Table of BSA solution absorbance of concentration at 595nm. BSA Concentration (ug/ml) OD595nm Group 1 Group 2 Group 3 Grou 0.126 0.133 0.125 0.12 25 0.190 0.146 0.164 0.2- 50 0.298 0.209 0.185 0.25 75 0.291 0.224 0.242 0.3: 100 0.325 0.188 0.266 0.3- 150 0.222 0.181 0.268 0.3.
Table of BSA solution absorbance of concentration at 595nm. BSA Concentration (ug/ml) OD595nm Group 1 Group 2 Group 3 Grou 0.126 0.133 0.125 0.12 25 0.190 0.146 0.164 0.2- 50 0.298 0.209 0.185 0.25 75 0.291 0.224 0.242 0.3: 100 0.325 0.188 0.266 0.3- 150 0.222 0.181 0.268 0.3.
Chapter9: Parenteral Medication Labels And Dosage Calculation
Section: Chapter Questions
Problem 4.6P
Related questions
Question
write a short description based on the table
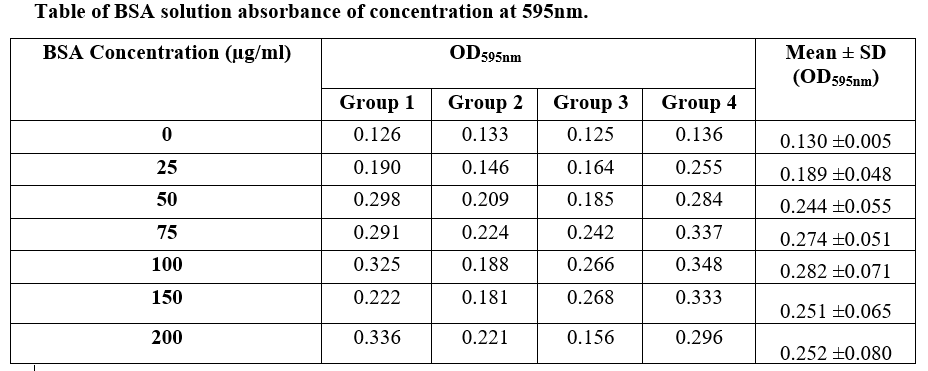
Transcribed Image Text:Table of BSA solution absorbance of concentration at 595nm.
BSA Concentration (ug/ml)
OD595nm
Mean + SD
(OD595nm)
Group 1
Group 2
Group 3
Group 4
0.126
0.133
0.125
0.136
0.130 ±0.005
25
0.190
0.146
0.164
0.255
0.189 ±0.048
50
0.298
0.209
0.185
0.284
0.244 ±0.055
75
0.291
0.224
0.242
0.337
0.274 +0.051
100
0.325
0.188
0.266
0.348
0.282 +0.071
150
0.222
0.181
0.268
0.333
0.251 +0.065
200
0.336
0.221
0.156
0.296
0.252 ±0.080
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Recommended textbooks for you







Essentials of Pharmacology for Health Professions
Nursing
ISBN:
9781305441620
Author:
WOODROW
Publisher:
Cengage