B. A Carnot heat engine, shown in Fig. below, receives 400 kJ of heat per cycle from a high-temperature source at 552°C and rejects heat to a low-temperature sink at 25°C. Determine (a) the thermal efficiency of this Carnot engine and (b) the amount of heat rejected to the sink per High tmetne rervoir Carnot
B. A Carnot heat engine, shown in Fig. below, receives 400 kJ of heat per cycle from a high-temperature source at 552°C and rejects heat to a low-temperature sink at 25°C. Determine (a) the thermal efficiency of this Carnot engine and (b) the amount of heat rejected to the sink per High tmetne rervoir Carnot
Refrigeration and Air Conditioning Technology (MindTap Course List)
8th Edition
ISBN:9781305578296
Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill Johnson
Publisher:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill Johnson
Chapter45: Domestic Refrigerators And Freezers
Section: Chapter Questions
Problem 12RQ: Refrigerators currently being manufactured in the United States are using______as their refrigerant.
Related questions
Question
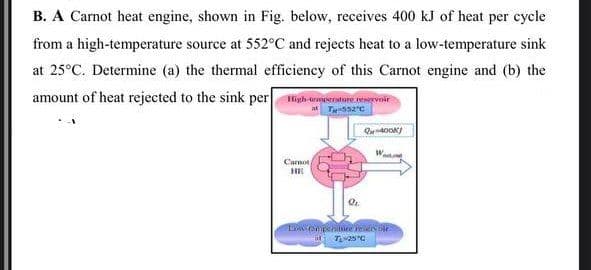
Transcribed Image Text:B. A Carnot heat engine, shown in Fig. below, receives 400 kJ of heat per cycle
from a high-temperature source at 552°C and rejects heat to a low-temperature sink
at 25°C. Determine (a) the thermal efficiency of this Carnot engine and (b) the
amount of heat rejected to the sink per igh urmesroture wrvoir
Cansot
HE
L Cmpeire er oe
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Refrigeration and Air Conditioning Technology (Mi…
Mechanical Engineering
ISBN:
9781305578296
Author:
John Tomczyk, Eugene Silberstein, Bill Whitman, Bill Johnson
Publisher:
Cengage Learning

Refrigeration and Air Conditioning Technology (Mi…
Mechanical Engineering
ISBN:
9781305578296
Author:
John Tomczyk, Eugene Silberstein, Bill Whitman, Bill Johnson
Publisher:
Cengage Learning