5. Turn your slope into a "for every" statement. Hint: The goes up for every 1 of ." BIU EEE E 6. Write out the equation for your line of best fit. Hint: Make sure you have: • Variables that match your experiment • Values for the the slope and intercept • Units for the slope and intercept
5. Turn your slope into a "for every" statement. Hint: The goes up for every 1 of ." BIU EEE E 6. Write out the equation for your line of best fit. Hint: Make sure you have: • Variables that match your experiment • Values for the the slope and intercept • Units for the slope and intercept
Related questions
Question
Pendulum experiment
Please help with #5 and 6
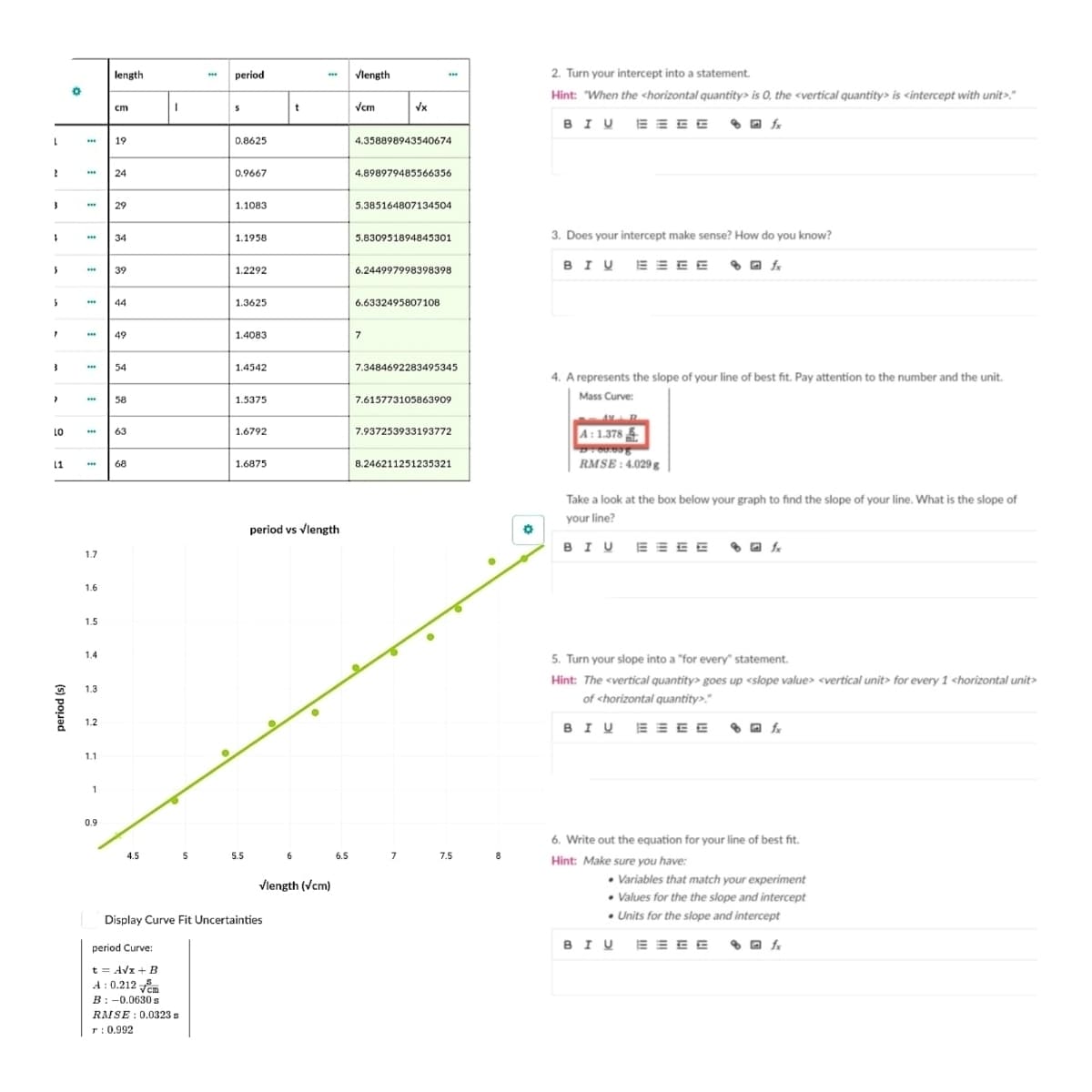
Transcribed Image Text:length
period
Vlength
2. Turn your intercept into a statement.
Hint: "When the <horizontal quantity> is 0, the <vertical quantity> is <intercept with unit>."
cm
t
Vcm
Vx
BIU EE EE O O
1.
19
0.8625
4.358898943540674
24
0.9667
4.898979485566356
29
1.1083
5.385164807134504
34
1.1958
5.830951894845301
3. Does your intercept make sense? How do you know?
BIU E=E E
39
1.2292
6.244997998398398
44
1.3625
6.6332495807108
49
1.4083
7
54
1.4542
7.3484692283495345
4. A represents the slope of your line of best fit. Pay attention to the number and the unit.
Mass Curve:
58
1.5375
7.615773105863909
7.937253933193772
A: 1.378
LO
63
1.6792
11
68
1.6875
8.246211251235321
RMSE : 4.029 g
Take a look at the box below your graph to find the slope of your line. What is the slope of
your line?
period vs Vlength
BIU E= EE
1.7
1.6
1.5
1.4
5. Turn your slope into a "for every" statement.
Hint: The <vertical quantity> goes up <slope value> <vertical unit> for every 1 <horizontal unit>
1.3
of <horizontal quantity>."
BIU E= EE
1.2
1.1
1
0.9
6. Write out the equation for your line of best fit.
Hint: Make sure you have:
4.5
5
5.5
6
6.5
7
7.5
8
• Variables that match your experiment
• Values for the the slope and intercept
• Units for the slope and intercept
Vlength (Vcm)
Display Curve Fit Uncertainties
BIU EE E E
period Curve:
t = Avx + B
A : 0.212
B:-0.0630 s
RMSE : 0.0323 s
r: 0.992
(S) pouad
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps
