The results of a temperature jump test are shown below. At one hour, the temperature was suddenly increased from 950 K to 1000 K while holding the stress constant at 100 MPa. 30) Estimate the activation energy, Q, in the equation È = A o" ert R is the universal gas constant R = 8.31 J/(mole K) 0.004 0.0035 T= 1000 K 0.003 0.0025 0.002 0.0015 T= 950 K 0.001 0.0005 1000 2000 3000 4000 5000 6000 Time (s) Strain
The results of a temperature jump test are shown below. At one hour, the temperature was suddenly increased from 950 K to 1000 K while holding the stress constant at 100 MPa. 30) Estimate the activation energy, Q, in the equation È = A o" ert R is the universal gas constant R = 8.31 J/(mole K) 0.004 0.0035 T= 1000 K 0.003 0.0025 0.002 0.0015 T= 950 K 0.001 0.0005 1000 2000 3000 4000 5000 6000 Time (s) Strain
Principles of Heat Transfer (Activate Learning with these NEW titles from Engineering!)
8th Edition
ISBN:9781305387102
Author:Kreith, Frank; Manglik, Raj M.
Publisher:Kreith, Frank; Manglik, Raj M.
Chapter9: Heat Transfer With Phase Change
Section: Chapter Questions
Problem 9.28P
Related questions
Question
Need neat and clean handwritten solution with step wise explaination and plagiarism free. Thanks.
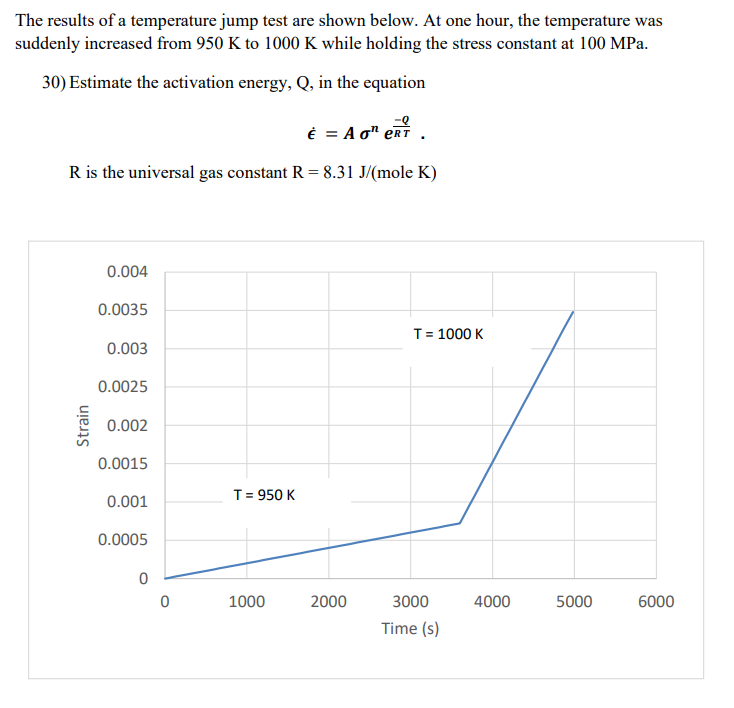
Transcribed Image Text:The results of a temperature jump test are shown below. At one hour, the temperature was
suddenly increased from 950 K to 1000 K while holding the stress constant at 100 MPa.
30) Estimate the activation energy, Q, in the equation
E = A o" erT
R is the universal gas constant R = 8.31 J/(mole K)
0.004
0.0035
T= 1000 K
0.003
0.0025
0.002
0.0015
T= 950 K
0.001
0.0005
1000
2000
3000
4000
5000
6000
Time (s)
Strain
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Heat Transfer (Activate Learning wi…
Mechanical Engineering
ISBN:
9781305387102
Author:
Kreith, Frank; Manglik, Raj M.
Publisher:
Cengage Learning

Principles of Heat Transfer (Activate Learning wi…
Mechanical Engineering
ISBN:
9781305387102
Author:
Kreith, Frank; Manglik, Raj M.
Publisher:
Cengage Learning