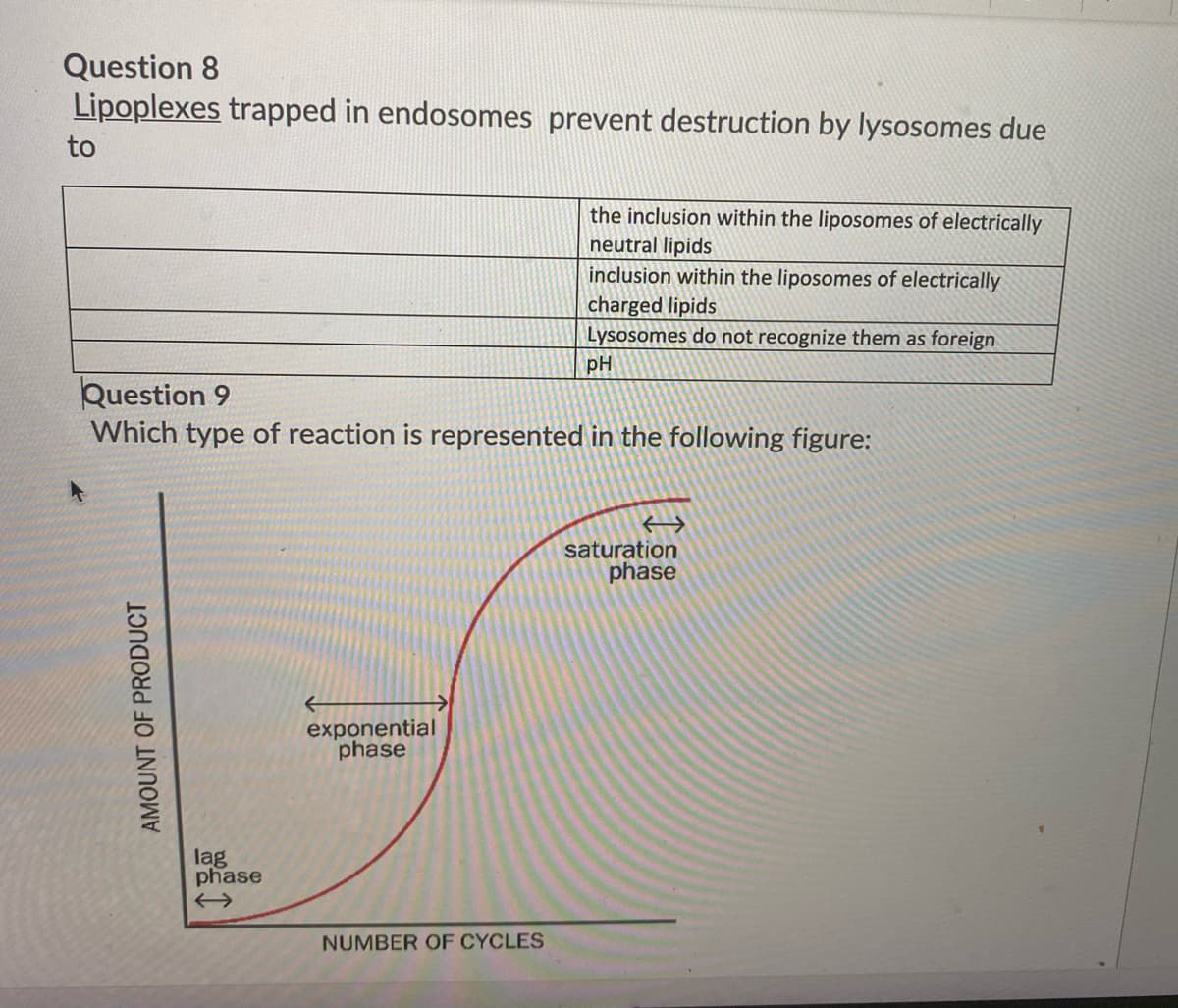
Question 8 Lipoplexes trapped in endosomes prevent destruction by lysosomes due to the inclusion within the liposomes of electrically neutral lipids inclusion within the liposomes of electrically charged lipids Lysosomes do not recognize them as foreign pH Ouestion 8
Question 8 Lipoplexes trapped in endosomes prevent destruction by lysosomes due to the inclusion within the liposomes of electrically neutral lipids inclusion within the liposomes of electrically charged lipids Lysosomes do not recognize them as foreign pH Ouestion 8
Human Anatomy & Physiology (11th Edition)
11th Edition
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:Elaine N. Marieb, Katja N. Hoehn
Chapter1: The Human Body: An Orientation
Section: Chapter Questions
Problem 1RQ: The correct sequence of levels forming the structural hierarchy is A. (a) organ, organ system,...
Related questions
Question
Transcribed Image Text:Question 8
Lipoplexes trapped in endosomes prevent destruction by lysosomes due
to
the inclusion within the liposomes of electrically
neutral lipids
inclusion within the liposomes of electrically
charged lipids
Lysosomes do not recognize them as foreign
pH
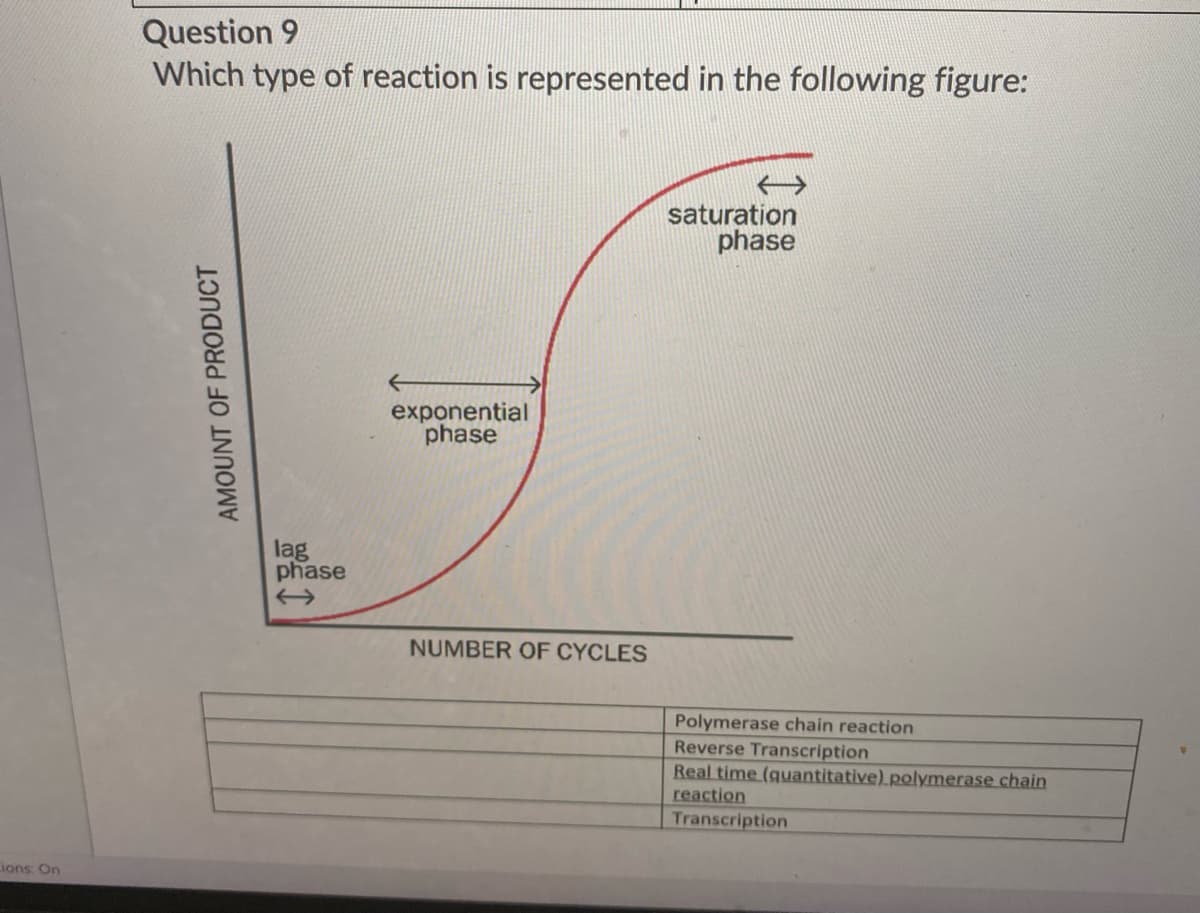
Question 9
Which type of reaction is represented in the following figure:
saturation
phase
exponential
phase
lag
phase
NUMBER OF CYCLES
AMOUNT OF PRODUCT
Transcribed Image Text:Question 9
Which type of reaction is represented in the following figure:
saturation
phase
exponential
phase
lag
phase
->
NUMBER OF CYCLES
Polymerase chain reaction
Reverse Transcription
Real time (quantitative) polymerase chain
reaction
Transcription
ions: On
AMOUNT OF PRODUCT
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Recommended textbooks for you

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:
9780134580999
Author:
Elaine N. Marieb, Katja N. Hoehn
Publisher:
PEARSON

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Anatomy & Physiology
Biology
ISBN:
9781259398629
Author:
McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:
Mcgraw Hill Education,

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:
9780134580999
Author:
Elaine N. Marieb, Katja N. Hoehn
Publisher:
PEARSON

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Anatomy & Physiology
Biology
ISBN:
9781259398629
Author:
McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:
Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:
9780815344322
Author:
Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:
W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:
9781260159363
Author:
Martin, Terry R., Prentice-craver, Cynthia
Publisher:
McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:
9781260231700
Author:
Sylvia S. Mader, Michael Windelspecht
Publisher:
McGraw Hill Education