Q: Match the lipid pathway with its description Question 27 options: dietary…
A: The objective of the question is to match the lipid pathway with its corresponding description.
Q: Calculate the solubility (in g/L) of CaSO4(s) in 0.450 M Na, SO4(aq) at 25°C. The Ksp of CaSO4 is…
A: Molarity of Na2SO4 = 0.450 MKsp of CaSO4 = 4.9310-5molar mass of CaSO4 = 136.14 g/mol
Q: 1. Calculate the concentration of a solution of H₂SO, with a pH of 1.06.
A: 1) pH = - log [H+] Thus, [H+] = 10-pH
Q: The titration curve below corresponds to a titration of a Hd A B C D Volume titrant added (mL) by a…
A: Answer:Analyte is the solution of unknown concentration that needs to be determined. It is measured…
Q: NH3 is a weak base (K₁ = 1.8 x 10-5) and so the salt NH4Cl acts as a weak acid. What is the pH of a…
A: NH4Cl is a salt of weak base NH3 and strong acid HCl. So in an aqueous solution of the salt the salt…
Q: What mass of precipitate (in g) is formed when 20.5 mL of 0.500 M Cu(NO₃)₂ reacts with 15.0 mL of…
A: The objective of this question is to find out the mass of the precipitate formed when a certain…
Q: A partially submerged tank is leaking 1 g/day of heptachlor into a pond (V = 30,000m3). a) Given a…
A: The objective of this question is to determine the steady-state concentration of heptachlor in the…
Q: How many mL of 0.500 M NaI would be required to make a 0.0490 M solution of NaI when diluted to…
A: The objective of this question is to find out the volume of 0.500 M NaI solution required to make a…
Q: Part A A 150.0 mL buffer solution is 0.100 mol L-1 in (CH3)2NH and 0.135 mol L-1 in (CH3)2NH2 Br.…
A: Answer:Buffer solution is a type of solution that resists the change in its pH on adding small…
Q: 1. What is the pH of a buffer that is 0.20 M HF and 0.085 M KF? pKa = 3.152. What is the pH of a…
A: 1. The concentration of The concentration of 2. The number of moles of The number of moles of The…
Q: OH CH3CH=CHCHCH2CH3 4-hexen-3-ol Four stereoisomers exist for 4-hexen-3-ol. Draw the structure of…
A:
Q: Can you show the stereoisomers and their stereochemistry?
A: Isomers having the same molecular formula and constitution but they differ in their space or…
Q: PLS HELP ASAP ON ALL ASKED QUESTIONS AND SHOW ALL WORK
A: 9. d. mol/L.s 10. d. Both (a) and (b) are correctExplanation:9. The rate of reaction refers to the…
Q: 3. Complete the following table: Fill in the mL used of the two starting materials (from the…
A: The answers and calculations are as follows.Explanation:
Q: Draw the enolate anion and the carbonyl that would be needed to make this product through an aldol…
A: Aldol condensation is the condensation between two aldehdyes or two ketones having at least one…
Q: Bra+light NBS CC Br or Br 2
A: NBS is N-bromosuccinimide. It is a source of cationic bromine for electrophilic addition and radical…
Q: The activation energy of a reaction is 50.5 kJ/mol and the frequency factor is 1.9\times 1011/s.…
A: Arrhenius equation is ln ( k ) = - + ln ( A ) Wherek = rate constantA = frequency factorEa =…
Q: What is a major difference between acid and base catalyzed hydration of a carbonyl?
A: Given question is based on organic chemistry.The given reaction is hydration of ketone.The hydration…
Q: What is the main product of the following reaction? (pay attention to regiochemistry and…
A: The question is based on pericyclic reaction.The Diels-Alder reaction is a type of cycloaddition…
Q: Consider the following chemical equilibrium: H2(g) +CO₂ (g) H₂O(g) +CO (g) Now write an equation…
A: Equilibrium constant is defined as the ratio of concentration of products to the concentration of…
Q: What is the IUPAC name for the compound shown? OHHHH H-C-C-c-c-C-O-H | | HH H H
A: IUPAC rule for carboxylic acid,1) Choose longest chain as parent chain.2) Numbering start from…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 1.03 2. Zn, HOAc Drawing + Q 6…
A: See the answer in the explanation fieldExplanation:
Q: Draw the product of this reaction and account for its formation, providing a curly arrow mechanism…
A: Reaction of benzene ring containing halogen group attached leads to the formation of benzyne as…
Q: IUPAC name: 이 2. 30 H2N ₂N H
A: IUPAC (International Union of Pure and Applied Chemistry) naming, also known as systematic naming or…
Q: Identify the missing reagent(s), reactant(s) or final product(s). Some transformations may require…
A: Given are organic reactions. Note: According to Bartleby Q&A guidelines we are supposed to…
Q: PLS HELP ASAP ON ALL ASKED QUESTIONS AND SHOW ALL WORK
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: Draw the products of this three-step synthesis. Br2 (1 equiv) CH3CO2H 1. (CH3CH2)2 Select to Draw…
A: In acidic medium, carbonyl compounds having enolisable alpha hydrogen undergo bromination reaction…
Q: 2 Na2Cr2.07) H2SO4, H2O HO
A:
Q: Starting with 3-pentanone as your only organic starting material, figure out and draw a series of…
A:
Q: Select the correct final major product. A 1. B 2. 0 3. D 4. + major product CH2MgBr 1. CH₂OH 2. H₂O…
A: The given reaction is based on acid- base reaction.
