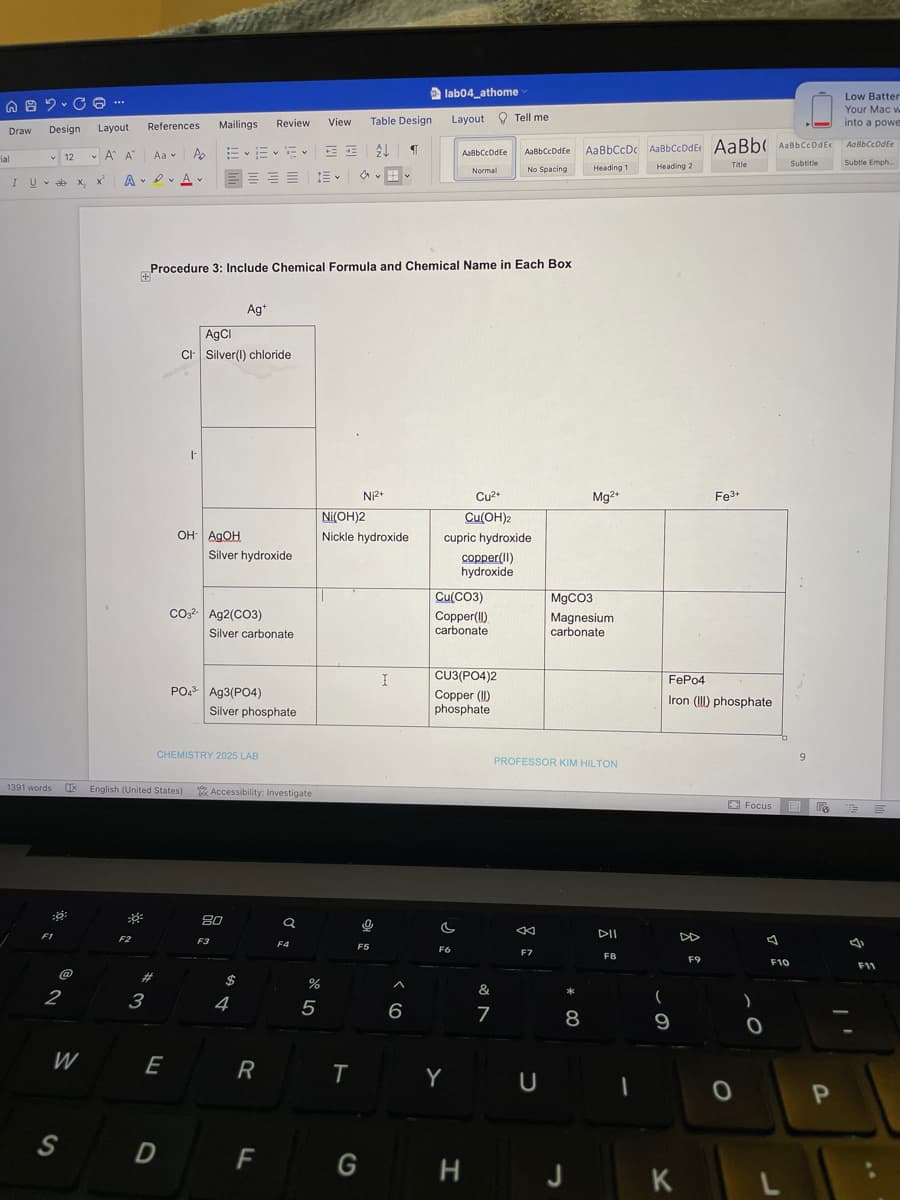
Procedure 3: Include Chemical Formula and Chemical Name in Each Box + Ag+ AgCl CI Silver(1) chloride F Cu²+ Cu(OH)2 cupric hydroxide OH- AgOH copper(II) hydroxide CO3²- Ag2(CO3) PO4 Ag3(PO4) Silver hydroxide Silver carbonate Silver phosphate Ni2+ Ni(OH)2 Nickle hydroxide I Cu(CO3) Copper(II) carbonate CU3(PO4)2 Copper (II) phosphate Mg2+ MgCO3 Magnesium carbonate Fe³+ FePo4 Iron (!) phosphate
Procedure 3: Include Chemical Formula and Chemical Name in Each Box + Ag+ AgCl CI Silver(1) chloride F Cu²+ Cu(OH)2 cupric hydroxide OH- AgOH copper(II) hydroxide CO3²- Ag2(CO3) PO4 Ag3(PO4) Silver hydroxide Silver carbonate Silver phosphate Ni2+ Ni(OH)2 Nickle hydroxide I Cu(CO3) Copper(II) carbonate CU3(PO4)2 Copper (II) phosphate Mg2+ MgCO3 Magnesium carbonate Fe³+ FePo4 Iron (!) phosphate
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter1: Chemical Foundations
Section: Chapter Questions
Problem 1RQ: Define and explain the differences between the following terms. a. law and theory b. theory and...
Related questions
Question
I need help completing this I just need the formula and names of each box, there are some that I did.
Transcribed Image Text:AB2-C6 ---
Draw
Layout
Design
12
ν Α' Α'
I Uab x, x
x₂
ial
1391 words IX English (United States)
::
30²
F1
@
2
lab04_athome
Review
Layout Tell me
References
Aa
~~~
AaBbCcDdEe. AaBbCcDc AaBbCcDdE: AaBb( AaBbCcDdEe
AaBbCcDdEe
Normal
Title
Subtitle
Heading 1
No Spacing
Heading 2
av
E
А ADA
Procedure 3: Include Chemical Formula and Chemical Name in Each Box
+
Ag+
AgCl
CI Silver(I) chloride
|
Cu²+
Cu(OH)2
cupric hydroxide
OH AQOH
copper(11)
hydroxide
CO32- Ag2(CO3)
PO4³- Ag3(PO4)
CHEMISTRY 2025 LAB
W
S
F2
#
3
E
D
Po
Mailings
Silver hydroxide
Silver carbonate
Silver phosphate
Accessibility: Investigate
80
Q
F3
$
4
R
F
F4
%
5
View
三
Ni2+
Ni(OH)2
Nickle hydroxide
T
Table Design
¶
F5
G
0
I
6
Cu(CO3)
Copper(!!)
carbonate
CU3(PO4)2
Copper (1)
phosphate
c
F6
Y
H
&
7
Mg2+
MgCO3
Magnesium
carbonate
PROFESSOR KIM HILTON
<K
DII
F7
FB
U
*
8
J
1
Fe3+
FePo4
Iron (III) phosphate
(
9
K
F9
0
9
Focus E
F10
)
0
L
FA
P
Low Batter
Your Mac w
into a powe
AOBBCCDdEe
Subtle Emph.....
E
F11
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9780078021558
Author:
Janice Gorzynski Smith Dr.
Publisher:
McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Elementary Principles of Chemical Processes, Bind…
Chemistry
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY