We have found the following equation for the amount of chemical C, X(t). 150 - X = ce180kt 120 - 2X To solve for the constants, we use the initial conditions. At time t = 0, no quantity of chemical C has been produced, or X(0) = 0. This can be used to solve for c. 150 - X ce180kt %3D 120 2X 150 – 0 ce180k(0) 120 C = We also know that 10 grams of C is formed after 9 minutes, or X(9) = 10, which can be used to solve for k. (Round your final answer to 7 decimal places.) 150 – 10 5180k(9) 120 – 2(10) = e1,620k 500 In = 1,620k 500 k =
We have found the following equation for the amount of chemical C, X(t). 150 - X = ce180kt 120 - 2X To solve for the constants, we use the initial conditions. At time t = 0, no quantity of chemical C has been produced, or X(0) = 0. This can be used to solve for c. 150 - X ce180kt %3D 120 2X 150 – 0 ce180k(0) 120 C = We also know that 10 grams of C is formed after 9 minutes, or X(9) = 10, which can be used to solve for k. (Round your final answer to 7 decimal places.) 150 – 10 5180k(9) 120 – 2(10) = e1,620k 500 In = 1,620k 500 k =
Algebra & Trigonometry with Analytic Geometry
13th Edition
ISBN:9781133382119
Author:Swokowski
Publisher:Swokowski
Chapter4: Polynomial And Rational Functions
Section: Chapter Questions
Problem 5T
Related questions
Question
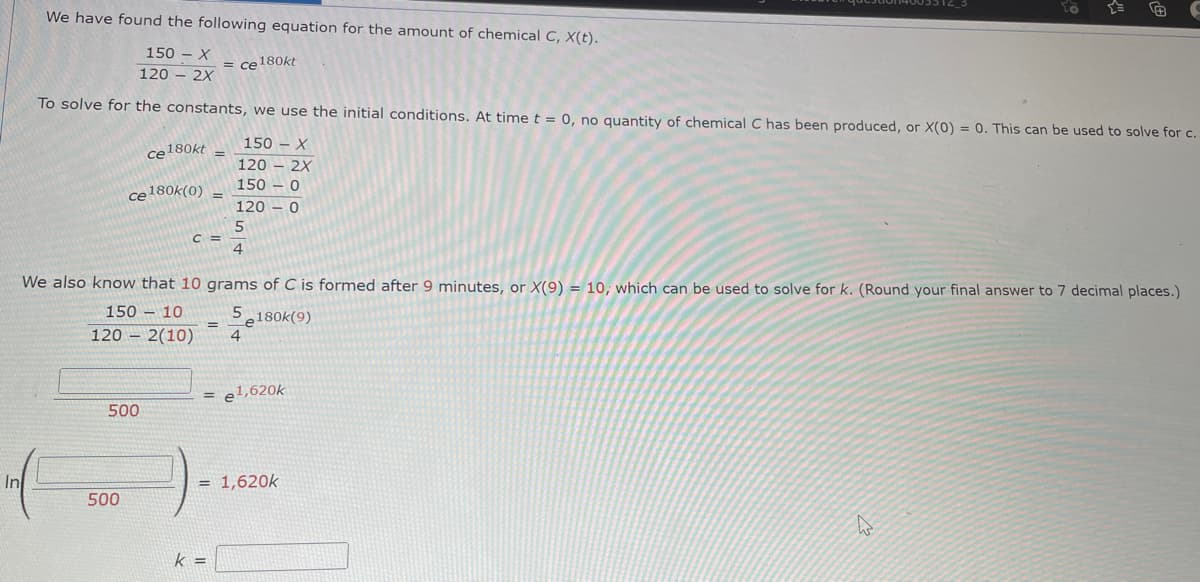
Transcribed Image Text:We have found the following equation for the amount of chemical C, X(t).
150 – X
= ce180kt
120 - 2X
To solve for the constants, we use the initial conditions. At time t = 0, no quantity of chemical C has been produced, or X(0) = 0. This can be used to solve for c.
150 - X
ce180kt
%3D
120
2X
150 – 0
ce180k(0)
120
C =
4
We also know that 10 grams of C is formed after 9 minutes, or X(9) = 10, which can be used to solve for k. (Round your final answer to 7 decimal places.)
150 – 10
5,180k(9)
120 – 2(10)
= e1,620k
500
In
= 1,620k
500
k =
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Algebra & Trigonometry with Analytic Geometry
Algebra
ISBN:
9781133382119
Author:
Swokowski
Publisher:
Cengage

Algebra & Trigonometry with Analytic Geometry
Algebra
ISBN:
9781133382119
Author:
Swokowski
Publisher:
Cengage