22. Read the medication label below and answer the following questions NDC 0703-0346-01 Rx only Ceftriaxone for Injection, USP 2 grams For I.M,or L.V. Use Single Use Vial Storage Prior to Reconstitution: Store powder at 20° to 25°C (68° to 77°F) For LM. Administration: Reconstitute with 4.2 mL [See USP Controlled Room Temperature). 1% Lidocaine Hydrochloride Injection (USP) or Protect from Light. Sterile Water for Injection (USP). Each 1 mL of solution contains approximately Omg equivalent of ceftriaxone. Storage After Reconstitution: See Package Insert 350 Mfd for: TEVA Each vial contains: ceftriaxone sodium powder equivalent to 2 grams ceftriaxone. For LV. Administration : Reconstitute with 19.2 ml of an IV. diluent specified in the accompanying package insert. Each 1 mL of solution contains approximately 100 mg equivalent of ceftriaxone. Withdraw entire contents and dilute to the desired concentration with the appropriate IV. diluent. Usual Dosage: See Package Insert Order: Ceftriaxone 875 mg IM STAT a. How much diluent is added to reconstitute the vial? b. What is the concentration of the medication? c. How many mLs should the nurse administer for per dose? d. Mark the syringe below with the appropriate dose. - - ม Teva Parenteral Medicines Irvine, CA 92618 Iss. 9/2009 3902302450909 35
22. Read the medication label below and answer the following questions NDC 0703-0346-01 Rx only Ceftriaxone for Injection, USP 2 grams For I.M,or L.V. Use Single Use Vial Storage Prior to Reconstitution: Store powder at 20° to 25°C (68° to 77°F) For LM. Administration: Reconstitute with 4.2 mL [See USP Controlled Room Temperature). 1% Lidocaine Hydrochloride Injection (USP) or Protect from Light. Sterile Water for Injection (USP). Each 1 mL of solution contains approximately Omg equivalent of ceftriaxone. Storage After Reconstitution: See Package Insert 350 Mfd for: TEVA Each vial contains: ceftriaxone sodium powder equivalent to 2 grams ceftriaxone. For LV. Administration : Reconstitute with 19.2 ml of an IV. diluent specified in the accompanying package insert. Each 1 mL of solution contains approximately 100 mg equivalent of ceftriaxone. Withdraw entire contents and dilute to the desired concentration with the appropriate IV. diluent. Usual Dosage: See Package Insert Order: Ceftriaxone 875 mg IM STAT a. How much diluent is added to reconstitute the vial? b. What is the concentration of the medication? c. How many mLs should the nurse administer for per dose? d. Mark the syringe below with the appropriate dose. - - ม Teva Parenteral Medicines Irvine, CA 92618 Iss. 9/2009 3902302450909 35
Essentials of Pharmacology for Health Professions
7th Edition
ISBN:9781305441620
Author:WOODROW
Publisher:WOODROW
Chapter8: Administration By The Gastrointestinal Route
Section: Chapter Questions
Problem 9CRQ
Related questions
Question
100%
Please help me answer!
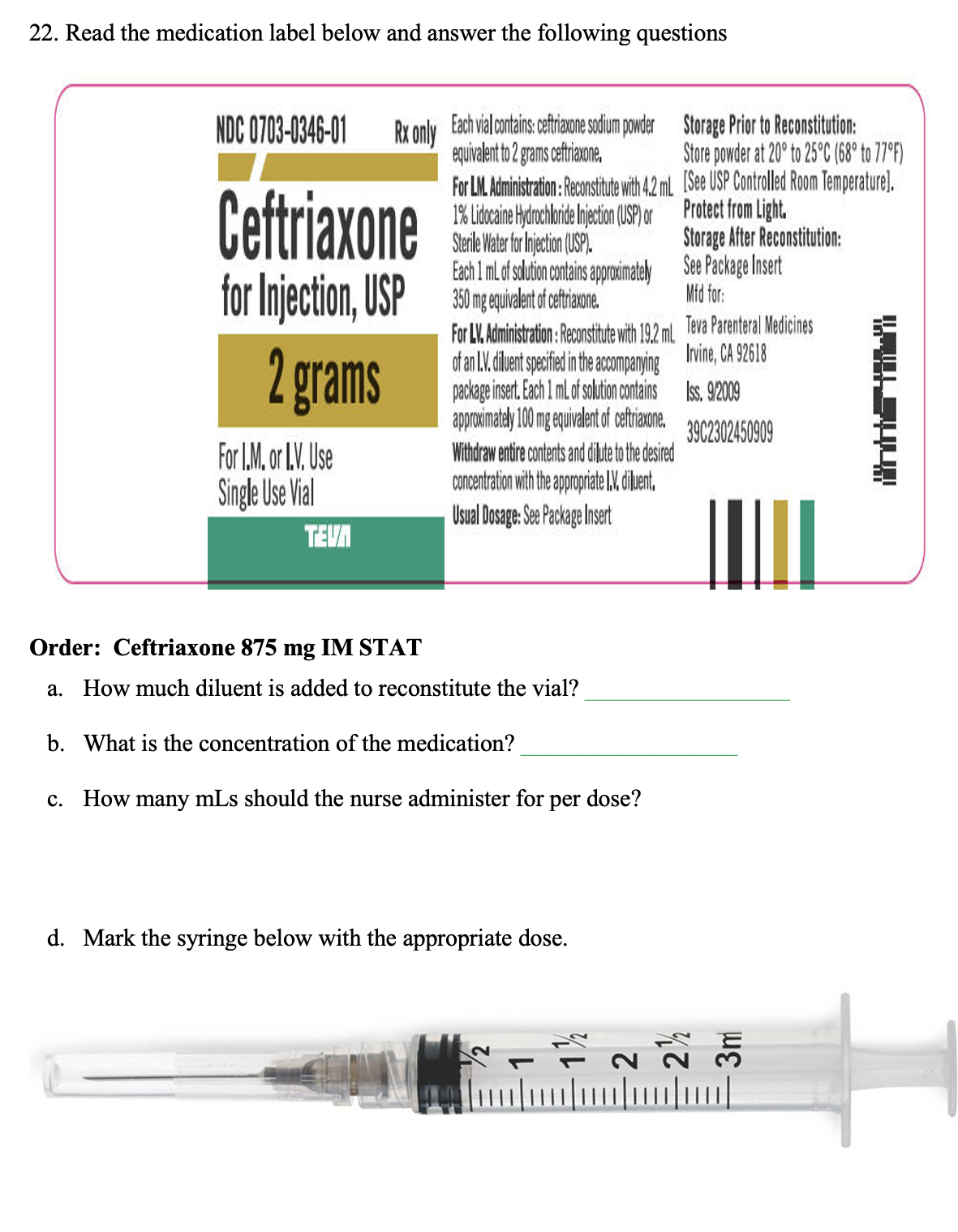
Transcribed Image Text:22. Read the medication label below and answer the following questions
NDC 0703-0346-01 Rx only Each vial contains: ceftriaxone sodium powder
equivalent to 2 grams ceftriaxone.
Ceftriaxone
for Injection, USP
2 grams
For I.M. or I.V. Use
Single Use Vial
TEVA
For LM. Administration: Reconstitute with 4.2 mL
1% Lidocaine Hydrochloride Injection (USP) or
Sterile Water for Injection (USP).
Each 1 mL of solution contains approximately
350 mg equivalent of ceftriaxone.
For LV. Administration: Reconstitute with 19.2 ml.
of an L.V. diluent specified in the accompanying
package insert. Each 1 mL of solution contains
approximately 100 mg equivalent of ceftriaxone.
Withdraw entire contents and dilute to the desired
concentration with the appropriate I.V. diluent.
Usual Dosage: See Package Insert
Order: Ceftriaxone 875 mg IM STAT
a. How much diluent is added to reconstitute the vial?
b. What is the concentration of the medication?
c. How many mLs should the nurse administer for
per dose?
d. Mark the syringe below with the appropriate dose.
Storage Prior to Reconstitution:
Store powder at 20° to 25°C (68° to 77°F)
[See USP Controlled Room Temperature).
Protect from Light.
Storage After Reconstitution:
See Package Insert
Mfd for:
Teva Parenteral Medicines
Irvine, CA 92618
Iss. 9/2009
3902302450909
S
[////
KOSUV
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Essentials of Pharmacology for Health Professions
Nursing
ISBN:
9781305441620
Author:
WOODROW
Publisher:
Cengage



Essentials of Pharmacology for Health Professions
Nursing
ISBN:
9781305441620
Author:
WOODROW
Publisher:
Cengage



Principles Of Radiographic Imaging: An Art And A …
Health & Nutrition
ISBN:
9781337711067
Author:
Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:
Cengage Learning