In standardization, 10.00 mL of Naz C2 04 solution required 39.17 mL of KMN04. Find the molarity of KMNO,. The unknown required 14.44 mL of MnO,. Find Ca in the urine. (Answer: 1.086 x 10 M, 7.840 x 10 M)
Q: B/ design circular tank to treat 5000m3/day of water with particles of 0.003cm, specific density…
A:
Q: A tank has a pure water flowing into it at 12 L/min. The content of the tank are kept thoroughly…
A:
Q: A municipal waste water has a BOD5 of 200 mg/L at 20 ˚C. The reaction rate constant k at 20 ˚C is…
A: Given data: Amount of BOD5 at 20°C (yt) = 200 mg/L Time (t)= 5 days (because, BOD5) Constant (k20)…
Q: Circular clarifiers are used to remove solid materials from water. They allow continuous treatment…
A:
Q: A sample is diluted to perform test 295 ml distilled water are added to 5mL sample to fill 300 ML…
A:
Q: Q/ A wastewater treatment use an activated sludge process for secondary treatment of 10000 m/day for…
A: Inflow Rate (Q) = 10000 m3/dayInitial BOD (S0) = 150 mg/L =150×10-610-3 kgm3 = 0.15 kg/m3Soluble…
Q: Effluent discharges from two secondary clarifiers were sampled. The secondary clarifier “A” is…
A:
Q: Available chlorine in bleaching powder solution is 0.5mg/ml. To obtain a chlorine dose of 0.1 mg/1…
A:
Q: Schlumberger Gen-6 140,000 (former Gen-9) General Resistivity of NaCI Water Solutions Gen grains/gal…
A:
Q: 100-mL water sample is mixed with 200 mL of deionized water for BOD analysis. The DO of the mixture…
A:
Q: A groundwater treatment plant adds chlorine to oxidize iron and manganese for removal by filtration.…
A: Given pH at 10oC = 7.8 t10 time through filtration = 1.8 min t10 time during peak pumpage = 8 min…
Q: A water contains a suspended solids concentration of 10.30 mg · L−1. Coagulation reduces this to 1.0…
A:
Q: A WASTEWATER TREATMENT PLANT OPERATING PRIMARY SETTLING TANKS GETS IN INFLOW OF 12.96 MILLION LITRES…
A: Answer We are given Flow rate Q = 12.96 MLD Influent suspended solid concentration Cin = 280 mg/l…
Q: Determine fixed and volatile solids concentration using the data given in example I and as under:…
A:
Q: 4 2.LacticacidhasaKa of1.38x10 . What is the pH of the solution that results when 25.0 mL of 0.300…
A: Since 25.0 mL of 0.300 M lactic acid are titrated with 20.0 mL of 0.375 M KOH solution. ⇒0.375 M KOH…
Q: The Volume of NaOH (0.01N) when use ph.ph indicator is 12.4mL and 3mL when use methyl Orange (M.O)…
A: Given data: Normality of NaOH = 0.01 N Volume of methyl orange (V1) = 3 mL Volume of phenolphthalein…
Q: Exercise-2 A surface water treatment plant is to process 30000 m'/d for a large city. A cubic rapid…
A: Given: Water Demand= 30000 m3/day Dose of Alum= 35 mg/L Detention time= 2 minutes Viscosity =…
Q: The following data were reported on the operation of primary settling tank and activated sludge…
A:
Q: .Determine the 5-day BOD (in mg/L) for a 15 ml sample that is diluted with dilution water to a total…
A: Given a 15 ml sample that is diluted with dilution water to a total volume of 300 ml when the…
Q: 1. If concentration of Na+, Cat+ and Mg++ in a water sample are 345, 60 and 18 mg/I respectively,…
A:
Q: A/Determine the dimensions of settling tank to treat 1000m/day of water with particles of 0.02mm and…
A:
Q: Example. Following observations were made on 4% dilution of waste water: no. of aerated water used…
A: Given data: Dilution percent=4% DO of aerated water=4 mg/l DO of diluted sample after 5 days…
Q: A Granulated Activated Carbon (GAC) adsorption laboratory column 10cm in diameter and 3m deep, is…
A: Given Area=7.854×10−3m3 Volume= 6.3660m/hr t=0.47123 Q=2.122 bed volume /hr
Q: Q.68 An activated sludge tank is 30 m long 9 m wide and has liquid depth of 4 m. The influent sewage…
A: Given data- Dimensions of the sludge tank=30m×9m×4mInfluent sewage flow Qo=3.5 MLDBOD So=130mg/LMLSS…
Q: A water sample was tested for water hardness. A 50.00 mL sample, prepared and buffered to pH 10,…
A: The given data is shown below:
Q: Determine the diameter of a trickling filter for a wastewater flowing at the rate of (13500/100)…
A: Wastewater flowing at the rate of (Q = 13500/100) gallons per minute. Concentration for the…
Q: For the measurement of the BOD of a wastewater sample from textile industry, five different…
A: Given- Assume K=-0.234day-1 t=5
Q: Suppose the estimated BOD of an influent sample is expected to be 150 mg/L and the dissolved oxygen…
A: Calculating BOD of raw sewage: Because we have given a condition:
