
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN: 9781305079250
Author: Mark S. Cracolice, Ed Peters
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10, Problem 42E
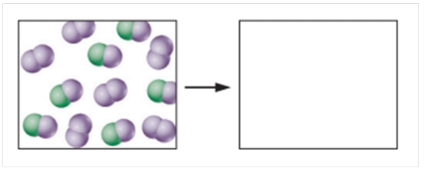
Carbon monoxide reacts with oxygen to form carbon dioxide at a temperature at which all species are in the gas phase. A tiny sample of the pre-reaction mixture is shown in the following diagram, in which purple spheres represent oxygen atoms and green spheres represent carbon atoms. Draw the product mixture. Give a written description of your reasoning.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Introductory Chemistry: An Active Learning Approach
Ch. 10 - The first step in the Ostwald process for...Ch. 10 - When hydrogen sulfide reacts with oxygen, water...Ch. 10 - Magnesium hydroxide is formed from the reaction of...Ch. 10 - In our bodies, sugar is broken down by reacting...Ch. 10 - Prob. 5ECh. 10 - Aqueous solutions of potassium hydrogen sulfate...Ch. 10 - The first step in the Ostwald process for...Ch. 10 - Butane, C4H10 is a common fuel used for heating...Ch. 10 - The explosion of nitroglycerine is described by...Ch. 10 - According to the reaction 2AgNO3+CuCu(NO3)2+2Ag,...
Ch. 10 - Prob. 11ECh. 10 - Prob. 12ECh. 10 - Prob. 13ECh. 10 - Prob. 14ECh. 10 - The hard water scum that forms a ring around the...Ch. 10 - Prob. 16ECh. 10 - Prob. 17ECh. 10 - Prob. 18ECh. 10 - The Solvay process is multistep industrial method...Ch. 10 - Prob. 20ECh. 10 - Prob. 21ECh. 10 - What mass of NaHCO3 must decompose to produce 448g...Ch. 10 - Prob. 23ECh. 10 - Solid ammonium chloride decomposes to form ammonia...Ch. 10 - What mass of magnesium hydroxide will precipitate...Ch. 10 - Prob. 26ECh. 10 - Prob. 27ECh. 10 - Prob. 28ECh. 10 - The reaction of a dry cell battery may be...Ch. 10 - Prob. 30ECh. 10 - Prob. 31ECh. 10 - Prob. 32ECh. 10 - Calcium cyanamide is a common fertilizer. When...Ch. 10 - Prob. 34ECh. 10 - The Haber process for making ammonia from nitrogen...Ch. 10 - Prob. 36ECh. 10 - Prob. 37ECh. 10 - The simplest example of the hydrogenation of a...Ch. 10 - Prob. 39ECh. 10 - Prob. 40ECh. 10 - Ammonia can be formed from a combination reaction...Ch. 10 - Carbon monoxide reacts with oxygen to form carbon...Ch. 10 - An experiment is conducted in which varying...Ch. 10 - The flasks below illustrated three trials of a...Ch. 10 - A solution containing 1.63g of barium chloride is...Ch. 10 - Prob. 46ECh. 10 - Prob. 47ECh. 10 - Prob. 48ECh. 10 - A mixture of tetraphosphorus trisulfide and...Ch. 10 - Sodium carbonate can neutralize nitric acid by the...Ch. 10 - Prob. 51ECh. 10 - Prob. 52ECh. 10 - Prob. 53ECh. 10 - Prob. 54ECh. 10 - Prob. 55ECh. 10 - Prob. 56ECh. 10 - Prob. 57ECh. 10 - Prob. 58ECh. 10 - Prob. 59ECh. 10 - Prob. 60ECh. 10 - Question 57 through 62: Thermochemical equations...Ch. 10 - Prob. 62ECh. 10 - Quicklime, the common name for calcium oxide, CaO,...Ch. 10 - What mass in grams of hydrogen has to react to...Ch. 10 - The quicklime produced in Question 63 is...Ch. 10 - Prob. 66ECh. 10 - What mass in grams of octane, a component of...Ch. 10 - Calculate the quantity of energy (kJ) transferred...Ch. 10 - Prob. 69ECh. 10 - Classify each of the following statements as true...Ch. 10 - Prob. 71ECh. 10 - What mass in grams of calcium phosphate will...Ch. 10 - Prob. 73ECh. 10 - Prob. 74ECh. 10 - A laboratory test of 12.8g of aluminum ore yields...Ch. 10 - How much energy is required to decompose 1.42g of...Ch. 10 - Prob. 77ECh. 10 - Prob. 78ECh. 10 - A sludge containing silver chloride is a water...Ch. 10 - Prob. 80ECh. 10 - Prob. 81ECh. 10 - Prob. 82ECh. 10 - Prob. 83ECh. 10 - Prob. 84ECh. 10 - In 1866, a young chemistry student conceived the...Ch. 10 - Prob. 86ECh. 10 - A student was given a 1.6240-g sample of a mixture...Ch. 10 - A researcher dissolved 1.382g of impure copper in...Ch. 10 - What mass in grams of magnesium nitrate, Mg(NO3)2,...Ch. 10 - Prob. 90ECh. 10 - Prob. 10.1TCCh. 10 - Solutions of zinc bromide and sodium hydroxide are...Ch. 10 - Prob. 2PECh. 10 - Prob. 3PECh. 10 - How mass of fluorine is formed when 3.0grams of...Ch. 10 - Prob. 5PECh. 10 - Prob. 6PECh. 10 - Prob. 7PECh. 10 - Prob. 8PECh. 10 - Prob. 9PECh. 10 - A solution containing 43.5g of calcium nitrate is...Ch. 10 - Prob. 11PECh. 10 - Prob. 12PECh. 10 - Prob. 13PECh. 10 - Prob. 14PECh. 10 - Prob. 15PECh. 10 - Prob. 1PCECh. 10 - Prob. 2PCECh. 10 - Prob. 3PCECh. 10 - Prob. 4PCECh. 10 - Prob. 5PCECh. 10 - Prob. 6PCECh. 10 - Eight problem-classification examples follow. Test...Ch. 10 - Prob. 8PCE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The “Chemistry in Focus” segment The Beetle That Shoots Straight discusses the bombardier beetle and the chemical reaction of the decomposition of hydrogen peroxide. m:math>H2O2(aq)H2O(l)+O2(g) e balanced equation given in the segment is m:math>2H2O2(aq)2H2O(l)+O2(g) y can’t we balance the equation in the following way? m:math>H2O2(aq)H2(g)+O2(g) e molecular-level pictures like those in Section 6.3 to support your answer.arrow_forwardIs there a difference between a homogeneous mixture of hydrogen and oxygen in a 2:1 mole ratio and a sample of water vapor? Explain.arrow_forwardMany over-the-counter antacid tablets are now formulated using calcium carbonate as the active ingredient, which enables such tablets to also be used as dietary calcium supplements. As an antacid for gastric hyperacidity, calcium carbonate reacts by combining with hydrochloric acid found in the stomach, producing a solution of calcium chloride, converting the stomach acid to water, and releasing carbon dioxide gas (which the person suffering from stomach problems may feel as a “burp”). Write the unbalanced chemical equation for this process.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY