05) Determine Assume the soil coefficient (log Koc). factor teem in your molecular structure is given as по Collection The followe : Calculations. Bekow CI Given, log Koc = ос NH2 0.53' x + 0.62 + Σ n; Pj, Where X is the first order molecular connectivity n; is the number of groups of type 'j molecule and Pj Is Collection factor in Index the for each group. (6 marks) [Last question Numerical
05) Determine Assume the soil coefficient (log Koc). factor teem in your molecular structure is given as по Collection The followe : Calculations. Bekow CI Given, log Koc = ос NH2 0.53' x + 0.62 + Σ n; Pj, Where X is the first order molecular connectivity n; is the number of groups of type 'j molecule and Pj Is Collection factor in Index the for each group. (6 marks) [Last question Numerical
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter21: Surface Characterization By Spectroscopy And Microscopy
Section: Chapter Questions
Problem 21.10QAP
Related questions
Question
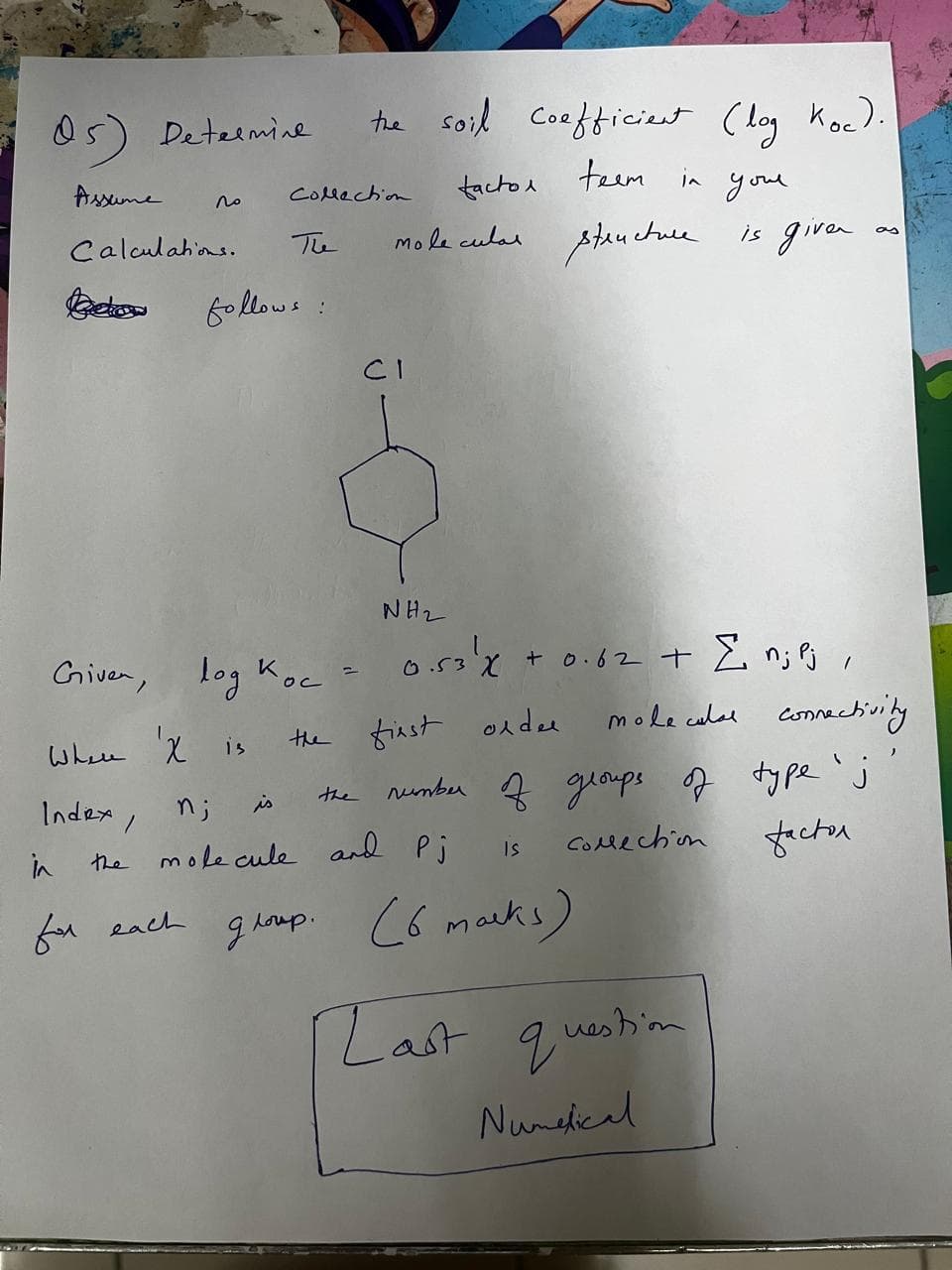
Transcribed Image Text:05) Determine
Assume
the soil coefficient (log Koc).
factor teem in your
molecular structure is given
as
по
Collection
The
followe
:
Calculations.
Bekow
CI
Given, log Koc
=
ос
NH2
0.53' x + 0.62 + Σ n; Pj,
Where X is
the first order
molecular connectivity
n;
is
the
number of groups of type 'j
molecule and Pj
Is
Collection
factor
in
Index
the
for each
group. (6 marks)
[Last question
Numerical
AI-Generated Solution
Unlock instant AI solutions
Tap the button
to generate a solution
Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning
