Which of the following groups in the following compound is important for the selectivity of ß-adrenoceptors? HO HO O The phenol OH O The amine O The alcohol IZ The tertiary butyl group The hydroxymethylene group
Which of the following groups in the following compound is important for the selectivity of ß-adrenoceptors? HO HO O The phenol OH O The amine O The alcohol IZ The tertiary butyl group The hydroxymethylene group
Essentials of Pharmacology for Health Professions
7th Edition
ISBN:9781305441620
Author:WOODROW
Publisher:WOODROW
Chapter22: Anticonvulsants, Antiparkinsonian Drugs, And Agents For Alzheimer’s Disease
Section: Chapter Questions
Problem 13CRQ
Related questions
Question
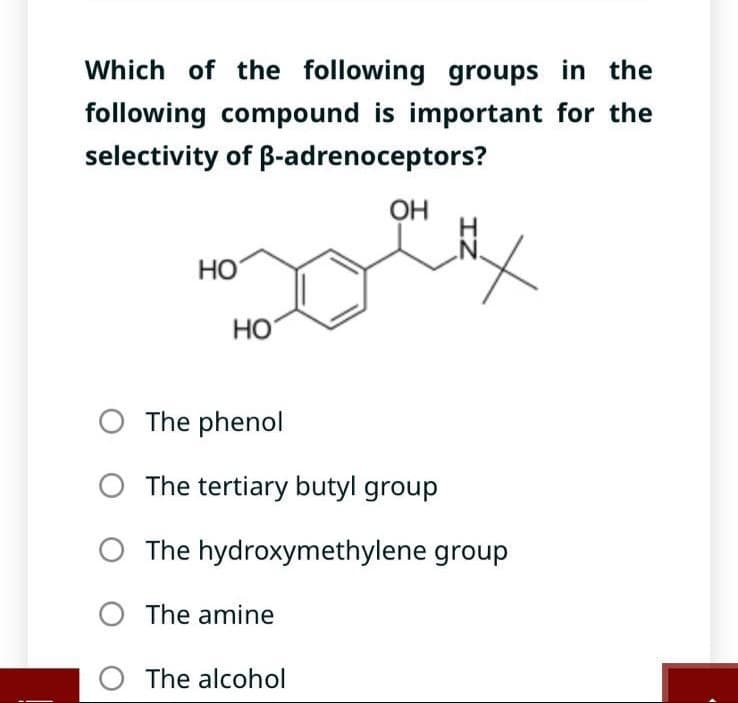
Transcribed Image Text:Which of the following groups in the
following compound is important for the
selectivity of B-adrenoceptors?
HO
HO
O The phenol
OH
O The amine
O The alcohol
IZ
The tertiary butyl group
The hydroxymethylene group
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Recommended textbooks for you

Essentials of Pharmacology for Health Professions
Nursing
ISBN:
9781305441620
Author:
WOODROW
Publisher:
Cengage


Essentials of Pharmacology for Health Professions
Nursing
ISBN:
9781305441620
Author:
WOODROW
Publisher:
Cengage
