Please check all answer(s) that apply Which of the following words describes "making a bond Exothermic Catabolic Dehydration Anabolic Hydrolysis Condensation | Endothermic Exergonic D Endergonic
Please check all answer(s) that apply Which of the following words describes "making a bond Exothermic Catabolic Dehydration Anabolic Hydrolysis Condensation | Endothermic Exergonic D Endergonic
Biology (MindTap Course List)
11th Edition
ISBN:9781337392938
Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Chapter3: The Chemistry Of Life: Organic Compounds
Section: Chapter Questions
Problem 14TYU: Hydrogen bonds and van der Waals interactions are much weaker than covalent bonds, yet they are...
Related questions
Question
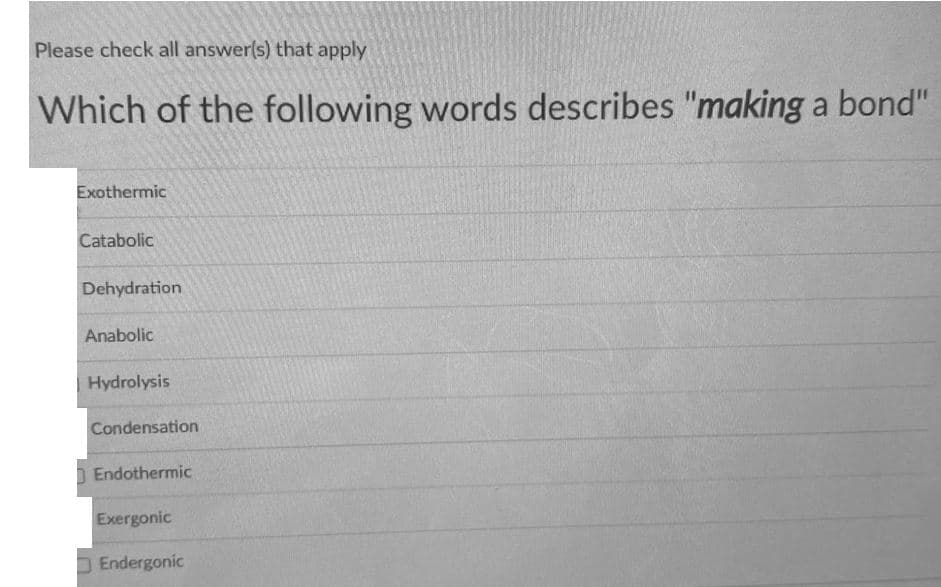
Transcribed Image Text:Please check all answer(s) that apply
Which of the following words describes "making a bond"
Exothermic
Catabolic
Dehydration
Anabolic
Hydrolysis
Condensation
| Endothermic
Exergonic
D Endergonic
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning