Q: 1 calorie is equal to 4.184 joules. true or false
A: The question is asking to verify the equivalence between the units of energy, calorie and joule.
Q: A Br 1. 0.9 eq LDA, rt Br 2. 3. NaOH, Br₂ 4. H+, H₂O Во OH HO Br Br D OH E O Br Br Br Major organic…
A: When LDA (lithium diisopropylamide) added to the ketone (3-methyl butan-2-one), it will deprotonate…
Q: of OH Starting materials 1 Br H,C OH 2 Br 6 Reagents a Mg / dry ether b 1. CO2 2. acidic workup c…
A: Find out suitable reagent for this reaction.
Q: true or false? the percentage of carbon in C2H6 is 21.9%
A: The objective of the question is to verify if the percentage of carbon in C2H6 (Ethane) is 21.9%.
Q: How many signals will be expected in the 1H NMR spectrum of the following compound? Don't give…
A: We have a structure for which we need to find out the number of unique hydrogen groups, i.e. the…
Q: Identify the product N in the following reaction. HO COOH COOH її СН, ОН/КОН Н N Give detailed…
A: We know K2Cr2O7 is an oxidizing agent, it will oxidize the phenanthrene to phenanthrene-9,10-dione.…
Q: 4. A 1.500-g sample of limestone rock is pulverized and treated with 30.00 mL of a 1.04 M HCI…
A: Mass percentage is defined as the quantity of solute present in 100 grams of solution.
Q: What is the coefficient of O2? - F652 + 02-> Fe2O3 + 502 05 О 10 0000
A: • Check whether the reaction is already balanced.• If not, balance each atom separately by…
Q: Draw the structure(s) of the major organic product(s) of the following reaction. 1. Dry Et₂O & +…
A: The products for both the reactions has drawn with proper mechanism. Please find the attachment…
Q: Provide the correct IUPAC name for the compound shown here. CI
A: Since,Rule of IUPAC-1) Longest chain as parent chain.2) Numbering start from those side where more…
Q: 22. Draw the major product of the following SN2 reaction. Draw a mechanism, showing the transition…
A: The bimolecular nucleophilic substitution reaction is a concerted reaction that occurs in a single…
Q: 1. The following data was obtained from decomposition of nitrogen oxide (NO). 2HX Hz + X2 T(K) 703…
A: The objective of the question is to calculate-The activation energyThe Arrhenius constantThe…
Q: 17. Draw a mechanism that explains the formation of the following product in this Friedel-Crafts…
A:
Q: 6. Predict the major product. H₂ H3C CC-CH3 a. Pd H₂ H3C-CC-CH3 b. Lindlar's catalyst NaNH, c. H3C…
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: please help!!!! I need a detailed reaction mechansim for this molecule and the name of the molecule!…
A: Esterification is a chemical reaction in which two reactants, typically an alcohol and an acid,…
Q: Provide the best method to synthesize the following alkene via Wittig.
A: The conversion of carbonyl compounds into alkene by the reaction with the triphenyl phosphonium…
Q: This molecule undergoes rearrangement via an oxo-di-p-methane pathway, a common metabolic route for…
A: This reaction is oxo-di-pi methane rearrangement.The most remarkable photochemical reaction of non…
Q: 4.4 (Pages 149-155); Watch KCV 4.4, IWE 4.6. Elemental phosphorus reacts with chlorine gas according…
A: Given reaction,Mass of Molar mass of Mass of Molar mass of Excess reactant left= ?
Q: what is the solubility of magnesium hydroxide in 0.0050 M MgCl2? Ksp= 6.0x10-12.
A: The question is based on the concept of solubility product principle.It states that when a weak…
Q: (CH3)2CHOH condensed structure
A: The objective of the question is to give the condensed structure of .
Q: Draw the product of this reaction. Ignore inorganic byproducts. Br Br a 1. excess NaNH2, A 2. H3O+ I…
A:
Q: What is the major elimination product to the reaction below? CI NaOEt HOEt, heat A. B. C. D.…
A: Given that, the elimination reaction reaction is:
Q: Consider the combustion of liquid C₅H₈ in oxygen gas to produce carbon dioxide gas and water vapor.…
A: The objective of this question is to calculate the percent error of the experimentally determined…
Q: Draw the structure of a compound, C4H8O3 that exhibits IR absorptions at 1710 and 2500-3000 cm-1 and…
A: The objective of the question is to draw the structure of the compound for the given data.
Q: Students were asked to find the identity of a wooden object. The mass of the object is 33.0 grams. A…
A: given that = dimension of object is 3.0cm*5.2cm*2.8cmmass of the object = 33.0 gramsdensity is a…
Q: Choose the reagent(s) that would be most likely to complete this reaction. HO 1. Hg(OAc)2, H₂O 2.…
A: Given,The reaction is:
Q: The equilibrium constant, Kc, for the following reaction is 77.5 at 600 K.CO(g) + Cl2(g)…
A: The objective of the question is to calculate the equilibrium concentrations of reactants and…
Q: What is pH with 0.00 mL NaOH added? What is pH 12.50 mL (half- way equivalence point) What is pH at…
A: pHHydrogen potentials are referred to as pH. It indicates the hydrogen ion concentration in a…
Q: Use the balanced equation, 4K + O2-->2K2O , to answer the following question. How many grams of…
A: The objective of the question is to find out how many grams of O2 are needed to react completely…
Q: Urea is prepared by reacting ammonia with carbon dioxide as shown in the reation equation image. In…
A: The objective of this question is to calculate the mass of urea formed when 637.2g of ammonia reacts…
Q: The equilibrium constant, K, for the following reaction is 5.10×10-6 at 548 K. NH4Cl (s)…
A: The objective of the question is to calculate the concentrations of the two gases.
Q: The equilibrium constant, Kc, for the following reaction is 83.3 at 500 K. PCl3(g) + Cl2(g) PCl5(g)…
A: The objective of the question is to determine the equilibrium concentrations of the reactants and…
Q: What is the IUPAC name of the following molecule:
A: According to IUPAC Nomenclature:Benzene derivatives compounds are named by using substituents as a…
Q: Which method would you use to perform these reactions, Grignard carboxylation, nitrile hydrolysis,…
A: Grignard carboxylationExplanation:In all the reactions, Grignard carboxylation is performed. Here,…
Q: Classify each carbocation a. as primary, secondary, or tertiary. b. t.
A: Primary carbocation are attached with 1 carbon atomSecondary carbocation are attached with 2 carbon…
Q: what is the empirical formula of a compound that percentage composition is 2.2% hydrogen, 26.7%…
A:
Q: Suppose that you have an enzyme concentration at the molarity calculated in question 2 and the…
A: To calculate the values of Kcat and KM for the reaction, we can use the Michaelis-Menten…
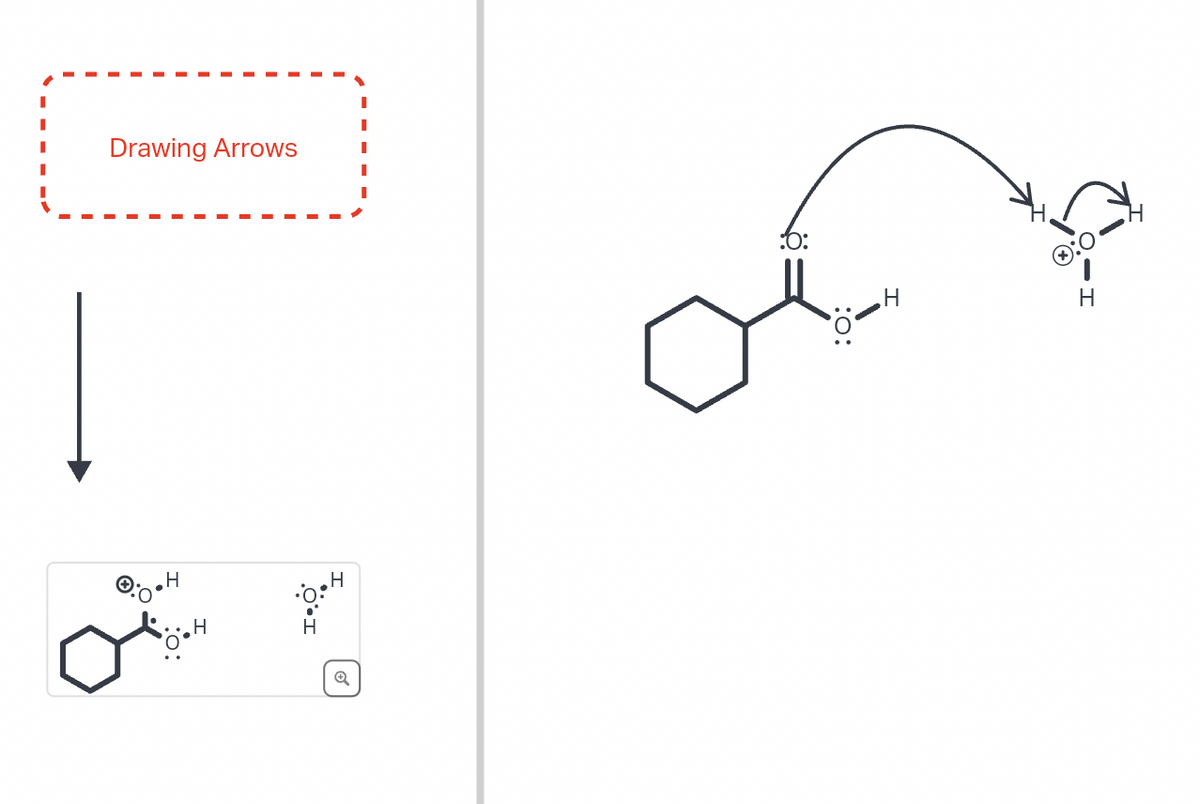
Q: Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and…
A: The objective of the question is to predict the product formed in the following reaction…
Q: Identify the reactants and products in a chemical equation, and complete the tables to indicate how…
A: In a chemical equation, reactants are on the left side of the arrow and products are on the right…
Q: Draw the major organic product of the reaction shown. 1. LDA, THF 2. CH31, HMPA 3. H*, H₂O
A: The alkylation reaction plays a fundamental role in organic synthesis, allowing chemists to…
Q: Calculate the solubility at 25 °C of PbCrO 4 in pure water and in a 0.0180 M Na2CrO 4 solution.…
A: Given that,The concentration of Ksp for Molar mass of Then,
Q: Which of the following two structures are related as resonance structures?
