20 400 K, 4 -0.03 cm 15 400 K, 4 -0.1 cm mol g-cates 400 K, 4 -0.6 cm 10 400 K, d -0.8 cm 300 K, d. -0.3 cm 300 K, d, -0.8 cm 500 1000 1500 2000 2500 3000 3500 4000 4500 Fro (molis)
20 400 K, 4 -0.03 cm 15 400 K, 4 -0.1 cm mol g-cates 400 K, 4 -0.6 cm 10 400 K, d -0.8 cm 300 K, d. -0.3 cm 300 K, d, -0.8 cm 500 1000 1500 2000 2500 3000 3500 4000 4500 Fro (molis)
Introduction to Chemical Engineering Thermodynamics
8th Edition
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Chapter1: Introduction
Section: Chapter Questions
Problem 1.1P
Related questions
Question
Concept problem:
The reaction
A⎯⎯→ B
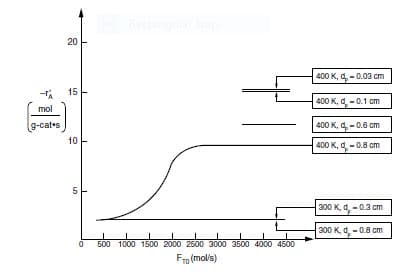
is carried out in a differential packed-bed reactor at different temperatures, flow rates, and particle sizes. The results shown in Figure P15-3B were obtained.
(a)What regions (i.e., conditions dp,T,FT0) are external mass transferlimited?
(b)What regions are reaction rate–limited?
(c)What region is internal-diffusion-controlled?
(d)What is the internal effectiveness factor at T=400 K and dp=0.8 cm?
Transcribed Image Text:20
400 K, 4 -0.03 cm
15
400 K, 4 -0.1 cm
mol
g-cates
400 K, 4 -0.6 cm
10
400 K, d -0.8 cm
300 K, d. -0.3 cm
300 K, d, -0.8 cm
500 1000 1500 2000 2500 3000 3500 4000 4500
Fro (molis)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to Chemical Engineering Thermodynami…
Chemical Engineering
ISBN:
9781259696527
Author:
J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:
McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind…
Chemical Engineering
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY

Elements of Chemical Reaction Engineering (5th Ed…
Chemical Engineering
ISBN:
9780133887518
Author:
H. Scott Fogler
Publisher:
Prentice Hall

Introduction to Chemical Engineering Thermodynami…
Chemical Engineering
ISBN:
9781259696527
Author:
J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:
McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind…
Chemical Engineering
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY

Elements of Chemical Reaction Engineering (5th Ed…
Chemical Engineering
ISBN:
9780133887518
Author:
H. Scott Fogler
Publisher:
Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:
9781285061238
Author:
Lokensgard, Erik
Publisher:
Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:
9780072848236
Author:
Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:
McGraw-Hill Companies, The