutase catalyzes the conversion of glucose 1-phosphate to glucose6-phosphate. Calculate the standard free energy change of this reaction if incubation of 20 mMglucose 1-phosphate (no glucose-6 phosphate initially present) yields a final equilibrium mixtureof 1.0 mM glucose 1-phosphate and 19 mM glucose 6-phosphate at 25°C and pH 7.0. c. If the rate of a nonenzymatic reaction is 1.2 x 10–2 μM s–1, what is the rate of the reaction at 37℃ inthe presence of an enzyme that reduces the activation energy by 30.5 kJ/mol?Calculate the overall ΔG° (report up to two decimal places) for the net reaction (see attached image). Answer: _____ kcal/mol Note: R = 1.98 x 10 -3 kcal/mol-KUsing Figure 1.3 of the Introduction as an example, a) draw all the structures of the tribasic amino acid lysine involved in the equilibrium reactions that would take place during titration against NaOH, starting with the fully protonated form below (draw the R-group in full). HAN+-CH- COOH (CH2)4 NH°+ b) indicate the numerical pa value of each equilibrium reaction, and which ionizable group is being dissociated in each step. c) indicate the net charge of the amino acid at each step and identify the zwitterion. d) Calculate the pI of this amino acid (show the calculation). e) What would be the predominant ionization states of this amino acid at physiological pH (7.4) and at this pH, what would the ratio of these two states be (show the calculation)?
- Beginning with the 1st tetrahedral intermediate, show the complete steps in chymotrypsin mechanism that occurs to form the 2nd chymotrypsin intermediate in the chymotrypsin active site. The substrate for chymotrypsin to be used is Ala-Tyr-Gly. Further, name the amino acid(s) that would be released as a result of the reactions you'd illustrated above.Write a balanced equation for each of the following reactions or reactionsequences.(a) The reaction catalyzed by PFK-2(b) The conversion of 2 moles of oxaloacetate to glucose(c) The conversion of glucose to UDP-Glc(d) The conversion of 2 moles of glycerol to glucose(e) The conversion of 2 moles of malate to glucose-6-phosphateWrite a balanced equation for each of the following reactions or reaction sequences. (a) The reaction catalyzed by PFK-2 (b) The conversion of 2 moles of oxaloacetate to glucose (c) The conversion of glucose to UDP-Glc (d) The conversion of 2 moles of glycerol toglucose (e) The conversion of 2 moles of malate to glucose-6-phosphate