A:
Q: How to obtain 2-bromo-4-nitro-1-(propan-2-yl)benzene from (propan-2-yl)benzene.
A: GivenReactant: (propan-2-yl)benzene or isopropyl benzene Product :…
Q: Calculate the frequency in hertz of electromagnetic radiation that has a wavelength of 680.0 nm. (c…
A: The objective of this question is to calculate the frequency of electromagnetic radiation given its…
Q: Indicate the mechanism of Hofmann degradation of the compound N-(2-phenylethyl)butan-1-amine.…
A: The objective of the question is to understand the mechanism of Hofmann degradation of the compound…
Q: Alkyl halides undergo nucleophilic substitution and elimination reactions. When the kinetics of the…
A: The given mechanism follows E2 mechanism.Explanation:
Q: Identify two ions that have the following ground-state electron configurations.[Ar]
A: The objective of the question is to identify two ions that have the ground-state electron…
Q: E 1. CH,MgBr 2.HIO* 1. Na(CN)BH, pH 4-5 2. HO CHÍNH, NGOH 1. CHICH₂NH2, Na(CN)BHz. pH 4-5 2.…
A: Find out suitable reagent for conversion of the reaction Alkyl cyanide into ketone
Q: Wine goes bad soon after opening because the ethanol CH3CH2OH in it reacts with oxygen gas O2 from…
A: The objective of the question is to find out the mass of ethanol that is consumed by the reaction of…
Q: 1. NaOMe 2. H*
A:
help fix please
Trending now
This is a popular solution!
Step by step
Solved in 1 steps with 1 images

- Which of the following structures are identical? (Green = Cl.)In the pic (yellow circle l). What is the above O means? Is it 5OH^- or 5 O^- H?The instructions are to draw the molecule oriented as : Br going towards me while OH is going away from me. The compound is : CH3CH(Br)CH(OH)CH3 want to make sure I drew it correctly
- __ Al(OH)3 + ___ HBr ------> ___ AlBr3 + ___ H2O Can yo balence this for meWhich bond is more polar?a. H - CH3 or Cl - CH3b. H - OH or H - H c. H - Cl or H - F d. Cl - Cl or Cl - CH3the context is "Cosmone is a molecule used by fragrance manufacturers to provide a rich and elegant musky essence to many perfumes. Cosmone has the molecular formula C15H26O." I need help on part (h).
- sotretinoin, a fatty acid, is a medication used to treat severe acne that has not responded to other drugs. Because it causes fetal abnormalities, it cannot be taken by pregnant women, and its prescription and use are carefully monitored. (a) Label each C = C as E or Z. (b) Label each joined σ bond joining two C = C’s as s-cis or s-trans.What is the approximate H−C−O bond angle in formaldehyde, H2C=O? a. 90° b. 109° c. 120° d. 180° Group of answer choices a b c dA compound of formala ( C20H29NO) absorbs 4 molar equivalents of hydrogen. How many rings does it contain ? select one : A) 3 B) 2 C) 1 D) 4
- Show the direction of the dipole moment in each of the following bonds : a. H3C¬Br b. H3C¬Li c. HO¬NH2 d. I¬Br e. H3C¬OH f. (CH3)2 N¬H12.) Which molecule below can be called hypervalent? Group of answer choices more than one of these choices SnF4 PF3 SeF4 GeF4each reagant must have 3 or less carbons . one image is the directions and one is the question.