Q: A wastewater sample was diluted 10% with tap water for conducting BOD Test. The DO content in…
A: Given that, Dilution percentage= 10% Initial D.O of waste water (D.oi)= 1.2mg/l Final D.O. of…
Q: The BOD, of a municipal wastewater is 200 mg/L at 20°C. The amount of organic matter remaining in…
A:
Q: () A wastewater sample is being analyzed to determine its BOD content. The sample is diluted in…
A: Volume of wastewater sample=5 mL Volume of distilled water added to sample for dilution=295 mL Total…
Q: 1. A groundwater contains the following constituents: CO2 = 9.6 mg/L Ca2+ = 95.2 mg/L Mg2+ = 25.8…
A: The given data is: Constituents of ground water CO2 = 9.6 mg/l Mg2+ = 25.8 mg/l Ca2+ = 95.2 mg/l…
Q: 1. Calculate the volume and weight of sludge generated per day in circular primary settling tam…
A: The given data is: Flow=10000 m3/d The inlet concentration=1100 mg/l The total suspended…
Q: MPN of effluent is 106/100 ml and it reduces to 102/100 ml after chlorination. Find percentage…
A: Given: Initial MPN value = 106/100 ml Final MPN value = 102/100 ml
Q: t more than 7 mg/L of soluble BOD in the effluent. The mixed liquor has a concentration of 3000 mg/L…
A:
Q: A 100-gal tank initially contains 25 gal of 50% salt solution. Fresh water is then poured into the…
A:
Q: Effluent discharges from two secondary clarifiers were sampled. The secondary clarifier “A” is…
A:
Q: OD of a wastewater sample is determined by incubating the BOD bottles at four different…
A: Ultimate BOD is independent of temperature.
Q: Water quality and characteristics discuss: 1. Organoleptic parameters 2. Nitrate 3. Solids
A: 1) Organoleptic Parameters- These are defined as the parameters in which results are obtained after…
Q: Water analysis was carried out for solid analysis. The test was performed and the following…
A: In question value of (mass of dish + ignited solid) is less than the value of (mass of empty dish)…
Q: A BOD analysis was conducted on a partially- treated sewage at 25°C. Only 20% of the volume of BOD…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: The waste water is flowing at a rate of 0.2 m³/s in a settling tank. The specific gravity of…
A: Given that: Flow rate of wastewater, Q=0.2m3/sSpecific Gravity of particles,…
Q: The following dissolved-oxygen values were found after 5days of incubation in 310-mL BOD bottles:…
A: Biochemical Oxygen Demand or BOD is defined as the oxygen required to oxidize the biodegradable…
Q: a. A stream has an average stream velocity of 0.92 [m/s] and an average depth of 1 [m]. Calculate…
A:
Q: 5 An air stripping tower is to be designed to lower the ethylbenzene (C,H0) concentration in a…
A:
Q: Wet coal is dried in a continuous rotary dryer that operates at atmospheric pressure. The chips with…
A: Molecular weight: Molecular weight is the weight of the given substance or molecule. The molecular…
Q: In determination of Acidity experiment, If the volume of 0.02N NaOH when use Methyl Orange (M.O) is…
A: Given data: Normality of NaOH = 0.02 N Volume of methyl orange (V1) = 0.8 mL Volume of…
Q: The mixture has a temp. = 20 °C %3D Deoxygenation const.= 0.28 day1 %3D Average flow = 0.3 m/s,…
A: Lo = BODu = 25.8mg/L DOmix = 7.2mg/L T (Temperature) = 20o C KD = 0.28/day Vav = 0.3m/s y(average…
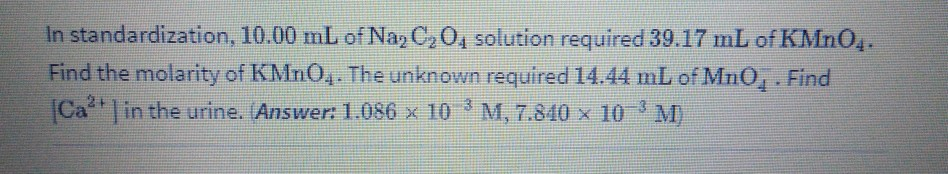
Trending now
This is a popular solution!
Step by step
Solved in 8 steps

- 3. Circular clarifiers are used to remove solid materials from water. They allow continuous treatment of water and can handle fairly large flows. Solids settle to the bottom of the clarifier and are carried away by a small stream of water (the waste stream). Most of the water exits the tank as treated effluent. Assume a clarifier receives a flow of 0.10 m3/s with a solid concentration of 800 mg/L. If the waste stream consists of 3% of the incoming flow and carries 90% of the incoming solids, a) what is the solids concentration in the waste stream, and b) what is the solids concentration in the treated effluent?For the measurement of the BOD of a wastewater sample from textile industry, five different dilutions (0.5%, 1%, 2%, 5% and 10%) of 1 L are prepared, with the addition of 2 mL of seed. Three different dilutions (0,5%, 2% and 5%) are used for seed corrections. The results of DO measurements are given below. Calculate the BOD5 concentration of the sample.-4 2.LacticacidhasaKa of1.38x10 . What is the pH of the solution that results when 25.0 mL of 0.300 M lactic acid are titrated with 20.0 mL of 0.375 M KOH?
