
Concept explainers
a)
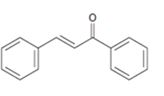
Interpretation:
The structure of the
Concept introduction:
Aldehydes and ketones with α-hydrogen undergo a base catalyzed carbonyl condensation reaction in aldol condensation. In this reaction two molecules of the reactant combine by forming a bond between α-carbon of one molecule and the carbonyl carbon within the same molecule or of the second molecule to give an aldol. The aldol dehydrates on heating to yield α, β-unsaturated aldehyde or ketone.
To show:
The structure of the aldehydes or ketones that should be used as a starting material to synthesize the compound given in an aldol reaction.
b)
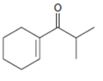
Interpretation:
The structure of the aldehydes or ketones that should be used as a starting material to synthesize the compound given in an aldol reaction is to be shown.
Concept introduction:
Aldehydes and ketones with α-hydrogen undergo a base catalyzed carbonyl condensation reaction in aldol condensation. In this reaction two molecules of the reactant combine by forming a bond between α-carbon of one molecule and the carbonyl carbon within the same molecule or of the second molecule to give an aldol. The aldol dehydrates on heating to yield α, β-unsaturated aldehyde or ketone.
To show:
The structure of the aldehydes or ketones that should be used as a starting material to synthesize the compound given in an aldol reaction.
c)
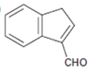
Interpretation:
The structure of the aldehydes or ketones that should be used as a starting material to synthesize the compound given in an aldol reaction is to be shown.
Concept introduction:
Aldehydes and ketones with α-hydrogen undergo a base catalyzed carbonyl condensation reaction in aldol condensation. In this reaction two molecules of the reactant combine by forming a bond between α-carbon of one molecule and the carbonyl carbon within the same molecule or of the second molecule to give an aldol. The aldol dehydrates on heating to yield α, β-unsaturated aldehyde or ketone.
To show:
The structure of the aldehyde that should be used as a starting material to synthesize the compound given in an aldol reaction.
d)
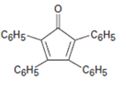
Interpretation:
The structure of the aldehydes or ketones that should be used as a starting material to synthesize the compound given in an aldol reaction is to be shown.
Concept introduction:
Mixed aldol condensation leads to a single product if i)one of the carbonyl partners contains no α hydrogen atoms but contains an unhindered carbonyl group and ii) if one of the carbonyl partners are much more acidic than the other and thus forms an enolate anion in preference to other.
To show:
The structure of the aldehydes or ketones that should be used as a starting material to synthesize the compound given in an aldol reaction.
Trending nowThis is a popular solution!

Chapter 23 Solutions
Organic Chemistry
- In this chapter, we will study three types of Condensation reactions: Michael, Aldol and Claisen. Match the right reaction with the statements below. ● This condensation reaction takes place between two esters. The electrophile in this reaction must be an a,B-unsaturated carbonyl compound. The B-hydroxy carbonyl products of this reaction are often dehydrated to form a,B- unsaturated carbonyl products.arrow_forwardBenzalacetone is a side product formed in the aldol condensation of dibenzalacetone. Show the reaction mechanism of how this side product, Benzalacetone, is formed.arrow_forwardPropose an appropriate intermediate for the following reactionarrow_forward
- Draw the structure of the major aldol product (prior to possible dehydration) of the following reaction without specifying stereochemistry. CH3 ལན། ཚིགས་. dilute aqueous NaOH 0-5° You do not have to consider stereochemistry. If no reaction occurs, draw the organic starting material. 0 + ChemDoodle [] заarrow_forwardA student tried to prepare the following compounds using aldol condensations. Which of these compounds was she successful in synthesizing? Explain why the other syntheses were not successful.arrow_forwardWhat are the products of an aldol cyclization for the following compound?arrow_forward
- A different way of synthesising the product 4,4- diphenylbut-3-en-2-one is via an aldol condensation. What starting materials would you use and what reaction conditions would you employ? Draw the full reactionarrow_forwardWhat is the product formed when each dicarbonyl compound undergoes an intramolecular aldol reaction, followed by dehydration.arrow_forwardSynthesis: Show how you would carry out the following synthesis. Include the reagents you would need for each step and the intermediate products formed in each step. You may use any inorganic reagents you need and organic reagents with 6 or fewer carbons.arrow_forward
- Synthesis: Show how you would carry out the following synthesis. Include the reagents you would need for each step and the structure of the intermediate products formed in each step. You may use any inorganic reagents you need and organic reagents of four or fewer carbons.arrow_forwardDraw the product of the following sequence of reactions. Show stereochemistry.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
