
Concept explainers
(a)
Interpretation:
Complete the given
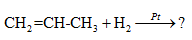
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms.
The chemical reaction which involves the addition of two hydrogen atoms across the double bond in the presence of catalyst is known as hydrogenation.
(b)
Interpretation:
Complete the given chemical reaction of alkene.
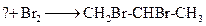
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that is double and triple bond.
The chemical reaction which involves the addition of two halogens across the double bond is known as halogenation.
(c)
Interpretation:
Complete the given chemical reaction of alkene.
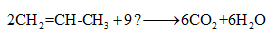
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that is double and triple bond.
The reaction in which burning of hydrocarbons takes place in oxygen to release water and carbon dioxide is known combustion reaction of hydrocarbon.
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
Introductory Chemistry: A Foundation
- Alcohols are very useful starting materials for the production of many different compounds. The following conversions, starting with 1-butanol, can be carried out in two or more steps. Show the steps (reactants/catalysts) you would follow to carry out the conversions, drawing the formula for the organic product in each step. For each step, a major product must be produced. (See Exercise 62.) (Hint: In the presence of H+, an alcohol is converted into an alkene and water. This is the exact reverse of the reaction of adding water to an alkene to form an alcohol.) a. 1-butanol butane b. 1-butanol 2-butanonearrow_forwardWhich of the following is the formula for an alkene? a. C3H7 b. C7H14 c. C5H12 d. C5H8 e. C6H6arrow_forwardName the alkene. CH2 H2 .C. CH3 H3C H2 name:arrow_forward
- Name the type of organic compound formed by each ofthe following reactions.a. elimination from an alcoholb. addition of hydrogen chloride to an alkenec. addition of water to an alkened. substitution of a hydroxyl group for a halogen atomarrow_forward4. Which of the following has isomeric forms?a. C2H3Clb. C2H5Clc. C2HCld. C2H4Cl2 5. Which of the following hydrocarbons always gives the same product when one of its hydrogen atoms is replaced by a chlorine atom.a. Hexaneb. Hex-1-enec. Cyclohexaned. Cyclohexenearrow_forwardName the alkene. CH2 H2 .C. CH3arrow_forward
- OH 1. POCl3 pyridine 2. Cl₂ H₂O ? Major Organic Productarrow_forwardThis type of hydrocarbons contain at least one double or triple bond. Unsaturated hydrocarbons Saturated hydrocarbons Polyunsaturated hydrocarbons O Supersaturated hydrocarbons The most acidic among the hydrocarbons. Alkynes Alkenes Alkanes Arenes The number of secondary carbon(s) in this compound. CH3 CH,-C CHCH, CH3 CIarrow_forwardWhat are the products of the complete combustion of 1-propanol, C,H,OH? A B C D carbon and oxygen carbon dioxide and water carbon monoxide and water carbon and hydrogenarrow_forward
- Write a balanced chemical equation for the reaction of ethene with oxygen. O a. C2H4(g) + 302(g) → 2CO2(g) + 2H2O(g) O b. 2C2H2(g) + 502(g) → 4CO2(g) + 2H2O(g) O c. CH4(g) + 2O2(g) → CO2(g) + 2H2O(g) O d. C2H4(g) + 202(g) → 2CO2(g) + 2H2(g) O e. 2C2H6(g) + 702(g) →> 4CO2(g) + 6H2O(g)arrow_forwardThe addition of hydrogen (H2). using a catalyst such as Ni or Zn and heat and/or pressure will change an aldehyde into a primary alcohol an alkene into an alkane all of these an. alkyne into an alkane a ketone into a secondary alcoholarrow_forwardFor the following reaction, draw the major organic product and select the correct IUPAC name for the organic reactant. If there is more than one major product, both may be drawn in the same box. When drawing hydrogen atoms on a carbon atom, either include all hydrogen atoms or none on that carbon atom, or your structure may be marked incorrect. H. HBr H. Select the correct IUPAC name for the organic reactant: 3-methyl-1-butene 2-methylbutene 2-methyl-3-butene O 4-methyl-1-pentenearrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning  Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





