
(a)
Interpretation:
Symbol of atom or ion has to be written that has 23 protons, 28 neutrons, and 20 electrons.
Concept Introduction:
Each and every element present in the Periodic table has a unique name. Some of the elements are named considering their
Chemical names are represented as atomic symbols. In the symbols, the mass number and atomic number are shown. The complete
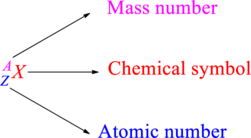
Atomic number is the total number of protons present in the atom of an element. Mass number is the total number of protons and neutrons present in nucleus of an atom.
(a)
Explanation of Solution
The species is said to contain 23 protons, 28 neutrons, and 20 electrons. Atomic number is equal to the number of protons and mass number is equal to the sum of protons and neutrons. Atomic number and mass number can be given as shown below.
The charge on the ion can be calculated considering the number of protons and electrons as shown below.
Element with the atomic number of 23 is vanadium. The atomic symbol of ion can be given as
(b)
Interpretation:
Symbol of atom or ion has to be written that has 53 protons, 74 neutrons, and 54 electrons.
Concept Introduction:
Refer part (a).
(b)
Explanation of Solution
The species is said to contain 53 protons, 74 neutrons, and 54 electrons. Atomic number is equal to the number of protons and mass number is equal to the sum of protons and neutrons. Atomic number and mass number can be given as shown below.
The charge on the ion can be calculated considering the number of protons and electrons as shown below.
Element with the atomic number of 53 is iodine. The atomic symbol of ion can be given as
(c)
Interpretation:
Symbol of atom or ion has to be written that has 44 protons, 58 neutrons, and 41 electrons.
Concept Introduction:
Refer part (a).
(c)
Explanation of Solution
The species is said to contain 44 protons, 58 neutrons, and 41 electrons. Atomic number is equal to the number of protons and mass number is equal to the sum of protons and neutrons. Atomic number and mass number can be given as shown below.
The charge on the ion can be calculated considering the number of protons and electrons as shown below.
Element with the atomic number of 44 is ruthenium. The atomic symbol of ion can be given as
(d)
Interpretation:
Symbol of atom or ion has to be written that has 15 protons, 16 neutrons, and 15 electrons.
Concept Introduction:
Refer part (a).
(d)
Explanation of Solution
The species is said to contain 15 protons, 16 neutrons, and 15 electrons. Atomic number is equal to the number of protons and mass number is equal to the sum of protons and neutrons. Atomic number and mass number can be given as shown below.
The charge on the ion can be calculated considering the number of protons and electrons as shown below.
As the charge is zero, this is an atom and not an ion.
Element with the atomic number of 15 is phosphorus. The atomic symbol of atom can be given as
Want to see more full solutions like this?
Chapter 2 Solutions
Chemistry: Principles and Practice
- The element bromine is Br2, so the mass of a Br2 molecule is the sum of the mass of its two atoms. Bromine has two isotopes. The mass spectrum of Br2 produces three peaks with relative masses of 157.836, 159.834, and 161.832, and relative heights of 6.337, 12.499. and 6.164, respectively. (a) What isotopes of bromine are present in each of the three peaks? (b) What is the mass of each bromine isotope? (c) What is the average atomic mass of bromine? (d) What is the abundance of each of the two bromine isotopes?arrow_forwardTwo basic laws of chemistry are the law of conservation of mass and the law of constant composition. Which of these laws (if any) do the following statements illustrate? (a) Lavoisier found that when mercury(ll) oxide, HgO, decomposes, the total mass of mercury (Hg) and oxygen formed equals the mass of mercury(ll) oxide decomposed. (b) Analysis of the calcium carbonate found in the marble mined in Carrara, Italy, and in the stalactites of the Carlsbad Caverns in New Mexico gives the same value for the percentage of calcium in calcium carbonate. (c) Hydrogen occurs as a mixture of two isotopes, one of which is twice as heavy as the other.arrow_forwardThe density of a mixture of gases may be calculated by summing the products of the density of each gas and the fractional volume of space occupied by that gas. (Note the similarity to the calculation of the molar mass of an element from the isotopic masses and fractional abundances.) Assume dry air with CO2 removed is 20.96% (by volume) oxygen. 78.11% nitrogen, and 0.930% argon. Determine the density of argon.arrow_forward
- he vigorous reaction between aluminum and iodine gives the balanced equation: :math>2Al(s)+3I2(s)2AlI2(s). mg src=Images/HTML_99425-9-2QAP_image001.jpg alt="" align="top"/> at do the coefficients in this balanced chemical equation tell us about the proportions in which these substances react on a macroscopic (mole) basis?arrow_forwardWhat are the live most abundant elements (by mass) in the earth’s crust, oceans, and atmosphere?arrow_forwardIn Groups 4A-6A, there are several elements whose symbols begin with S. Name these elements, and for each one give its symbol, atomic number, group number, and period. Describe each as a metal, metalloid, or nonmetal.arrow_forward
- The mass spectrum of bromine (Br2) consists of three peaks with the following characteristics: Mass (u) Relative Size 157.84 0.2534 159.84 0.5000 161.84 0.2466 How do you interpret these data?arrow_forward3. (a) Magnesium oxide was formed when the metal burns in air. (i) If 1.25 g of magnesium oxide contains 0.754 g of magnesium, determine the mass ratio of magnesium magnesium oxide. (ii) Calculate the weight of magnesium in 534 g of magnesium oxide.arrow_forwardAntimony has many uses, for example, in infrared devices and as part of an alloy in lead storage batteries. The element has two naturally occurring isotopes, one with mass 120.904 amu, the other with mass 122.904 amu. (a) Write the AX notation for each isotope. (b) Use the atomic mass of antimony Z from the periodic table to calculate the natural abundance of each isotope.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





