The table shows the variation of the rate constant with temperature for the first-order reaction. 2N205(g) → 2N2O4(g) + O2(g) Determine graphically the activation energy for the reaction. Round your answer to 2 significant digits. T (K) k (1) 298. 1.74 × 10 ·5 308. 6.61 × 10 318. 2.52 x 10 4 328. 7.59 x 10 338. 2.40 x 10 Note: Reference the Fundamental constants table for additional information. kJ mol x10
The table shows the variation of the rate constant with temperature for the first-order reaction. 2N205(g) → 2N2O4(g) + O2(g) Determine graphically the activation energy for the reaction. Round your answer to 2 significant digits. T (K) k (1) 298. 1.74 × 10 ·5 308. 6.61 × 10 318. 2.52 x 10 4 328. 7.59 x 10 338. 2.40 x 10 Note: Reference the Fundamental constants table for additional information. kJ mol x10
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter11: Chemical Kinetics: Rates Of Reactions
Section: Chapter Questions
Problem 39QRT
Related questions
Question
Only typed solution.
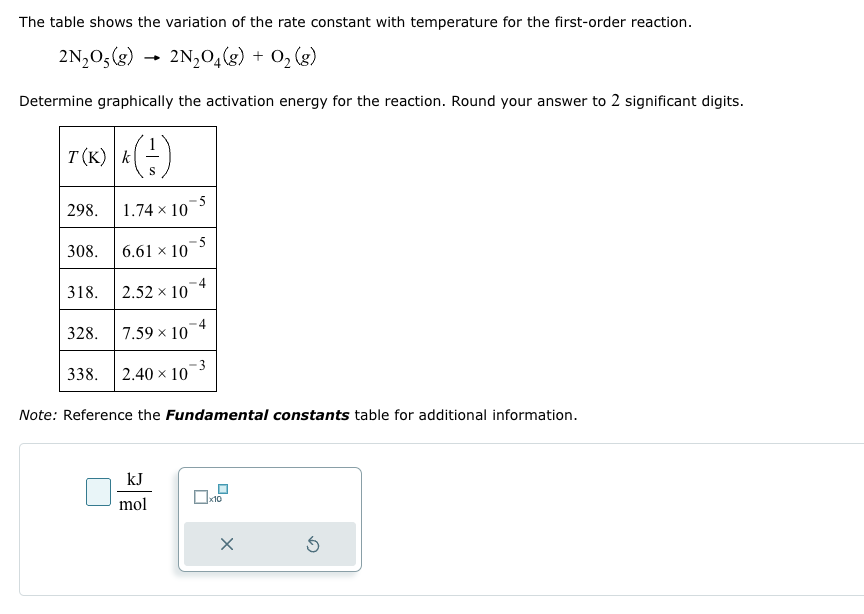
Transcribed Image Text:The table shows the variation of the rate constant with temperature for the first-order reaction.
2N205(g)
→
2N2O4(g) + O2(g)
Determine graphically the activation energy for the reaction. Round your answer to 2 significant digits.
T (K) k (1)
298. 1.74 × 10
·5
308.
6.61 × 10
318.
2.52 x 10
4
328.
7.59 x 10
338.
2.40 x 10
Note: Reference the Fundamental constants table for additional information.
kJ
mol
x10
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 1 steps with 1 images

Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning