Q: Question 1. Indicate at which position(s) an electrophilic substitution will take place and justify…
A: When all the properties of a molecule cannot be explained by a single structure, we draw more than…
Q: An equilibrium mixture contains 0.350 mol of each of the products (carbon dioxide and hydrogen gas)…
A: The equilibrium reaction is:CO(g) + H2O(g) CO2(g) + H2(g)Moles of CO2(g) and H2(g) = 0.350 molMoles…
Q: Which of the following is the required starting material for the given reaction? ? Ph₂CuLi Ph Br…
A: A Gilman reagent is an organometallic compound with the formula R2CuLi, where R is an alkyl or aryl…
Q: 15. What is the major organic product obtained from the following reaction? OH MgBr 1. 2. H3O+ 3 LOH…
A: Given,The reaction is:
Q: 74) Identify the reagents of product required. Add any Side products not shown. a) C b) AICI AICI…
A: Electrophilic aromatic substitution:Halogenation: Benzene on reaction with halogen in presence of…
Q: A solution contains 0.021 M Cl- and 0.017 M I. A solution containing copper (I) ions is added to…
A: Solubility product is defined as the product of concentration of cations and anions formed at…
Q: Compound ∆Hf (kJ/mole) ∆S (J/mole K) CO2(g) –393.5 +213.8 CO2 (aq) –413.8 +117.6…
A: The objective of the question is to determine whether the given reactions are spontaneous or…
Q: Consider the reaction of a 20.0 mL of 0.220 M C H NHCI (Ka = 5.9 x 10°) with 12.0 mL of 0.237 M…
A: Answer:When acid and base solutions are mixed then neutralization reaction takes place and its…
Q: A molecule phosphoresces with a single peak wavelength of 700 nm. The single fluorescence peak is at…
A: The answer is given in attachment below.Explanation:Step 1:Step 2: Step 3: Step 4:
Q: Please don't provide handwritten solution ...
A: The objective of this question is to calculate the pH of an aqueous solution of potassium acetate.…
Q: balance each of the following equations. Show all coefficients including "one". Classify each…
A: 1. H₂ + O₂ → H₂OBalanced equation: 2H₂ + O₂ → 2H₂OClassification: Combination 2. N₂H₄ → NH₃ +…
Q: Rank the three compounds in order of increasing heat of hydrogenation. The smallest heat of…
A: We have to provide the order and predict the product.
Q: or products of amidation What is the missing reactant in this organic reaction? R+ NH2 A N + H₂O о…
A:
Q: Calculate solubility in the presence of a common ion. Calculate the solubility of PbCl 2 (a) in pure…
A:
Q: Wine goes bad soon after opening because the ethanol (CH, CH₂OH) in it reacts with oxygen gas (0)…
A: We will use the balanced equation for the calculation of mass of consumption of ethanol with given…
Q: Draw step 3 of the mechanism. CH3 CH3 H₂C- H₂C CH2 CO H3C CH3 CH3 CH3
A: The given reaction scheme is shown below.We have to draw the mechanism of the above reaction.
Q: Use the reaction quotient to predict whether a precipitate will form. If 15.0 mL of 3.74×104 M Cal₂…
A: Given:Calculate concentrations of ions in solution after mixing solutions of different salts…
Q: If wavelength of photon is 2.2 × 10-11m and h = 6.6 × 10-34Js, then momentum of photon
A:
Q: Only typed solution.
A: Ea = 1.0x102 kJ/molExplanation:Temperature…
Q: Identify the mechanism by which each of the reactions above proceeds from among the mechanisms…
A: Given are organic reactions. The starting compound of reaction 1 is alcohol.The starting compound of…
Q: 2. H₂O ? H-NO $croyCl Na₂Or 07 H.80%
A: Given,The reaction:
Q: Propose an appropriate base to carry out the transformation below and use it as part of a curly…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: 2. Predict the major product(s) of the following reactions: (a) CH3CH2C AICI3 ? (b) O CH3CH2COCI…
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: Free energy What might the reaction coordinate diagram look like for the following reaction? ii Ryn…
A: The nucleophilic substitution reaction in which a nucleophile group displace a group bonded at…
Q: Prepare a report (formatting specifications below) outlining the 2020 COVID-19 drug authorization…
A: The objective of this question is to understand the 2020 COVID-19 drug authorization process in…
Q: Determine the pH of a solution that is 1.30% NaOH by mass. Assume that the solution has a density of…
A: Calculation of moles of NaOHGiven, mass of NaOH = 1.30 % by mass which means 1.30 g of NaOH present…
Q: Calculate the pH when 20.0 mL of 0.150 M KOH is mixed with 26.0 mL o 0.300 M HBrO (Ka = 2.5 × 109)
A:
Q: Organic Chemistry problem. Please help. Thank you.
A: The required answer is given below Explanation:Step 1:This reaction is known as Baeyer Villiger…
Q: Calculate solubility in the presence of a common ion. Calculate the solubility of PbCl 2 (a) in pure…
A: We have to calculate the solutbility.
Q: Synthesize this from benzene
A: In summary, a benzene reactant can form a meta directed bromine and amine group through the…
Q: Draw the product of the reaction shown below. Ignore inorganic byproducts. HзN. BOC-Ala DCC Drawing
A: The given reaction is shown below.We have to provide the major product of the given reaction.
Q: Consider the reaction: H2O+ HCI H₂O + CI Which of the following molecular orbital pictures…
A: In the Lewis structure of , oxygen has a formal charge of +1, and the molecule has a trigonal…
Q: gives either G or H. G is formed if the aldehyde is added at -78°C whereas H is formed if the…
A: The objective of this question is to explain the processes behind the aldol condensation and…
Q: LOH + pK = 9.9 CH,0 which side does the equilibrium lie? Right side Left side. Not enough…
A: Given an acid-base reaction.Determine whether the equilibrium lie.
Q: 2. Explain using mechanisms the regiochemistry and stereochemistry observed for the following…
A: A more stable carbocation intermediate is formed during the regioselective mechanism of HBr's free…
Q: Draw the final organic product for this reaction: NaOH
A: The objective of this question is to draw the product formed from the reaction given.
Q: Will KCIO precipitate when 20 mL of a 0.050 M solution of K is added to 80 mL of a 0.50 M solution…
A: Given data:Concentration of K+ ions = 0.050 M,Concentration of ClO4- ions = 0.50 M,Volume of K+ ions…
Q: The natural product (-)-N-methylwelwitindolinone C isothiocyanate is isolated from a blue-green…
A: This is an example of benzyne mechanism. In this reaction there is dehydrohalogenation to produce…
Q: What is the major organic product in the following sequence of reactions?...
A: The objective of this question is to choose the correct organic product obtained from the reaction.
Q: Which of the following describes the reaction shown? OH O neither O reduction O oxidation
A:
Q: The alkyne below is correctly drawn with correct geometries for all atoms
A: AlkaneC-Csp3108.5oTetrahedralAlkeneC=Csp2120oTrigonal planarAlkyneC≡Csp180oLinear
Q: 5. Draw the approximate isotope patterns (M, M+1, and M+2 only) for the molecular ions of the…
A: Isotopic abundance of the atoms present in the given compounds:Carbon: It has 2 major isotopes. 12C:…
Q: Predict the major product for the reaction. اف H Cl2, CH3CH2OH
A: Let us discuss the reaction mechanism to find out the products.
Q: If a solution contains 0.0020 mol CrO2 of per liter, what concentration of Ag ion must be reached by…
A: The dissociation of solid Ag2CrO4 isThe solubility product Ksp expression is
Q: Select the correct final major product. (anhydre = anhydrous, no water present) A Compound I ☑…
A: We have to choose the correct option.
Q: Draw the molecular orbital (MO) electron diagram for the Be2 molecular ion. Be sure your diagram…
A: According to the molecular orbital theory the atomic orbitals combine to form molecular orbitals.…
Q: Draw the major organic product of the reaction shown below. + PBг3 OH
A: Answer:PBr3 has a phosphorous atom that has vacant d-orbitals which makes it an electrophilic…
Q: Draw the aromatic compounds named below. o-bromophenol Draw Your Solution
A:
Q: 1) A) Show the ionization reaction for the side chain of cysteine. B) Why does the arginine side…
A: The objective of the question is to understand the ionization reaction of the side chain of…
Q: VO 4^-3 + C2 O 4^-2 ---->VO ^+1 + CO 2 (acidic) Solve and balance this redox equation
A: The objective of this question is to balance the given redox equation in an acidic medium. The redox…
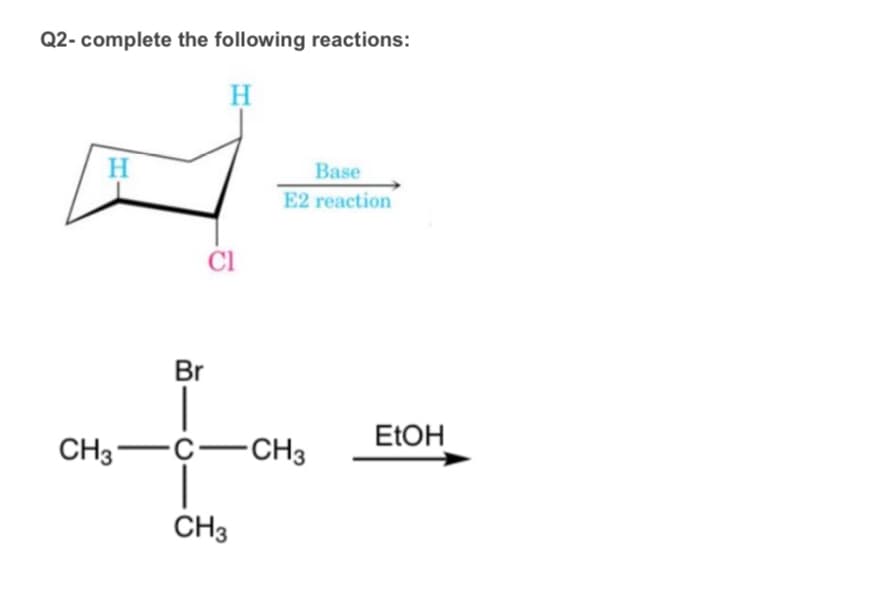
Step by step
Solved in 1 steps with 1 images

- reactions and products for C9H8O4 C9H8O4 + O2 --> C9H8O4 + H2O --> C9H8O4 + HCl --> C9H8O4 + OH- --> C9H8O4 + Na --> C9H8O4 + F -->The following reaction has a ΔSsystem < 0 O2(g) → 2O(g) T or F can you explain why this is false?Complete the following reaction equations :(i) C6H5Cl + CH3COCl →(ii) C2H5NH2 + C6H5SO2Cl →(iii) C2H5NH2 + HNO2 →
- N(CH2CH3)3 + HNO3 --------> a.) rewrite the reaction using bond-line structure of reagents and products of the reaction b.) supply the curved arrows explaining the mechanism of the reactionComplete the following reactions, with the major organic product. A. CH3-CH=CH-CH3 +KMnO4 + H2O ---> B. CH3CH2-CH=CH-CH2CH3 + H2SO4 --->predict the products: XCL2+ E3PO4 --->
- KMnO4, warm, conc'd reacts with hept-1-ene to yield __________. CO2, hex-1-ene CO2, hexanoic acid Formic acid, pentanoic acid Ethanoic acid, pentanal Formic acid, hexanoneWhat products would ypu obtain from reaction of 2,4-dimethyl-2-pentene with BH3, followed by H2O2, OH-? Show its complete reaction mechanism.Is the reagent for all elimination reactions conc. H2SO4? or just the reactions ones with OH?