Q: Propose a plausible mechanism for the following transformation: 1) Excess MeMgBr 2) H₂O HO OH
A: The objective of the question is to predict the product formed in the given reaction.
Q: Assuming complete dissociation, what is the pH of a 3.09 mg/L Ba(OH)2 solution? pH =
A:
Q: For the reaction: 2 A (g) + B (s) ⇌ 2 C (s) + D (g) At 298 K in a 10.0 L vessel, the equilibrium…
A: K = 17.5 Explanation:Given:…
Q: Please help me , i need fast solution. Please don't provide handwritten solution ...
A: The objective of this question is to calculate the pH of a solution of NH3 after the addition of…
Q: 44.0 mL of water was added to 100.0 mL of 0.250 M CaCl₂ solution. Assume the volumes are additive.…
A: The objective of this question is to find the concentration of CaCl2 in the diluted solution after…
Q: At 700 K, 0.500 mol of HI is added to a 2.00 L container and allowed to come to equilibrium.…
A: To solve this problem, we'll use the equilibrium constant expression and the stoichiometry of the…
Q: Please write a paragraph of the objective stated using the two images attached below Please please…
A: Acid-base titrations are analytical methods that are employed in the determination of an unknown…
Q: What CO2 partial pressure (torr) is required to yield a velocity of 0.045 M-s1 for the reaction?…
A: The velocity is given as v = 0.045 M/sIt is required to find the pressure in torr.
Q: Predict the FINAL (?) product for each of the following reaction or synthetic chain: +MgBr 1. 03 ?…
A: Given,The reaction:
Q: In the laboratory a "coffee cup" calorimeter, or constant pressure calorimeter, is frequently used…
A:
Q: Use the reaction quotient to predict whether a precipitate will form. If 15.0 mL of 2.88x10^-4 M…
A: The objective of this question is to apply principles of solubility equilibrium, calculating…
Q: The following is an HPLC separation of deuterated benzenes on a C18 column that 440 cm long. The…
A: 1. mobile phase velocity (<v>) = 10.552 cm/min 2. retention factor (k') of C6D6 is 3.511 3.…
Q: 3. Calculate the pH of a 1.94x103 M solution of CH COOH at 25°C. K, 4.55x100
A: Concentration of acetic acid(CH3COOH) = 1.9410-3 MKa of CH3COOH = 4.5510-10
