Q: For the reaction below, Kc = 9.2 × 10⁻⁵. Note Kc is sometimes called K. What is the equilibrium…
A: Equilibrium constant Kc for a reaction is defined as the product of the molar concentrations of the…
Q: How are Mesons different from Bosons? A) Both are the same except the Bosons have a mass equal to…
A: Option (a) is incorrect. Bosons can have a range of masses, including zero (such as photons, the…
Q: Imagine an alternate universe where the value of the Planck constant is 6.62607 × 10-40 J-s. In that…
A: If the wavelength is very small than the dimensions of the object then it falls under classical…
Q: A chemical engineer is studying the following reaction: N₂(g) + 3H₂(g) → 2NH₂(9) At the temperature…
A: The reaction involved is N2 (g) + 3H2 (g) <-----> 2NH3 (g) Kp of the reaction = 0.0043…
Q: Balance the following reaction in your notes, assuming basic conditions (use the smallest whole…
A: Answer: Given chemical reaction is the redox reaction where one specie is getting oxidized and other…
Q: Which of the following species is NOT AROMATIC? (Note all species have a planar geometry) - Select…
A:
Q: For the reaction below, Kc = 4.60 × 10⁻⁶. Note Kc is sometimes called K. What is the equilibrium…
A:
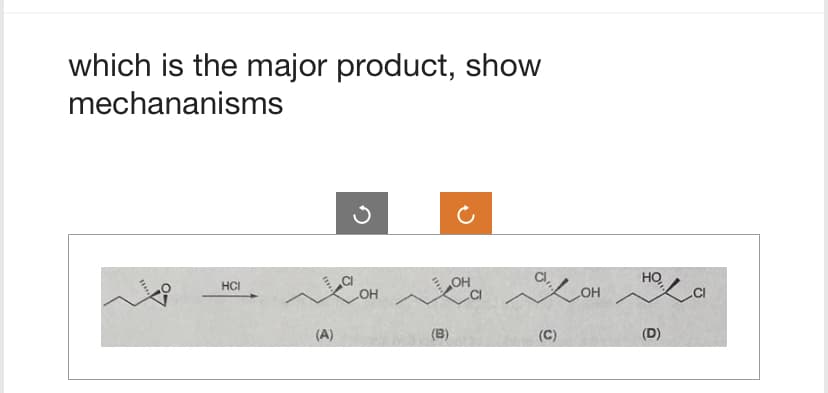
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 1. CH3CH2MgBr 2. Neutralizing…
A: The question is based on the reactions of aldehyde. this is an example of nucleophilic addition…
Q: For each of the salts on the left, match the salts on the right that can be compared directly, using…
A: Answer: For a sparingly soluble salts it is considered that all the dissolved quantity of the…
Q: A solution contains 7.00×10-3 M sodium sulfide and 6.87×10-3 M sodium phosphate. Solid silver…
A:
Q: A chemical engineer is studying the following reaction: HCH3CO₂(aq) + CH3NH₂(aq) → CH3CO₂(aq) +…
A: The direction in which the reaction proceeds to reach equilibrium can be predicted by determining…
Q: The decomposition of urea, (NH₂)₂CO, in 0.10 M HCI follows the equation (NH₂)₂CO (aq) + 2H+ (aq) +…
A:
Q: The first-order rate constant for the conversion of cyclobutane to ethylene at 1000.°C is 101 s²¹.…
A: Given that, the reaction is C4H8 2C2H4. The temperature is 1000°C. The reaction follows the first…
Q: Suppose a 250. mL flask is filled with 1.3 mol of H₂ and 1.2 mol of HI. The following reaction…
A:
Q: Why does benzene undergo substitution reactions, but not addition reactions? Select one: O A.…
A: We have to select the correct statement with respect to reactions of benzene
Q: 2. Fill in the missing information. MgBr 1. 2. H3O+ H3O+ mCPBA
A:
Q: The energy E of the electron in a hydrogen atom can be calculated from the Bohr formula: R E=- In…
A:
Q: For a given process, which proceeds at a constant pressure, the change in Gibbs can en- ergisomic…
A: For a given process, which proceeds at a constant pressure, the change in Gibbs free energy as a…
Q: Identify the precipitate(s) formed when solutions of Ca(ClO4)2(aq), K2CO3(aq), and NaNO3(aq) are…
A: The question is based on the concept of chemical reactions. we need to identify any precipitate…
Q: Shown on the next two pages are the mass, IR and NMR spectra for an unknown organic molecule. Based…
A: The question is based on the concept of organic spectroscopy . we have to analyse the spectral data…
Q: Anhydrous CuCl2 (0.12g) and triphenylphosphine oxide Ph3PO (0.5g) are reacted together to form what…
A: The reaction between anhydrous copper(II) chloride (CuCl2) and triphenylphosphine oxide (Ph3PO)…
Q: 14-36 Reaction of isoprene (2-methyl-1,3-butadiene) with ethyl propenoate gives a mixture of two…
A: Diels-Alder reaction is the type of pericyclic reaction. It is the cycloaddition reaction between…
Q: 1. a) Which non-metallic compounds have density values between 1.4 and 2.9 g/mL? b) What…
A: Non metal having density in between 1.4 to 2.9g/ml are benzophenone, bromo form, chloroform,…
Q: Lead thiocyanate, Pb(SCN)2, has a Kp value of 2.00 x 10- AB(s) A+ (aq) +B (aq) Le Châteller's…
A: Given: The value of Ksp of PbSCN2 is 2.00×10-5. The molarity of KSCN is 0.800 M.
Q: When the volume of a gas is changed from 3.75 L to 6.52 L, the temperature will change from 65.0 °C…
A:
Q: Consider a particle in state n = 4 moving in a 1D box of length / = 1.0 Å. Determine the probability…
A: The probability of finding a particle in a given region is proportional to the square of the…
Q: The following data were collected for the reaction of cyclobutane, C₂H₂(g), to form ethylene,…
A: Given: Part 1: To plot the graph of concentration versus time for the formation of C2H4 and the…
Q: 40 mL of a 0.200M HC4H7O2 solution was titrated with 100.0mL of 0.100M Sr(OH)2 Ka for HC4H7O2 is…
A: The reaction between acid and base is written below. 2C3H7COOH (aq) + Sr(OH)2 (aq) ⇌ 2H2O (l) +…
Q: Compound H₂C O₂N NH₂ NH₂ Conjugate acid H3C O₂N + NH3 + NH3 pk, value 5.08 1.00
A: According to Bronsted-Lowry acid base theory the compounds which have ability to donate proton or…
Q: A heat engine operates on a Carnot cycle. In this cycles, it absorbs 360J of energy while it expands…
A: Efficiency of a carnot engine given as (Q1–Q2)/Q1 = (Th—Tc)/Th Where Q1= heat of source Q2 =Heat of…
Q: What mass of LP gas is necessary to heat 1.7 L of water from room temperature (25.0 °C) to boiling…
A: Answer: In this we have to calculate the heat required to raise the temperature of water when…
Q: Solid cobalt(II) acetate is slowly added to 150 mL of a 0.0653 M potassium hydroxide solution. The…
A: The question is based on the concept of solubility product principle. It that states that when a…
Q: 24.98 Draw the condensed structural formula for and give the name of the amino acid formed when the…
A:
Q: P-V work (w = -PΔV) occurs in the following system. Assuming all gases are ideal such that PΔV =…
A: Given: Temperature = 296 K R = 8.314 J/mol·K w = -PΔV
Q: A 40.0 mL volume of 0.100 M HCl is titrated with 0.100 M NaOH. Part A Calculate the pH of the…
A: The balanced chemical reaction between NaOH and HCl is NaOH(aq) + HCl(aq) → NaCl(aq) + H2O(l)…
Q: Alcohols are soluble in water with up to how many carbons? 05 6 4 3 7
A: There are various types of intermolecular hydrogen bonding such as hydogen bonding, London…
Q: Give detailed Solution with explanation needed
A: Ketals and acetals on hydrolysis gives ketone as the main product and alcohols as byproducts.
Q: Write a balanced net ionic equation to show why the solubility of AgOH(s) increases in the presence…
A: The net ionic equation shows only the species that are involved in the reaction. Ksp for AgOH = 2.0…
Q: Calculate the ▲Gº for the following process at 100 K and 300 K. Use the thermodynamic data from…
A: Introduction: The problem can be solved using the following relation,…
Q: H H cat HCI H₂O
A: The question is based on the concept of organic reactions. we need to identify the product formed…
Q: 4. Give the structural formula of the compound Q (note the alkene isomerism). J Cat. C PCY3 Ru CI…
A:
Q: Consider the insoluble compound silver hydroxide, AgOH. The silver ion also forms a complex with…
A:
Q: To make 12 pancakes, combine 2 cups of mix, 2 eggs, and 1 cup of milk. This recipe could be…
A:
Q: Draw the most stable chair conformation of cis- 1,3-dimethylcyclohexane and trans- 1,3-…
A: Internal angles of a regular hexagon are 120°. The cyclohexane ring's carbon-carbon bonds, on the…
Q: 20. Which of the following is the correct rate law for this reaction? The following data was found…
A:
Q: A student performed the first determination in Part 2. They reacted 61.33 g of 1 M HCl with 0.6615 g…
A:
Q: What is the electron configuration for phosphorus anion
A: Phosphorus has an atomic number of 15, which means it has 15 electrons. To determine the electron…
Q: what volume of 5.00 M NaOH is used to make 4.00 L of 0.625 M NaOH?
A: Given M1 = 5 M M2 = 0.625 M V2 = 4 L
Q: ORGANIC FUNCTIONAL GROUPS Understanding the names of carboxylate salts What is the systematic name…
A: We know according to Baldwin rule 5 membered ring formed faster than any other higher membered ring…
Q: Calculate the pH of a 0.515 M aqueous solution of quinoline (C₂H,N, K = 6.3x10-10). pH =
A: We have to calculate the pH of solution
Step by step
Solved in 2 steps with 2 images

- The conversion of 3 alcohols into alkenes under acidic conditions involves two cationic intermediates. For each reaction, draw the complete mechanism using curved arrows.Under certain reaction conditions, 2,3-dibromobutane reacts with twoequivalents of base to give three products, each of which contains twonew π bonds. Product A has two sp hybridized carbon atoms, product Bhas one sp hybridized carbon atom, and product C has none. What arethe structures of A, B, and C?Which compounds (B–F) are identical to A? (b) Which compounds (B–F) represent an isomer of A?
- The alcohol compound güven the formula below is used in perfume making.bromine benzeme and -1 synthesis of this compound using the necessary organic and inor chemicals showA chiral alkyne A with molecular formula C6H10 is reduced with H2 and Lindlar catalyst to B having the R conguration at its stereogenic center. What are the structures of A and B?a) Draw two different enol tautomers of 2-methylcyclohexanone. (b) Draw two constitutional isomers that are not tautomers, but contain a C = C and an OH group.
- Label compounds B–D as stereoisomers, conformations, or constitutional isomers of A.Draw the products formed when both cis- and trans-but-2-ene are treated with OsO4, followed by hydrolysis with NaHSO3 + H2O. Explain how these reactions illustrate that syn dihydroxylation is stereospecic.Draw the products formed when both cis- and trans-but-2-ene are treated with OsO4, followed by hydrolysis with NaHSO3 + H2O. Explain how these reactions illustrate that syn dihydroxylation is stereospecific.
- An alkene G (C6H12) reacts with cold basic KMnO4 to produce H (C6H14O2). Hydrogenation of Gproduces I (C6H14). Ozonolysis of G produces propanone (CH3COCH3) and propanal(CH3CH2CHO).a) Draw the structure of G, H, I.b) State the reagent(s) and condition for ozonolysis reactionOne mole of an unknown hydrocarbon, compoundC,in the presence of a platinum catalyst, adds 98.9L of hydrogen, measured at 744mm Hg and 22°C, to form a saturated alkanewhich containsone ring. Whenone mole of compound Cis reacted with ozone,followed by reduction with (CH3)2S, four moles of only oneproductwasformed, whose condensed molecular formula is CHO-CHO. Give the structure ofcompound C. Explain your reasoning.When cyclohexanone was reacted with an amine A. It the reaction formed an enamine, whichof the following is the most likely identity of amine A?





