
UNIVERSITY PHYSICS,VOL.2 (OER)
16th Edition
ISBN: 2810022325764
Author: OpenStax
Publisher: XANEDU C
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 4, Problem 48P
For the Carnot cycle of Figure 4.12, what is the entropy change of the hot reservoir, the cold reservoir, and the universe?
Figure 4.11 The four processes of the Carnot cycle. The working substance is assumed to be an ideal gas whose
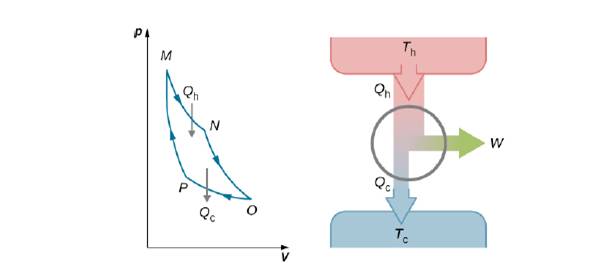
Figure 4.12 The total work done by the gas in the Carnot cycle is shown and given by the area enclosed by the loop MNOPM.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
a) Suppose 1.0 mole of nitrogen is confined to the left side of the container in the figure below. The valve is opened and the volume of the gas doubles. What is the entropy change of the gas in this irreversible process? Treat gas as ideal.
b) After having calculated what was asked in letter a, answer what is the variation of entropy if the gas had expanded, doubling the volume, through a free and adiabatic expansion? Justify your answer, explain the differences and similarities between the cases and what is linked to the entropy variation in this case.
QUESTION 4
a) A process involving an ideal gas is carried out in which the temperature changes (AT) at
constant volume (V). Consider the following two processes
Process A: for a fixed value AT, the mass of the gas (m) is doubled.
Process B: the process is repeated with the same initial mass but AT is doubled.
State which process has a greater change in entropy (AS). Explain your answer in terms
of AS for each process.
Write your answer here
b) One mole of N2 at 20.5 °C and 6.00 bar undergoes a transformation to the state described
by 145 °C and 2.75 bar. Calculate change in entropy (AS) (in unit J K1). (Assume ideal
gas behaviour).
Given that the molar heat capacity at constant pressure
T
(CP.m)[J mol¬1 K = 30.81 – 11.87 × 10-3– + .
K?
+ 2.3968 × 10-5
K
1.0176 × 10¬8
K3
The attached images have 3 parts, solve those 3 parts and also because of limited images to upload, this is the 4th part, iam typing here:
Calculate the total change in entropy for entire system, in J/K.
Chapter 4 Solutions
UNIVERSITY PHYSICS,VOL.2 (OER)
Ch. 4 - Check Your Understanding What is the efficiency of...Ch. 4 - Check your Understanding Show that QhQh=QcQc for...Ch. 4 - Check Your Understanding A Carnot engine operates...Ch. 4 - Check Your Understanding A Carnot refrigerator...Ch. 4 - Check Your Understanding In Example 4.7, the...Ch. 4 - Check Your Understanding A quantity of heat Q is...Ch. 4 - Check Your Understanding A 50-g copper piece at a...Ch. 4 - State an example of a process that occurs in...Ch. 4 - Explain in practical terms why efficiency is...Ch. 4 - If the refrigerator door is left what happens to...
Ch. 4 - Is it possible for the efficiency of a reversible...Ch. 4 - In the text, we showed that if the Clausius...Ch. 4 - Why don't we operate ocean liners by extracting...Ch. 4 - Discuss the practical advantages and disadvantages...Ch. 4 - The energy output of a heat pump is greater than...Ch. 4 - Speculate as to why nuclear power plants are less...Ch. 4 - An ideal gas goes from state (pi,vi,) to state...Ch. 4 - To increase the efficiency of a Carnot engine,...Ch. 4 - How could you design a Carnot engine with 100%...Ch. 4 - What type of processes occur in a Carnot cycle?Ch. 4 - Does the entropy increase for a Carnot engine for...Ch. 4 - Is it possible for a system to have an entropy...Ch. 4 - Are the entropy changes of the system in the...Ch. 4 - Discuss the entropy changes in the systems of...Ch. 4 - A tank contains 111.0 g chlorine gas l2), which is...Ch. 4 - A mole of ideal monatomic gas at 0 and 1.00 atm...Ch. 4 - A mole of an ideal gas at pressure 4.00 atm and...Ch. 4 - After a free expansion to quadruple its volume, a...Ch. 4 - An engine is found to have an efficiency of 0.40....Ch. 4 - In performing 100.0 J of work, an engine...Ch. 4 - An engine with an efficiency of 0.30 absorbs 500 J...Ch. 4 - It is found that an engine discharges 100.0 J...Ch. 4 - The temperature of the cold reservoir of the...Ch. 4 - An engine absorbs three times as much heat as it...Ch. 4 - A coal power plant consumes 100,000 kg of coal per...Ch. 4 - A refrigerator has a coefficient of performance of...Ch. 4 - During one cycle, a refrigerator removes 500 J...Ch. 4 - If a refrigerator discards 80 J of heat per cycle...Ch. 4 - A refrigerator has a coefficient of performance of...Ch. 4 - The temperature of the cold and hot reservoirs...Ch. 4 - Suppose a Carnot refrigerator operates between Tc...Ch. 4 - A Carnot engine operates between reservoirs at 600...Ch. 4 - A 500-W motor operates a Carnot refrigerator...Ch. 4 - Sketch a Carnot cycle on a temperature-volume...Ch. 4 - A Carnot heat pump operates between 0 and 20 ....Ch. 4 - An engine between heat reservoirs at 20 and 200 ...Ch. 4 - Suppose a Carnot engine can be operated between...Ch. 4 - A Carnot engine is used to measure the temperature...Ch. 4 - What is the minimum work required of a...Ch. 4 - Two hundred joules of heat are removed from a heat...Ch. 4 - In an isothermal reversible expansion at 27 , an...Ch. 4 - An ideal gas at 300 K is compressed isothermally...Ch. 4 - What is the entropy change of 10 g of steam at 100...Ch. 4 - A metal is used to conduct heat between two...Ch. 4 - For the Carnot cycle of Figure 4.12, what is the...Ch. 4 - A 5.0-kg piece of lead at a temperature of 600 is...Ch. 4 - One mole of an ideal gas doubles its volume in a...Ch. 4 - One mole of an ideal monatomic gas is confined to...Ch. 4 - (a) A 5.0-kg rock at a temperature of 20 is...Ch. 4 - A copper rod of cross-sectional area 5.0 cm2 and...Ch. 4 - Fifty grams of water at 20 is heated until it...Ch. 4 - Fifty grams of water at 0 are changed into vapor...Ch. 4 - In an isochoric process, heat is added to 10 mol...Ch. 4 - Two hundred grams of water at 0 is brought into...Ch. 4 - Suppose that the temperature of the water in the...Ch. 4 - Two hundred grams of water at 0 is brought into...Ch. 4 - (a) Ten grams of H2O stats as ice at 0 . The ice...Ch. 4 - The Carnot cycle is represented by the...Ch. 4 - A Carnot engine operating between heat reservoirs...Ch. 4 - A monoatomic ideal gas (n moles) goes through a...Ch. 4 - A Carnot engine has an efficiency of 0.60. When...Ch. 4 - A Carnot engine performs 100 J of work while...Ch. 4 - A Carnot refrigerator exhausts heat to the air,...Ch. 4 - A 300-W heat pump operates between the ground,...Ch. 4 - An engineer must design a refrigerator that does...Ch. 4 - A Carnot engine employs 1.5 mol of nitrogen gas as...Ch. 4 - A 5.0-kg wood block starts with an initial speed...Ch. 4 - A system consisting of 20.0 mol of a monoatomic...Ch. 4 - A glass beaker of mass 400 g contains 500 g of...Ch. 4 - A Carnot engine operates between 550 and 20 ...Ch. 4 - An ideal gas at temperature T is stored in the...Ch. 4 - A 0.50-kg piece of aluminum at 250 is dropped...Ch. 4 - Suppose 20 g of ice at 0 is added to 300 g of...Ch. 4 - A heat engine operates between two temperatures...Ch. 4 - A thermal engine produces 4 MJ of electrical...Ch. 4 - A coal power plant consumes 100,000 kg of coal per...Ch. 4 - A Carnot engine operates in a Carnot cycle between...Ch. 4 - A Carnot engine working between two heat baths of...Ch. 4 - A Carnot cycle working between 100 and 30 is...Ch. 4 - (a) infinitesimal amount of heat is added...Ch. 4 - Using the result of the preceding problem, show...Ch. 4 - With the help of the two preceding problems, show...Ch. 4 - A cylinder contains 500 g of helium at 120 atm and...Ch. 4 - A diatomic ideal gas is brought from an initial...Ch. 4 - The gasoline internal combustion engine operates...Ch. 4 - An ideal diesel cycle is shown below. This cycle...Ch. 4 - Consider an ideal gas Joule cycle, also called the...Ch. 4 - Derive a formula for the coefficient of...Ch. 4 - Two moles of nitrogen gas, with =7/5 for ideal...Ch. 4 - A Carnot refrigerator, working between 0 and 30 ...
Additional Science Textbook Solutions
Find more solutions based on key concepts
A refrigerator extracts energy from its contents at the rate of 95 W. How long will it take to freeze 750 g of ...
Essential University Physics (3rd Edition)
Choose the best answer to each of the following. Explain your reasoning. 9.If you had been present in the unive...
The Cosmic Perspective Fundamentals (2nd Edition)
3. Draw a circuit diagram for the circuit of Figure P23.3.
Figure P23.3
College Physics: A Strategic Approach (4th Edition)
Calculate the displacement and velocity at times of (a) 0.500, (b) 1.00, (c) 1.50, and (d) 2.00 s for a ball th...
College Physics
Whether the angle of the boat's path across the river is greater than, less than or equal to 12° .
Physics (5th Edition)
The speed of the person sitting on the chair relative to the chair and relative to Earth.
Conceptual Physics (12th Edition)
Knowledge Booster
Similar questions
- Two hundred grams of water at 0 is brought into contact into thermal equilibrium successively with reservoirs at 20 , 40 , 60 , and 80 . (a) What is the entropy change of the water? (b) Of the reservoir? (c) What is the entropy change of the universe?arrow_forwardA copper rod of cross-sectional area 5.0 cm2 and length 5.0 m conducts heat from a heat reservoir at 373 K to one at 273 K. What is the time rate of change of the universe's entropy for this process?arrow_forwardOne mole of an ideal monoatomic gas is subjected to the processes in the figure below: State 1 has pressure P=2.8 atm, volume V=4.93 m^3 and temperature T=260 K . Volume V' is 4 times larger than V.What are the temperatures of states 2,3 and the pressure of state 2?T2=KT3=KP2= atmWhat is the entropy change, in J/K, of each thermodynamic process?ΔS1-2W= J/KΔS2-3W= J/KΔS3-1W=J/Karrow_forward
- What is the entropy change for an energy reservoir at 700 degrees Celsius that transfers 1500kJ of heat to a heat engine. Please include your calculations in answer.arrow_forwardPlease help me with these questions a. What is the name given to this cycle, and why is it important? The volume of the ideal gas at point 1 is V1, etc. By analysing the adiabatic processes, we find that: V_2/V_1 = V_4/V_3 b. The work done during the isothermal processes is WC (between points 1 and 2) and WH (between points 3 and 4). Find an expression for the fraction WC / WH in terms of TC and TH. c. Apply the First Law of Thermodynamics to the isothermal processes to show that the efficiency of this cycle is (please see the image with the corresponding formula for question c) d. Use the Second Law of Thermodynamics to explain why no heat engine can be more efficient than a perfectly reversible engine. You may wish to include a heat flow diagram in your answer.arrow_forwardIn a heat engine, 2.00 mol of a diatomic gas are carried through the cycle ABCDA, shown in Figure. (The PV diagram is not drawn to scale.) The segment AB represents an isothermal expansion, and the segment BC is an adiabatic expansion. The pressure and temperature at A are 5.00 atm and 600 K. The volume at B is twice the volume at A. The pressure at D is 1.00 atm. A) What is the pressure at B? B) What is the temperature at C? C) Find the total work done by the gas in one cycle.arrow_forward
- In a heat engine, 2.00 mol of a diatomic gas are carried through the cycle ABCDA, shown in Figure. (The PV diagram is not drawn to scale.) The segment AB represents an isothermal expansion, and the segment BC is an adiabatic expansion. The pressure and temperature at A are 5.00 atm and 600 K. The volume at B is twice the volume at A. The pressure at D is 1.00 atm.A) What is the pressure at B?B) What is the temperature at C?C) Find the total work done by the gas in one cycle.arrow_forwardProblem 1: Describe a situation in which the entropy of a container of gas is constant. In other words, come up with your own problem where the answer is that AS = 0.arrow_forwardMethane is heated in a steady-flow process at approximately atmospheric pressure. 1000 kJ of heat is added to 10 moles of methane initially at 500.0K. Note: There can be more than 1 exiting temperature that can give a heat of 1000 kJ. For this one, the exiting temperature does not exceed 3500 K. What is the final temperature? What is the average specific heat capacity, cp, for the temperature range of this heating process? What is the entropy change of the gas?arrow_forward
- Imagine that you are rolling three typical six-sided dice. Each way that you can roll a particular outcome using these three dice represents a microstate for that outcome. How many ways can you roll a five with these three dice? That is, how many microstates exist for a roll of five with three dice? What is the entropy associated with an outcome of five in this situation?arrow_forwardIn an experiment, the students used an unknown substance with a molecular weight of 28. They utilized 1kg of the unknown substance with a specific heat of 1.04kJ/kgK at constant pressure. The students also assumed that the substance operates in the same way as an Ideal Gas does. Show unit analysis and conversion. Find: a. the interval in the entropy of the substance from 47degree Celsius and 2atm to 327degree Celsius and 5atm. b. the substance's interval in internal energy.arrow_forwardThe molar heat capacities of gases are not perfectly constant, as we've assumed, but increase slowly with temperature. An empirical formula for the molar heat capacity at constant volume of nitrogen is Cv = (20.6 -1.6 × 10-³T+8.0 × 10-6T²) J/mol K, where T is in K Part A What is the entropy increase of the gas if 4.2 g of nitrogen in a rigid container are slowly heated from 300°C to 500°C? Express your answer with the appropriate units. AS = 0.15 Submit μА J K Previous Answers Request Answer < Return to Assignment X Incorrect; Try Again; One attempt remaining ? Provide Feedbackarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