Q: The cell potential (Ecell) of a galvanic cell made using the redox couples H+/H2 and Zn2+/Zn is…
A:
Q: 1. Predict the product and provide the mechanism for the following reversible nucleophilic addition.…
A: See answer in the explanation fieldExplanation:
Q: PLS HELP ASAP ON ALL ASKED QUESTIONS AND SHOW ALL WORK
A: a. Step 1, i.e., 2B → Eb. Intermediates: E, K, EK and F c. Overall reaction: 2B+2A→D+C d. Catalyst:…
Q: A sheet of gold weighing 9.0 g and at a temperature of 13.8 °C is placed flat on a sheet of iron…
A: The objective of this question is to find the final temperature of the combined metals, gold and…
Q: In the disorder of renal tubular acidosis, patients have difficulty maintaining the pH gradient due…
A: The objective of the question is to identify the cause of difficulty in maintaining the pH gradient…
Q: part a and b please
A: In a Diels-Alder reaction between adieneandadienophile, a cyclic structure is a product. The two…
Q: Suppose a 500. mL flask is filled with 0.80 mol of O2 and 0.30 mol of NO. This reaction becomes…
A: The objective of the question is to find the initial, change, and equilibrium molarity of each…
Step by step
Solved in 3 steps with 3 images

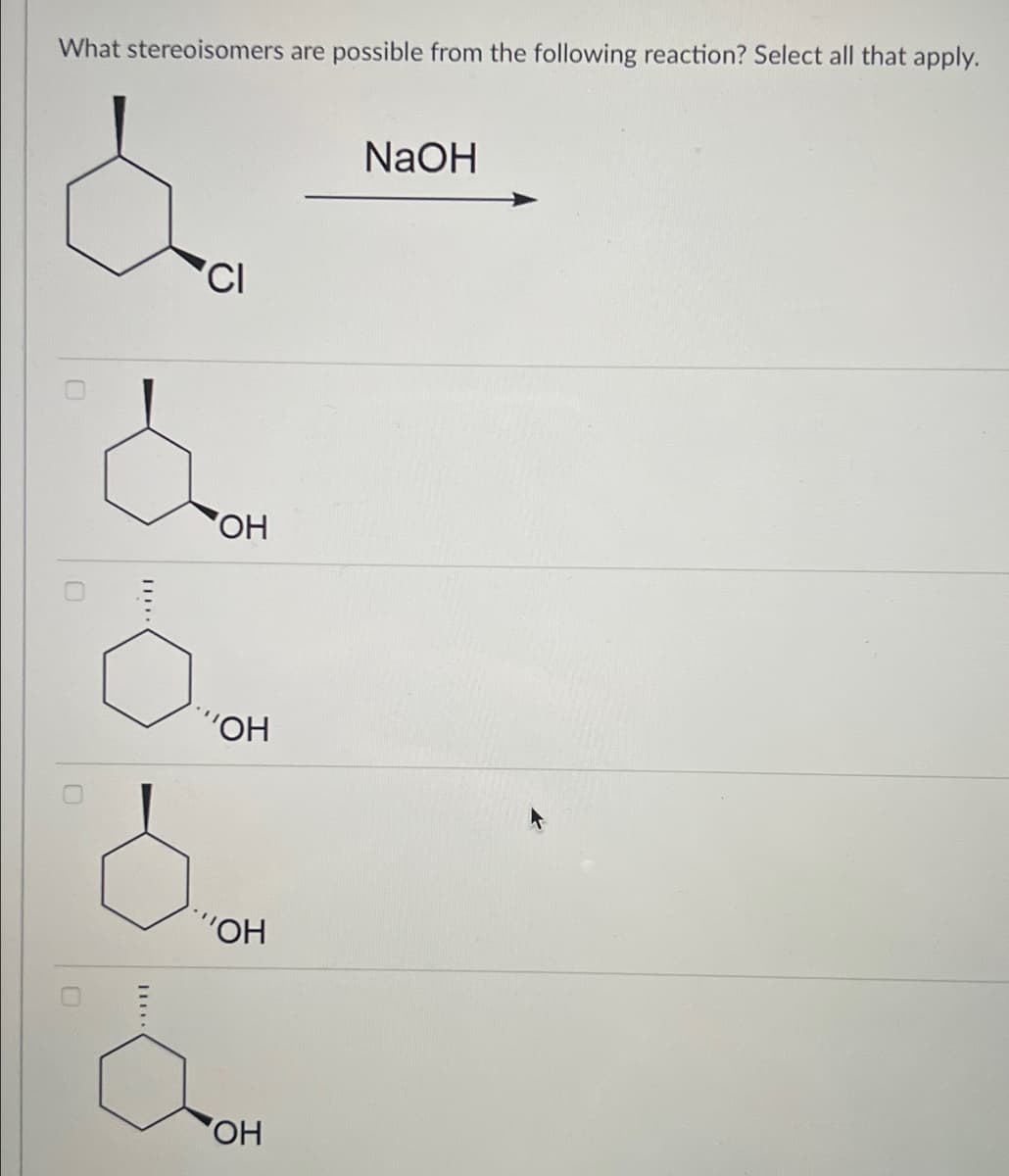
- Pls help ASAP, thank you! "which of the following is the most stable eclipsed conformation of this alkane?"For which double bonds are stereoisomers possible? (See attached)what is the most stable newman conformation around the star bond Please provide only typed answer solution no handwritten solution needed allowed
- How many stereoisomers are possible for the molecule formed by the reaction described in the preceding question? Group of answer choices a. 8 b. 16 c. 32 d. 64 (preceding question is image #2)please do only the highlighted qouestionWhich of the following structures is an E isomer? Use image as reference
- Solve correctly please, with detailed explaination. When solving alkene addition reactions, how do you know which double bond undergoes the addition reaction if there is more than one? (Gpt/Ai wrong answer not allowed)caq5 Organic Chemistry problem. Please help. Thank youneed to draw RR SS meso isomers ,check if my drawing is correct. if not please help to correct them

