2.2. An engine uses a high capacity to flow coolant around the engine and through the engine to heat up faster a positioned in the cooling system. When cold, che To assist the is is closed allowing coolant to flow around the engine only.
2.2. An engine uses a high capacity to flow coolant around the engine and through the engine to heat up faster a positioned in the cooling system. When cold, che To assist the is is closed allowing coolant to flow around the engine only.
Principles of Heat Transfer (Activate Learning with these NEW titles from Engineering!)
8th Edition
ISBN:9781305387102
Author:Kreith, Frank; Manglik, Raj M.
Publisher:Kreith, Frank; Manglik, Raj M.
Chapter7: Forced Convection Inside Tubes And Ducts
Section: Chapter Questions
Problem 7.1DP
Related questions
Question
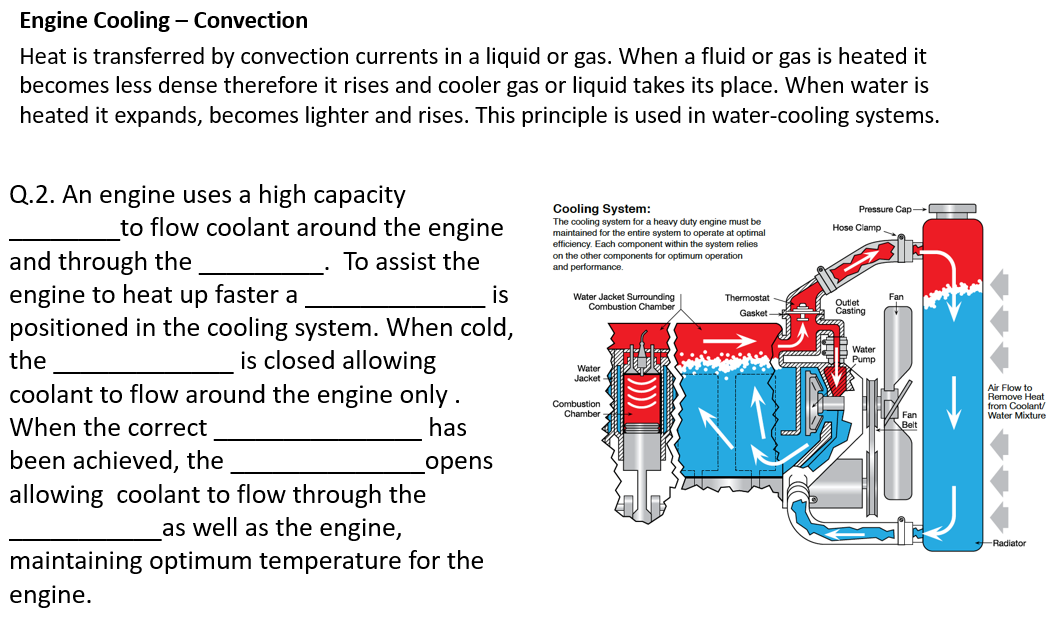
Transcribed Image Text:Engine Cooling – Convection
Heat is transferred by convection currents in a liquid or gas. When a fluid or gas is heated it
becomes less dense therefore it rises and cooler gas or liquid takes its place. When water is
heated it expands, becomes lighter and rises. This principle is used in water-cooling systems.
Q.2. An engine uses a high capacity
Cooling System:
The cooling system for a heavy duty engine must be
maintained for the entire system to operate at optimal
efficiency. Each component within the system relies
on the other components for optimum operation
and performance.
Pressure Cap-
to flow coolant around the engine
Hose Clamp.
and through the
engine to heat up faster a
positioned in the cooling system. When cold,
To assist the
is
Water Jacket Surrounding
Combustion Chamber
Thermostat -
Outlet
Casting
Gasket
the
is closed allowing
B Water
Pump
Water
Jacket
coolant to flow around the engine only.
Air Flow to
Remove Heat
from Coolant/
Water Mixture
Combustion
Chamber
Fan
When the correct
has
been achieved, the
opens
allowing coolant to flow through the
as well as the engine,
-Radiator
maintaining optimum temperature for the
engine.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Heat Transfer (Activate Learning wi…
Mechanical Engineering
ISBN:
9781305387102
Author:
Kreith, Frank; Manglik, Raj M.
Publisher:
Cengage Learning

Refrigeration and Air Conditioning Technology (Mi…
Mechanical Engineering
ISBN:
9781305578296
Author:
John Tomczyk, Eugene Silberstein, Bill Whitman, Bill Johnson
Publisher:
Cengage Learning

Principles of Heat Transfer (Activate Learning wi…
Mechanical Engineering
ISBN:
9781305387102
Author:
Kreith, Frank; Manglik, Raj M.
Publisher:
Cengage Learning

Refrigeration and Air Conditioning Technology (Mi…
Mechanical Engineering
ISBN:
9781305578296
Author:
John Tomczyk, Eugene Silberstein, Bill Whitman, Bill Johnson
Publisher:
Cengage Learning