- A tank is initially filled to capacity with 300 L pure water. At t=0, brine containing 2 g/L salt is pumped into the tank at a rate of 25 L/min. The well-mixed solution is pumped out at a larger rate of 50 L/min. Determine the concentration of the salt inside the tank after 5 min. Use dQ/dt = ci vi - co vo as a formulaA tank has a total capacity of 100L, but initially it contains 15L of pure water. Water flows into the tank 8L per min and contains 2kg of salt. The solution in the tank is being mixed well. If the well mixed solution in the tank exits 7L per minute, determine the amount of salt in the solution just before the tank overflowsThe results of a jar test on a surface water supply sample revealed that the optimum dose of alum (Al2(SO4)3.14H2O) as coagulant is 17 mg/L. Furthermore, the water has total alkalinity = 50 mg/L as CaCO3; pH = 7.8; and residual turbidity at optimum alum dose = 4 NTU. The water treatment plant has a mean flow of 10m3/hour. Determine: The kg of alum per year needed in the operation of the plant. The residual alkalinity in the sample, following the addition of alum at optimum dose. Solve parts (a) and (b) if instead of alum, ferric chloride (FeCl3) were used as coagulant. Assume the numerical jar test results are the same as those with alum.
- The BODs of a domestic wastewater sample is considered to be about 200 mg/l and the DO in it is measured as 0 mg/L. The laboratory staff are preparing to set up a BOD test to determine the actual BOD of the sample. The DO of the dilution water was measured to be 9 mg/I. Which of the following volume ratios of (total volume of BOD bottle = 300 mL) / (volume of wastewater sample) would be most appropriate to use in setting up the BOD bottle for incubation? Select one among the following: (a) 20/1; (b) 50/1; (c) 100/1; (d) 500/1. Justify your selectionGiven:Flow rate of water = 500 L/minSuspended solids concentration in influent water = 100 mg/LRequired effluent suspended solids concentration = 10 mg/LFiltration rate = 10 m^3/m^2/hrFiltration tank dimensions: Length = 5 m, Width = 3 m, Depth = 4 mCalculate:o Calculate the total suspended solids (TSS) load in the influent water:o Calculate the filtration tank surface area:o Calculate the required filtration tank capacity:o Calculate the required detention time in the filtration tank: Q2) DisinfectionGiven:Flow rate of water = 2000 m^3/hChlorine residual required in effluent = 0.5 mg/LInitial chlorine concentration in influent water = 2 mg/LChlorine decay rate in disinfection tank = 0.05 per hourDisinfection tank volume = 300 m^3Calculate:o Calculate the chlorine demand in the influent water:o Calculate the chlorine dosage required:o Calculate the chlorine dosage rate:o Calculate the actual chlorine dosage rate considering chlorine decay:A BOD analysis was performed on a municipal wastewater. The 300 mL test bottle was filled with 2.0% wastewater mixed with dillution water, The initial dissolved oxygen (DO) was 8.3 mg/L. The sample is then put in a incubator and set at 20°C. Since the 5th day falls on public holiday, the final DO does not get measured until the 7th day. The final DO is measured at 4.5 mg/L. Assuming k30 (base e) is 0.2/day, determine the BOD3 @ 20°C BOD5 @ 30°C
- Suppose the estimated BOD of an influent sample is expected to be 150 mg/L and the dissolved oxygen concentration of the oxygen saturated dilution water used in the BOD test is 8.5 mg/L. If you are using a 300 mL BOD bottle, estimate the maximum and minimum amount of sample you would add to the BOD bottle to ensure satisfactory test results. NOTE: Acceptable BOD test should comply with the two essential criteria: DO final ≥ 2 mg/L and DO initial – DO final ≥ 2 mg/L. Assume that the dilution water is the only source of oxygen.A 5-day BOD test if performed on an industrial wastewater sample that contains no bacteria; therefore, a "seeded" BOD test is run. Ten ml of "seed" are added to 20 liters of dilution water. Thirty milliliters of industrial wastewater are added to a 300-ml BOD bottle, and the remaining volume consists of "seeded" dilution water. The average dissolved-oxygen concentrations of the diluted wastewater samples and blanks (seeded dilution water) on the first day of the test were 7.5 mg/L and 9.0 mg/L, respectively. After incubation of separate BOD bottles at 20 C for 5 days, the average DO concentrations of the diluted wastewater BOD bottles and seeded dilution water BOD bottles were 3.1 and 8.5 mg/L, respectively. Calculate the 5-day BOD of the industrial wastewater. I am unsure of how to solve for f given this information, where f = (% seed in D1) / (% seed in B1)A 100-mL water sample is mixed with 200 mL of deionized water for BOD analysis. The DO of the mixture is 8.2 mg/L. The DO after 5 days is 4.9 mg/L. What is the BOD of the water sample?

