
Microbiology with Diseases by Body System (4th Edition)
4th Edition
ISBN: 9780321918550
Author: Robert W. Bauman Ph.D.
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 2VI
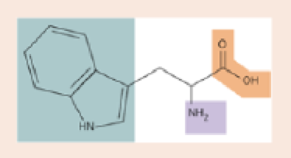
Shown is the amino acid tryptophan. Put the letter “C” at the site of every carbon atom. Label the amino group, the carboxyl group, and the side group.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Identify the amino acid shown below. (Note: single letter code is provided as answer)
H
H₂N-C-COOH
CH₂
CH₂
S
CH₁
M
Ow
F
Oc
Please clearly identify each numbered amino acid side chain, below. Your description should include the name or 3 letter code
for the amino acid and the type (i.e., chemical class) of the amino acid.
1
4
OH
OH
CH,
CH,
CH2
CH2
CH,
"H,N-C-C-0
"H,N-C-C-0
"H,N-C-C-0
_0-5-5-N H.
H O
H O
HO
но
Write down the abbreviations (both 1 letter and 3 letter) for the amino acids given below:Tryptophan, Glutamine, Isoleucine, Cysteine, Arginine
Chapter 2 Solutions
Microbiology with Diseases by Body System (4th Edition)
Ch. 2 - Electrons zip around the nucleus at about 5...Ch. 2 - Chlorine and potassium atoms form ionic bonds,...Ch. 2 - Why are decomposition reactions exothermic, that...Ch. 2 - Why does the neutralization of an acid by a base...Ch. 2 - Raw Oysters and Antacids: A Deadly Mix? The highly...Ch. 2 - Why do the cell membranes of microbes living in...Ch. 2 - Prob. 1MCCh. 2 - The atomic mass of an atom most closely...Ch. 2 - One isotope of iodine differs from another in...Ch. 2 - Prob. 4MC
Ch. 2 - Which of the following terms most correctly...Ch. 2 - In water, cations and anions of salts dissociate...Ch. 2 - Prob. 7MCCh. 2 - Which of the following statements about a...Ch. 2 - Proteins are polymers of ___________. a. amino...Ch. 2 - Which of the following are hydrophobic organic...Ch. 2 - Fill in the Blanks 1. The outermost electron shell...Ch. 2 - Fill in the Blanks 2. The type of chemical bond...Ch. 2 - Prob. 3FIBCh. 2 - Prob. 4FIBCh. 2 - Fill in the Blanks 5. Groups of atoms such as NH2...Ch. 2 - Fill in the Blanks 6. The reverse of dehydration...Ch. 2 - Fill in the Blanks 7. Reactions that release...Ch. 2 - Fill in the Blanks 8. All chemical reactions begin...Ch. 2 - Fill in the Blanks 9. The ____________ scale is a...Ch. 2 - Prob. 10FIBCh. 2 - Label a portion of the molecule below; label two...Ch. 2 - Shown is the amino acid tryptophan. Put the letter...Ch. 2 - List three main types of chemical bonds, and give...Ch. 2 - Name five properties of water that are vital to...Ch. 2 - Prob. 3SACh. 2 - What is the difference between atomic oxygen and...Ch. 2 - Explain how the polarity of water molecules makes...Ch. 2 - Prob. 1CTCh. 2 - Prob. 2CTCh. 2 - Two freshmen disagree about an aspect of...Ch. 2 - When an egg white is heated, it changes from...Ch. 2 - Prob. 5CTCh. 2 - The poison glands of many bees and wasps contain...Ch. 2 - Prob. 7CTCh. 2 - Prob. 8CTCh. 2 - The deadly poison hydrogen cyanide has the...Ch. 2 - Triple covalent bonds are stronger and more...Ch. 2 - How can hydrogen bonding between water molecules...Ch. 2 - How can a single molecule of magnesium hydroxide...Ch. 2 - Prob. 13CTCh. 2 - Prob. 14CTCh. 2 - A textbook states that only five nucleotide bases...Ch. 2 - Prob. 1CM
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- After a polypeptide chain has been synthesized, certain amino acids in the peptide may become modified. For each modified amino acid, identify the standard amino acid from which it is derived. Enter the unabbreviated name of the standard amino acid. OH I *NH,CH,–C–CH,CH,—CH–COO H C *NH₂ standard amino acid: Identify the modification that yields the first amino acid depicted. addition of a hydroxyl group addition of an amino group modification of the existing amino group modification of the existing hydroxyl group elongation of the side chain by the addition of a -CH₂- grouparrow_forwardGive the Essential amino acids with the carboxyl group, amino group, and the Radical/Side Chain in the proper positions. Show in RED ink the amide bonds in the polypeptide formed in the essential amino acids. Give the name of the polypeptide. Copy and complete the table below.arrow_forwardFor the protein given in the attached picture: Write the name of these 5 amino acids corresponding to their abbreviation of 3 letters. Describe precisely how the functional groups in the amino acids are involved in bonding between two successive amino acids in the protein.arrow_forward
- Identify the amino acid shown below. (Note: single letter code is provided as answer) H H₂N-C-COOH CH₂ CH₂ CH₂ Эс Ow COM OFarrow_forwardWrite the structure formula, three-letter and one-letter abbreviation for each essential amino acid at pH 7. Histidine Arginine Lysinearrow_forwardidentify an amino acid that contains a sulfhydryl side chain (complete name)arrow_forward
- Draw the stereoisomers of the following amino acids. Indicate pairs of enantiomers and pairs of diastereomers. leucineCH3CHCH2 CHCOO−CH3 +NH3 isoleucineCH3CH2CH CHCOO−CH3arrow_forwardWrite the following oligopeptide using the one letter code for the amino acids: Cys-His-lle-Leu-Glu N.B. Use upper case characters only, pleasearrow_forwardDraw one aromatic polar amino acid at pH 7. Label with full name, three letter abbreviation, and single letter symbol.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Macromolecules | Classes and Functions; Author: 2 Minute Classroom;https://www.youtube.com/watch?v=V5hhrDFo8Vk;License: Standard youtube license