TABLE 2.3 Lattice Structure of Metals Crystal Structure Atomic Radius (nm) Crystal Structure Atomic Metal (nm) Metal Radius (nm) Aluminum FCC 0.1413 Molybdenum BCC 0.1363 Cadmium НСР 0.1490 Nickel FCC 0.1246 Chromium BCC 0.1249 Platinum FCC 0.1387 Cobalt НСР 0.1253 Silver FCC 0.1445 Copper FCC 0.1278 Tantalum BCC 0.1430 Gold FCC 0.1442 Titanium НСР 0.1445 Iron BCC 0.1241 Tungsten BCC 0.1371 Lead FCC 0.1750 Zinc НСР 0.1332
TABLE 2.3 Lattice Structure of Metals Crystal Structure Atomic Radius (nm) Crystal Structure Atomic Metal (nm) Metal Radius (nm) Aluminum FCC 0.1413 Molybdenum BCC 0.1363 Cadmium НСР 0.1490 Nickel FCC 0.1246 Chromium BCC 0.1249 Platinum FCC 0.1387 Cobalt НСР 0.1253 Silver FCC 0.1445 Copper FCC 0.1278 Tantalum BCC 0.1430 Gold FCC 0.1442 Titanium НСР 0.1445 Iron BCC 0.1241 Tungsten BCC 0.1371 Lead FCC 0.1750 Zinc НСР 0.1332
Solid Waste Engineering
3rd Edition
ISBN:9781305635203
Author:Worrell, William A.
Publisher:Worrell, William A.
Chapter4: Mechanical Processes
Section: Chapter Questions
Problem 4.20P
Related questions
Question
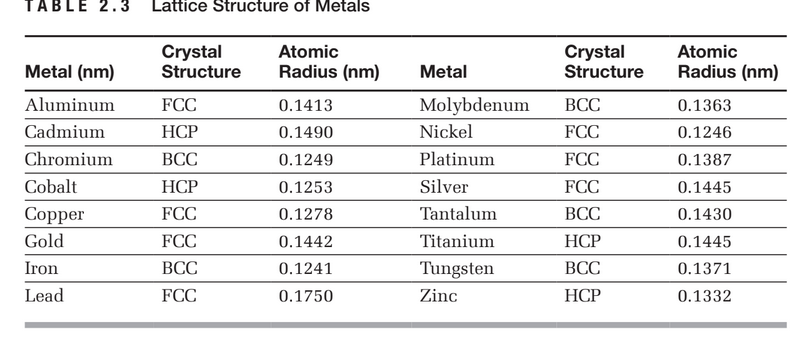
Using the information available in Table 2.3, calculate the density of molyb-denum given that it has an atomic mass of 95.94 g/mole.
Transcribed Image Text:TABLE 2.3
Lattice Structure of Metals
Crystal
Structure
Atomic
Radius (nm)
Crystal
Structure
Atomic
Metal (nm)
Metal
Radius (nm)
Aluminum
FCC
0.1413
Molybdenum
BCC
0.1363
Cadmium
НСР
0.1490
Nickel
FCC
0.1246
Chromium
BCC
0.1249
Platinum
FCC
0.1387
Cobalt
НСР
0.1253
Silver
FCC
0.1445
Copper
FCC
0.1278
Tantalum
BCC
0.1430
Gold
FCC
0.1442
Titanium
НСР
0.1445
Iron
BCC
0.1241
Tungsten
BCC
0.1371
Lead
FCC
0.1750
Zinc
НСР
0.1332
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, civil-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Solid Waste Engineering
Civil Engineering
ISBN:
9781305635203
Author:
Worrell, William A.
Publisher:
Cengage Learning,

Solid Waste Engineering
Civil Engineering
ISBN:
9781305635203
Author:
Worrell, William A.
Publisher:
Cengage Learning,