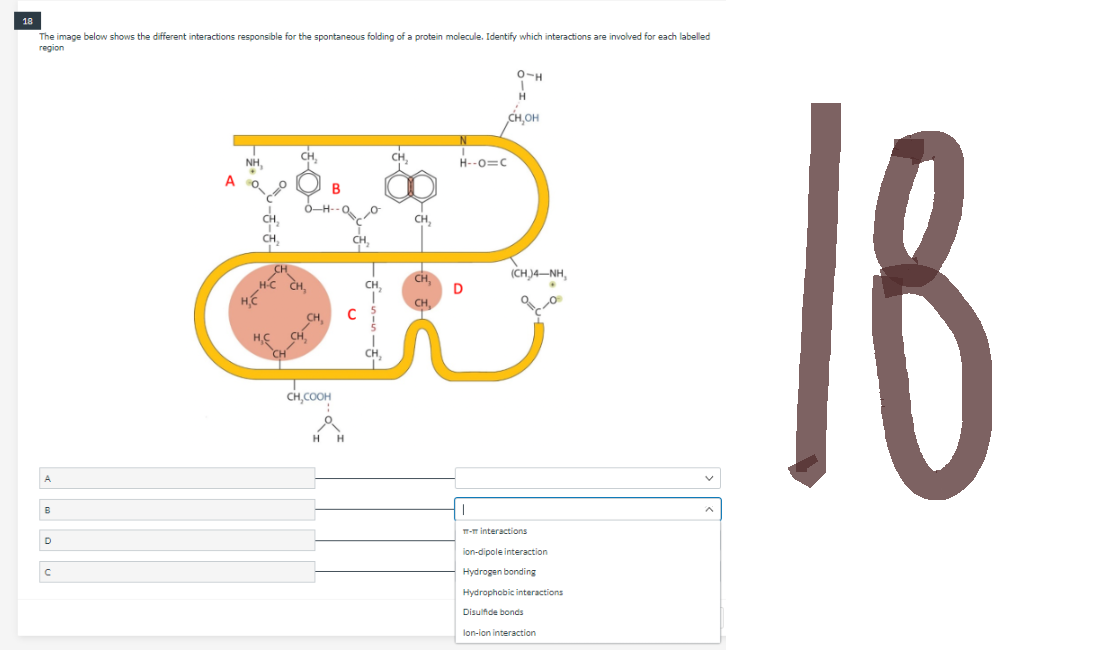
The image below shows the different interactions responsible for the spontaneous folding of a protein molecule. Identify which interactions are involved for each labelled region 0-H 1 H CH,OH NH, B A B с A CH H-C H.C CH, CH,COOH H H CH₂ C CH, CH, H--O=C D KCHJANH, m-m interactions ion-dipole interaction Hydrogen bonding Hydrophobic interactions Disulfide bonds lon-ion interaction
Q: II. ATP ACCOUNTING Provide what is being asked for. Show all relevant calculations and summarize…
A: 1 mole of lactose is hydrolyzed to produce 1 mole of glucose and 1 mole of galactose. Metabolism of…
Q: 2. Below is an image of the NADH cofactor, labeled A. (a) What role does this cofactor play, and…
A: NADH is vital in to metabolism and especially to the reactions that occur during release of energy…
Q: For the binding of a ligand to a protein, what is the relationship between the Ka (association…
A: The ligand in protein-ligand binding is typically a molecule that generates a message by connecting…
Q: Consider a 17:0 fatty acid in a mammalian cell where propionyl CoA is completely oxidized. For each…
A: β-oxidation of fatty acid occurs in mitochondria. Before β- oxidation, activation of fatty…
Q: feedback inhibition. Which step must be inhibited to regulate cholesterol synthesis?
A: The question is about the biomolecule synthesis, i.e cholesterol. What are the steps required for…
Q: What is the similarity among eicosanoids, cholesterol and phospholipids in terms of their…
A: Eicosanoids are biologically active molecules, which act as mediators of inflammation, regulators of…
Q: How does placing the solution in a boiling bath terminate the hydrolysis reaction mediated by…
A: The protein enzyme α-amylase is a key player in the hydrolysis reaction of starch molecules.
Q: At pH-4, what is the net charge of the peptide GKPAD? O+0.5 O-1.0 Oo -0.5
A: The sequence of the given peptide is as follows: Gly-Lys-Pro-Ala-Asp The amino acids are composed of…
Q: What is Bio Chemistry? Explain.
A: Biochemistry- Can be used for studying properties of the biological molecules.
Q: What is the Reverse transcriptase (RT) inhibitors? Explain with an example and describe its…
A: Question 1: Introduction: Reverse transcriptase (RT), also known as RNA-dependent DNA polymerase,…
Q: The "energy balance model" of body mass is based on the idea that biological systems neither create…
A: Energy intake, energy expenditure, and energy storage are the three basic components of energy…
Q: 5. cis 18:3(46,9,12) 18:3;6,9,12 w6,9,12 6. trans 18:3(46,9,12) 18:3;6,9,12 w6,9,12
A: Fatty acids are termed as cis and trans based on the position of hydrogen atoms on double bonded…
Q: Effects of low NADP+ levels in the non-cyclic light reactions of photosynthesis? (Increase,…
A: Photosynthesis can be divided into two stages: the Calvin cycle and light-dependent processes. At…
Q: Name the given substance. What pharmacological group does it belong to? CI -OH
A: The answer to the above question is provided below.
Q: What is the relationship between Warburg effect and oncogenesis? Explain why cancer cells have…
A: The Warburg effect is connected to increased glycolysis as a consequence of increased expression of…
Q: The following strand of DNA is transcribed: 5'-GACCTCCGAATGC-3' Write the sequence of the…
A: Transcription- formation of mRNA from DNA In , RNA A pairs with U G pairs with C
Q: 6. Succinate dehydrogenase couples the activity of the citric acid cycle with that of the electron…
A: Succinate dehydrogenase is an enzyme located in the inner mitochondrial membrane. It has FAD as its…
Q: Use the table below to identify the oil sample being described/ asked. Oil sample % Fatty acid by…
A: Unsaturated fatty acids are the fatty acids with one or more double bonds. Iodine is a halogen that…
Q: 111 The disease is caused by single amino acid mutation in the beta globin chain of hemoglobin and…
A: The single amino acid mutation can cause amino acid to change and thus can result in any disease or…
Q: why is energy released when IMFs form in between molecules as they approach eachther
A: In molecular terms, intermolecular forces are the forces of attraction and repulsion between…
Q: Do you think globalization affects the environment? Why or why not? Support your answer with…
A: Globalization is a process in which people of different countries or continents are interacting with…
Q: A3 Glucosylacetamide has a structure similar to the transition state during catalysis by the enzyme…
A: The chemical compounds that have structure similar to that of transition state substrate are called…
Q: If a particular 180 amino acid polypeptide is known to sample 3 preferred states as it folds into…
A: Anfinsen's experiment showed that the protein is intrinsically capable of finding its lowest-energy…
Q: Please, explain and write about what type of evidence, biochemically, led to the cyclic nature of…
A: The glyoxylate cycle is an anabolic mechanism found in fungus, bacteria, plants, protists, and…
Q: How do nuclear import receptorsnegotiate the tangled gel-like interior ofa nuclear pore complex so…
A: Nuclear pore complex is a large complex main up of protein, this complex allows small molecule and…
Q: Lipids like nucleic acids and proteins are made of strings of similar subunits. True or false.…
A: The four classes of biological macromolecules are lipids proteins, nucleic acids and carbohydrates.…
Q: Consider the mRNA sequence below. Assume that the following mRNA segment has been translated.…
A: The genetic code is a set of three-letter combinations of nucleotides called codons, each of which…
Q: Bile acids are detergents made by the liver O True O False
A: Bile acids is the product secreted by liver by found in bile juice. Bile juice is stored in gall…
Q: Question:- What is an example of a heterotrimeric G protein and what role does it play?
A: G proteins are guanosine binding proteins. That is, they can bind to GTP and GDP. Monomeric G…
Q: Which polysaccharide is the most common storage form in animals> Why is this so?
