Q: The heat of vaporization of water at 100°C is 40.66 kJ/mol. Calculate the quantity of heat that is…
A: Given,mass of steam water = 5.00 gThe heat of vaporization of water at 100 oC = 40.66 kJ/mol
Q: Predict the major product of halogenation of the given alkyne. + 5 P Br 2 Click and drag to start…
A: When one equivalent of the halogen is used the product of the reaction is trans…
Q: What is the IUPAC name for the compound shown? OHHHH H-C-C-c-c-C-O-H | | HH H H
A: IUPAC rule for carboxylic acid,1) Choose longest chain as parent chain.2) Numbering start from…
Q: A student set up a Cu-Fe cell according to the procedure in Part 2. They measured the cell voltage…
A: The temperature is setup at 298 KThe cell voltage average is V = 0.682 V
Q: What is the pH of a buffer that is 120 M in formic acid, HCHO2 and 0.080 M in potassium formate…
A: The objective of this question is to calculate the pH of a buffer solution. The buffer solution is…
Q: Iminodiacetic acid (IDA) is often used in ion exchange resins as a way to remove heavy metals from…
A: The formation constant for a reaction represents the equilibrium constant for the formation of a…
Q: Propose an appropriate base to carry out the transformation below and use it as part of a curly…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: The area of a 15-inch pizza is 153.4 in. Express this area in square centimeters. (1 in 2.54 cm)…
A: The area of a 15 inch pizza is 153.4 in2Convert the area in cm2
Q: Question Aqueous hydrobromic acid HBr reacts with solid sodium hydroxide NaOH to produce…
A: Maas of HBr = 2.4 gMass of NaOH = 1.6 gMass of H2O produced = 0.155 g
Q: 9. Part 1: Draw the structure for compound A. Part 2: Draw the structure for compound B. Part 3:…
A: We have to complete the synthesis of the given transformation.
Q: A correct IUPAC name for the following compound is: 3,3,5-trimethyl-2-hexene…
A: IUPAC rule for alkene,1) Choose longest chain as parent chain.2) Numbering start from those carbon…
Q: Find the order of reaction when half life of a reaction is found to be inversely proportional to the…
A: Given that the half-life of the reaction is inversely proportional to the sixth power of its initial…
Q: Cyclopentadienone is a highly reactive ketone. Draw an anti- aromatic resonance structure that…
A: The compound should have internal conjugation with a total no. of 4n pi electrons.Anti aromatic is…
Q: A compound containing Na, C and O is found to have 1.06 mol Na, 0.528 mol C, and 1.59 mol O. What is…
A: The empirical formula of a compound gives the simplest ratio of the number of different atoms…
Q: I am stuck on question 4, I found everything
A: The objective of the question is to determine the weight in grams and weight percentage of magnesium…
Q: A 20.0 mL sample of 0.150 M ethylamine CH3CH2NH2 is titrated with 0.0981 M HCl. What is the pH after…
A: The objective of this question is to calculate the pH of a solution of ethylamine before any HCl is…
Q: Please draw all possible resonance structures, provided with an explanation
A: Please see attachment below for resonance structure.Explanation:Step 1: Resonance structures in the…
Q: edict the product of this organic reaction: OH + OH P+H₂O Specifically, in the drawing area below…
A: Carboxylic acid reacts with alcohol to form an ester in the presence of an acid catalyst, the…
Q: 4. Draw the major product(s) for each reaction. Label all reactants and products (R,S,E,Z) when…
A: Given is organic reaction. The given reaction is Hoffman reaction. Amines undergo Hoffman reaction…
Q: This chapter covered many C-C bond-forming reactions, including aldol reactions, Claisen…
A: Given reaction is an example of intramolecular nucleophilic addition reaction of carbonyl compounds.
Q: You wish to know the enthalpy change for the formation of liquid PCl3 from the elements. P4(s)+6…
A:
Q: The half-equivalence point of a titration occurs half way to the equivalence point, where half of…
A: Given,moles of monoprotic weak acid = 0.480 molKa of acid = 5.3 x 10-5
Q: Please predict the products for each of the following reactions. Hint: one of the reactions is the…
A:
Q: What CO2 partial pressure (torr) is required to yield a velocity of 0.045 M-s1 for the reaction?…
A: The velocity is given as v = 0.045 M/sIt is required to find the pressure in torr.
Q: Rank the three compounds in order of increasing heat of hydrogenation. The smallest heat of…
A: We have to provide the order and predict the product.
Q: What is the major neutral organic product for the following sequence? 1. KÊN 2. H+ 3. H3O+, heat ?…
A: -> Carbonyl compounds have electrophilic center and KCN has nucleophilic part which can give…
Q: Which is the major organic product formed by this reaction? Studying the summary chart in Lesson…
A: -> NaBH4/CH3OH is reducing agent which can reduce carbonyl compounds(aldehydes and ketones) to…
Q: Draw the structure of the starting material A for the reaction below when one equivalent of H2/Pt is…
A: The reagent used is H2 (1 equiv.) in the presence of Pt catalyst.The product formed is…
Q: %Transmittance 100 M 4000 'H NMR 300 MHz -3344 2958 2931 2873 3000 2000 Wavenumber (cm-1) -1466…
A: The objective of the question is to analyze the given spectrum and predict the structure of the…
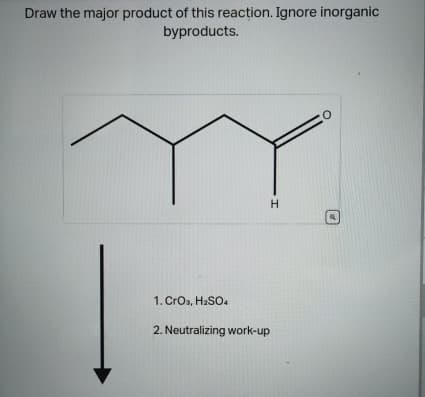
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the carboxylic acid side…
A: Given,The reaction:
Q: 11. Treatment of compound F with lithium diisopropylamide followed by cyclohexanone gives either G…
A: The objective of this question is to elucidate the mechanisms behind the formation of ethers via…
Q: Calculate the pH when 20.0 mL of 0.150 M KOH is mixed with 26.0 mL o 0.300 M HBrO (Ka = 2.5 × 109)
A:
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A: The objective of this question is to balance the given redox reaction under acidic conditions and…
Q: Vanadium is a transition metal that can assume multiple different positive charges. Vanadium forms…
A: The objective of the question is to determine the formula and the name of two vanadium oxides, given…
Q: When heated, which of these will give a product which has a molar mass exactly lower by 44? OH LOH…
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: What mass of precipitate (in g) is formed when 20.5 mL of 0.500 M Cu(NO₃)₂ reacts with 15.0 mL of…
A: The objective of this question is to find out the mass of the precipitate formed when a certain…
Q: Select the correct final major product. A 1. B 2. 0 3. D 4. + major product CH2MgBr 1. CH₂OH 2. H₂O…
A: The given reaction is based on acid- base reaction.
Q: 8. Use the following reactions and Draw the transition state in the following reactions: CH3 CH3 a)…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: 2. H₂O ? H-NO $croyCl Na₂Or 07 H.80%
A: Given,The reaction:
Q: 3) Give the products of the reactions. O=C=N- Ph -N=C=O n HO OH (HCI), H2O, A
A: To write the products of the given reactions.
Q: A 1.00-mol sample of an ideal monatomic gas is taken through the three-stage cycle as shown The…
A:
Q: Predicting the reactants or products of esterification hat is the missing reactant in this organic…
A: Esters is formed by react carboxylic acid and alcohol as in acidic medium.
Q: (A)Write the Hückel Hamiltonian matrix for benzene. (B) The pictures below represent a top view of…
A: The objective of the question is to write the Huckel Hamiltonian matrix for benzene and to label the…
Q: Use the reaction quotient to predict whether a precipitate will form. If 15.0 mL of 3.74×104 M Cal2…
A: The objective of the question is to predict whether the precipitate will occur or not.
Q: HO 777 ΤΗΝ
A: The objective of the question is to complete the given reaction by explaining each step of preparing…
Q: P1A.12* Balloons are still used to deploy sensors that monitor meteorological phenomena and the…
A: (a) Volume of balloon is:
Q: Please write the procedure in your own words Please write it in a paragraph Please please please…
A: The objective of this question is to evaluate the laboratory procedure for determining the…
Q: Q.10 5-Hydroxy-1,3-dioxane is more stable with the OH group in the axial position than in the…
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: Explain why the addition of bromine to compound A gives a product in which the two bromine atoms…
A: In both compounds, the addition of bromine to double bond results in the formation of the…
Q: What is the major organic product in the following sequence of reactions? i TBDMS-CI 1. CH₂MgCl TBAF…
A: The objective of the question is to identify the major organic product resulting from the given…
Please don't provide handwritten solution ...
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- Answer is Reaction 7? Can you explain to me, please?Draw the major product of this reaction. Ignore inorganic byproducts and the alkoxide side product.Draw the major product of this reaction. Ignore inorganic byproducts. Assume thatthe water side product is continuously removed to drive the reaction towards products. Reagent= (CH2OH)2, TsOH
- Use the product of the next reaction product. (d) please!!Draw the products of each reaction. Assume excess halogen is present.1. what reaction and meachanism is this ? 2. Explain mechanism what happenning in each step on this picture 3. why is it says "stop" when NaOEt added ? does reaction stop there ? why does it stop ? ? what principle ?

