Q: Q 16.23: The following reaction will produce a(n) H RCO₂H A ether. B carboxylic acid. C ester. D…
A: The objective of the question is to predict the product formed in the following reaction given.
Q: Maleic anhydride reacts with 2,3-dimethyl-1,3-butadiene to give a single product, C10H1203, in more…
A: This is an example of Diels-alder reaction. In this reaction a conjugated diene undergoes…
Q: On the basis of this scheme, write down expressions for the net rates of change of theconcentrations…
A: The objective of the question is to derive the rate expressions for the concentrations of various…
Q: 口味 Will the carbocation intermediate below undergo a rearrangement? If it will, change the drawing…
A: Carbocations are electron-poor and typically have three substituents. The carbon atom is sp2…
Q: When 173. mg of a certain molecular compound X are dissolved in 100 g of cyclohexane (C6H12), the…
A: Answer:For a non-electrolyte and non-volatile solute dissolved in a solvent, depression in freezing…
Q: OTf KI EtOH, 35 °C
A: The reaction is taking place between C6H5CH(OTf)CH2CH3 (benzyl trifluoromethanesulfonate) and KI…
Q: Use the reaction quotient to predict whether a precipitate will form. If 15.0 mL of 3.74×104 M Cal₂…
A: Given:Calculate concentrations of ions in solution after mixing solutions of different salts…
Q: Can you please answer this?
A: Given:E=−0.525 VE∘ (standard cell potential) = 0.244 VEAg+/Ag∘ (standard reduction potential for…
Q: After running various experiments, you determine that the rate law for the following reaction…
A: Please see attachment below for mechanism.Explanation:Step 1: Step 2:Above reaction followed by an…
Q: Devise a biosynthesis for the natural product (II) from an appropriate starter co-enzyme A and…
A: The image you sent depicts a biosynthetic pathway for a natural product (II) using a starter…
Q: 13. The Ksp of SrCO3 1.6 x 109. What is the solubility of SrCO, (in mol/L) in pure water?
A: Since you have asked multiple questions, we will answer the first question for you. If you want any…
Q: (A)Write the Hückel Hamiltonian matrix for benzene. (B)The pictures below represent a top view of…
A: The objective of the question is to write the Huckel Hamiltonian matrix for benzene and to label the…
Q: A solution contains 1.06×102 M lead acetate and 6.05×103 M magnesium nitrate. Solid potassium…
A: The question is based on the concept of solubility product principle.It states that when a weak…
Q: HO OH cat. H2SO4 H₂O cat. H2SO4 Complete both mechanisms above. BRIEFLY explain how the different…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Consider the SN2 reaction between the reactants shown below. ö:
A: Answer:Nature of alkyl halideStrength of nucleophilestability of leaving groupType of solvent used…
Q: Free energy What might the reaction coordinate diagram look like for the following reaction? ii Ryn…
A: The nucleophilic substitution reaction in which a nucleophile group displace a group bonded at…
Q: Draw a major resonance contributor of this enamine. Include all lone pairs in your structure.
A: The objective of this question is to draw the resonating structures for the given enamine compound.
Q: Malic acid, (HO2C)CH2CH(OH) (CCH) has two acidic protons. What is the pKa for the following…
A: The pKa value is a measure of acidic strength of a solution. The higher the pKa value, the lower the…
Q: Draw the major substitution products you would expect for the reaction shown below. If substitution…
A: Organic reactions are those in which organic reactants react to form organic products. The objective…
Q: Provide the curved arrow mechanism for the electrophile elimination reaction between the given…
A: In an elimination reaction carbon carbon double bond is formed by breaking of two Sigma bonds.
Q: What are the effects of aerosols on climate change?
A: An aerosol is a colloid of fine solid particles or liquid droplets, in air or another gas.
Q: Which of the following compounds canot undergo elimination upon heating with NaOMe? A Br B OTS CI C…
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: A reaction shown below, which of the following could be the possible intermediate for it? OH (A) H₂O…
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: Draw a reasonable mechanism for the following transformation: For the mechanism, draw the curved…
A: The given reactant is a carbonyl compound and the reagents are sodium hydroxide, water and heat. The…
Q: 16.20 Give an acceptable name for each carbonyl compound depicted in the ball-and-stick models. a.…
A: a.The ball-and-stick models of the carbonyl compound is given below :The above carbonyl compound is…
Q: Organic Chemistry Problem. Please help with reaction question. Thank you.
A: Sequence of (A) and (C) is not converted from cyclohexane to cyclohexane…
Q: Draw the product of the reaction shown below. Ignore inorganic byproducts. HзN. BOC-Ala DCC
A: This is an example of DCC mediated acid-amine coupling reaction.
Q: - Write the systematic (IUPAC) names for the molecules. H3C IUPAC name: CH3 H3C CH3 IUPAC name:
A: The objective of the question is to find the IUPAC (International Union of Pure and Applied…
Q: The hydrocarbon whose formula is shown was subjected to complete hydrogenation. Use the given data…
A: The objective of the question is to determine the number of rings and pi bonds in the reactant…
Q: Which of the following accurately describes the major species in solution at point C on the…
A:
Q: Consider the reaction of a 20.0 mL of 0.220 M C H NHCI (Ka = 5.9 x 10°) with 12.0 mL of 0.237 M…
A: Answer:When acid and base solutions are mixed then neutralization reaction takes place and its…
Q: 1. What can you tell from the structure of the amino acid above? Select one:
A: Amino acids are building blocks of proteins. All amino acids contain one carboxyl group and one…
Q: Find the amount of energy given to the 4 He nucleus in the reaction 3 n +³ He →ª He + y, using the…
A: The reaction is given as:
Q: Free energy What might the reaction coordinate diagram look like for the following reaction? Ryn…
A: This is a nucleophilic substitution reaction at carbonyl center. Carbonyl carbons are electrophilic…
Q: Please predict the products for each of the following reactions. Hint: one of the reactions is the…
A:
Q: State your calculated average cell voltage for the Ag-AgBr cell. Using this value, and 298 K as the…
A: The objective of the question is to calculate the standard cell potential and the solubility product…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 1. (Ph)2CuLi 2. H3O+ Drawing Q
A: Major product is given below. Explanation:Step 1: Step 2: Step 3: Step 4:
Q: please DRAW the carbene mechanism for the formation of this five membered ring. Please Draw. H3CO2C…
A: The objective of this question is to describe the carbene mechanism for the formation of a…
Q: draw the product of the reactions shown below at physiological pH (7.4). Ignore inorganic…
A: Product of the reaction shown in explanation section.Explanation:
Q: Part A MISSED THIS? Watch KCV: Finding the [H3O+] and pH of Strong and Weak Acid Solutions IWE:…
A: Given that,Ka for Part A) The concentration of Part B) The concentration of pH of the solution in…
Q: Consider the SN2 reaction between the reactants shown below.
A: Answer:S in sn2 represents substitution, n represents nucleophilic and 2 means bimolecular, so, sn2…
Q: Identify the mechanism by which each of the reactions above proceeds from among the mechanisms…
A: Given are organic reactions. The starting compound of reaction 1 is alcohol.The starting compound of…
Q: Write the name of the following compound. (You need to mention R or S with its name) H CH3 HO "Cl…
A: The rules for naming enantiomers are given as follows:In a substituted product, the substituent with…
Q: Only typed solution.
A: Ea = 1.0x102 kJ/molExplanation:Temperature…
Q: Cyclopentadienone is a highly reactive ketone. Draw an anti- aromatic resonance structure that…
A: The compound should have internal conjugation with a total no. of 4n pi electrons.Anti aromatic is…
Q: Which reagent was used to convert 3-pentanone to the molecule shown below? О a) ammonia b) hydrazine…
A: Carbonyl compounds react with ammonia derivatives to form imine derivatives. The reaction proceeds…
Q: S 702 t N02 C2H4 20 vl mil A, 02 Free 40 NO2 CH2N2 NO2
A: alkenes act as both electrophiles and nucleophiles. In case of cyclo-addition reaction, periferial…
Q: What is the major organic product in the following sequence of reactions? i TBDMS-CI 1. CH₂MgCl TBAF…
A: The objective of the question is to identify the major organic product resulting from the given…
Q: Organic Chemistry problem. Please help. Thank you.
A: Alkynes undergo ozonolysis to give acid anhydrides or diketones. If water is present in the…
Q: Antigen/Antibody (Ag/Ab) combination assays can detect HIV infection on average______ days earlier…
A: The objective of the question is to determine how much earlier an Antigen/Antibody (Ag/Ab)…
Step by step
Solved in 1 steps with 3 images

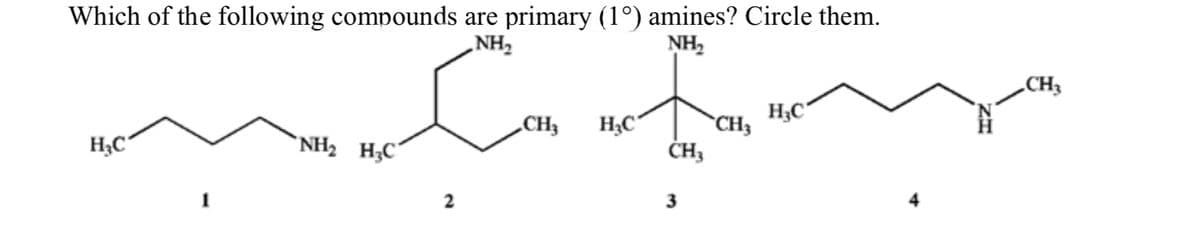
- Draw and name compounds that meet the following descriptions: a. Two primary, 2 secondary, and 2 tertiary amines having the formula C5H13NArrange the following amines in order of increasing boiling point, lowest bp to highest bp:(CH3)2CHCH2CH2NH2, (CH3)2CHN(CH3)2, and (CH3)2CHCH2NHCH3.1. Classify each of the following compounds as either a primary, secondary, or tertiary amine:
- 1. Which statement best described the ability of amines to hydrogen bond? A. Primary, secondary and tertiary amines can all hydrogen bond with molecules identical to themselves. B. Primary and secondary amines can hydrogen bond with molecules identical to themselves, but tertiary amines cannot. C. Only primary amines can hydrogen bond with molecules identical to themselves. Secondary and tertiary amines cannot. D. Primary, secondary and tertiary amines cannot hydrogen bond with molecules identical to themselves, but the can hydrogen bond with water.Which of the following amines will not show intermolecular hydrogen bonding? a) CH3NH2 b) (CH3)2NH c) (CH3)3N d) all of theseClassify the amine below by its degree. H3C-NH2-CH3 Select the correct answer below: primary amine secondary amine tertiary amine quaternary amine
- Which 13C chemical shift may indicate the presence of an amine functional group? δ 110 – 120 δ 40 – 60 δ 10 – 20 δ 150 - 160WHich of the following molecules are all amines based off? H20 CO2 NO3 NH3* The compound CH3CH2NH2 is classified as a A) primary amine. B) secondary amine. C) tertiary amine. D) quaternary amine. E) hydrated amine. * Which of the following compounds is an amine? A) (CH3CH2)2NH B) CH3CH2CH2CH2CO2CH3 C) CH3CH2CH2CH2-O-CH2CH2CH3 D) CH3CH=O E) CH3COCH3