A: A polysaccharide is a long chain of carbohydrate molecules. They are typically composed of multiple…
Q: ENZYME/PATHWAY 1. phosphofructokinase-1 2. pyruvate dehydrogenase complex 3. Krebs cycle 4. lactic…
A: Introduction Phosphofructokinase 1(PFK1)is a kinase enzyme that phosphorylates fructose…
Q: 2. A 4-month-old child has signs of rickets. Digestive disorders were not noted. The child has had…
A: Calcitriol (1,25-DHCC) is the biologically active form of vitamin D. It regulates the plasma levels…
Q: intrinsic factor is serected by intrinsic factor O Parietal cells O Chief cells O G cells O
A: Structure of Intrinsic factor: Genuine Cbl is identified by an inherent component detected in…
Q: The pyruvate dehydrogenase (PDH) complex gates the entry of acetyl-CoA into the citric acid cycle,…
A: Under aerobic condition carbohydrates, fatty acids and most amino acids enter Krebs cycle through…
Q: 20 2
A: Microscope : This is an image of simple microscope, that has a magnifying glass that has double…
Q: How does PFGE separate larger fragments more efficiently than standard electrophoresis? 2. Why is…
A: Gel electrophoresis is a analytical method for characterization of DNA and RNA and the visualization…
Q: 3 from left to right. The table below shows which mRNA codons code for each type of amino acid.…
A: Genetic information in our body is stored in form of DNA. DNA multiples itself by replication. DNA…
Q: All steps in the calculations must be reported, incomplete reporting of these leads directly to…
A: Acetyl coenzyme is classified as a type of allosteric activator of the enzyme referred to as…
Q: 5. Dipeptide a. Draw a dipeptide with one polar amino acid connected to one nonpolar amino acid b.…
A: Peptides and polypeptides are linear polymers made up of amino acids. A dipeptide is composed of two…
Q: 1. ATP ACCOUNTING, Provide what is being asked for. Show all relevant calculations and summarize…
A: In beta oxidation of saturated fatty acid, First step produces FADH2 catalyzed by Acyl CoA…
Q: Show the reaction mechanism for the catabolism of proline.
A: Proline plays an important role in metabolism and is being recognised as an important amino acid in…
Q: Tumor cells often lack an extensive capillary network and must function under conditions of limited…
A: Tumor cells show uncontrolled growth. Tumor cells do not differentiate into various types of cells.…
Q: Please answer fast In an enzyme-catalyzed reaction, the rate of the reaction depends on which of…
A: Enzymes are biocatalysts which increase the rate of biochemical reactions. The rate of an enzyme…
Q: Arrange each compound according to its solubility in water. Assume that each acylglycerol contains…
A: Hydrophobicity is the association that is observed if nonpolar groups or molecules are placed in an…
Q: Explain the biochemical principle behind the separation of carbohydrates molecules by TLC as…
A: Thin Layer Chromatography (TLC) demonstrated the separation of each component as spots. Each…
Q: When is fatty acid synthesis highest? When there is excess acetyl CoA in the mitochondria. When…
A: Fatty acid synthesis occurs in the cytoplasm of the cell. The precursor molecule in the fatty acid…
Q: Please state if the statements are true or false. 1. An l-sugar is a sugar where the -OH group at…
A: Sugars are also called carbohydrates. Carbohydrates are composed of carbon, hydrogen, and oxygen in…
Q: Cancer stem cells (CSCs) are cancer cells (found within tumors or hematological cancers) that…
A: Regular stem cells like hematopoietic stem cell that give birth to the entire blood cell lineages,…
Q: In the given structure below, 1. What protein was used as the template for modelling the structure…
A: the mRNA sequence is used as a template to assemble in order the chain of amino acids that form…
Q: Every amino acid that undergoes catabolic breakdown give rise to an ammonia molecule that must be…
A: There are 20 naturally occurring amino acids that are found in proteins. Amino acids can be…
Step by step
Solved in 2 steps

- Which of the following (could be more than one) would not be a rational explanation for why the three-dimensional structure of a protein is driven and stabilized largely by noncovalent rather than covalent bonds?a) Proteins may be degraded for energy, and if their three-dimensional structures were heldtogether by mostly covalent bonding, this might be too difficult to accomplishb) Proteins will need to be unfolded to cross biological membrane, and if their three-dimensionalstructures were held together by mostly covalent bonding, this might be too difficult toaccomplish.c) Protein function (transport, enzyme catalysis, etc...) may require flexibility in the three-dimensional structure to allow for conformational change, and if protein three-dimensionalstructure were held together by mostly covalent bonding, this might be too difficult toaccomplish.d) All of the answer choices are rational explanations for why the three-dimensional structure of protein is driven and stabilized largely…Define the following terms:a. salt bridgeb. oligomerc. allosteric transitiond. protein denaturatione. amphipathic moleculePlease Answer numbers 1, 2 & 3 thank you 1. Which of the following intermolecular molecular forces of attraction is disrupted when a native protein is added with acetic acid? a. Hydrogen bond b. Peptide bond c. Disulfide bond d. Salt bridge e. van der Waals force 2. Suppose a protein sample with a fragment containing the following amino acid sequence is subjected to various chemical assay/tests. - Ala-Gly-Phe-Met-Cys- which of the following test will the sample be positive? a. Lead-sulfide test b. Hopkins cole's test c. Millon's test d. Xanthoproteic test 3. Suppose a protein sample with a fragment containing the following amino acid sequence is subjected to various chemical assay/tests. - Ala-Gly-Trp-Phe-Met-Cys- What is observed when the protein sample is subjected to Millon’s test? a. Violet interface b. Red precipitate/solution c. Brown/black precipitate d. Yelllow product e. No observable result
- Indicate the level(s) of protein structure to which each of thefollowing contributes:a. amino acid sequenceb. b-pleated sheetc. hydrogen bondd. disulfide bondIn the tertiary structure of a protein, which pair of amino acid side chains would be most likely to participate in hydrophobic interactions? Asn/lys ser/cys gln/tyr val/thr phe/lêuWhich of the following statements are correct about the molten globular state of protein folding (select all that appy)? A. May be short-lived or long-lived intermediate in protein folding B. Contains substantial levels of secondary structure C. Has loose packing of hydrophobic core D. Larger radius of gyration than dentaured state E. Is a more compact state than the native confomation
- Which of the following, if any, is correct about protein folding? a)2° structure formation is primarily entropic, 3° structure formation is primarily entropic b)2° structure formation is primarily entropic, 3° structure formation is primarily enthalpic c)2° structure formation is primarily enthalpic, 3° structure formation is primarily entropic d)2° structure formation is primarily enthalpic, 3° structure formation is primarily enthalpic e)None of the aboveAlthough all of these may play a part, the major driving force in protein folding is: a) hydrogen bond formation b) salt bridge formation c) steric interactions d) the hydrophobic effect A partially folded protein is called a a) peptide template b) active domain c) molten globule d) precursor protein Which of these processed is NOT seen in protein denaturation? A) subunits are dissociated b) folding is unraveled c) alpha helix and beta sheets are unraveled d) peptide bonds are broken. A protein which has more than one stable conformation is called a: a) chaperone protein b) molten globule c) flexible protein d) inherently unstructured protein If a denatured protein does not spontaneously renature upon removal of denaturing agents, it may require a ____ to renature. a) chaperone protein b) molten globule c) flexible protein d) inherently unstructured protein Prion protein and amyloid protein have which trait in common? A) They denature very easily…a) In the article the authors reference the canonical forces in protein folding. Describe how these forces come into play when a protein folds. Why do the authors suggest that other intermolecular interactions must be important to fully understand folding processes? https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6995338/pdf/nihms-1067149.pdf
- Which of the following statements are correct about protein structure (select all that apply)? A. Post-translational modifications such as glycosylation or phosphorylation may alter the structure of a protein B. Only amino acids with a net charge may interact with other amino acids C. The 3D structure of a protein is determined primarily by the protein backbone/main chain conformation while the amino acid sidechains play only a minor role. D. Hydrophobic interactions play a key role in protein folding E. Amino acid sidechains contribute to 3D structure through their ability to form hydrogen bonds with other amino acidsWhich of the following statements are correct about the thermodynamics of protein folding (select all that appy)? A. The deltaG for protein folding is negative B. Burial of hydrophobic side provides a positive entropy change that drive protein folding C. The overall entropy change of protein folding is favorable D. Intercations between amino acids provide a large negative deltaH that helps favor the native state. E. The free energy change of protein folding is dependent on the temperaturePredict the protein 3° structure of the following protein sequence. Provide detail from 2° structure principles Nterm – SLDVTFSPGAEITFKWNPGSFNSLKDTIRQVTDK – Cterm

