Biology: The Unity and Diversity of Life (MindTap Course List)
15th Edition
ISBN:9781337408332
Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Chapter5: Ground Rules Of Metabolism
Section: Chapter Questions
Problem 11SQ: Solutes tend to diffuse from a region where they are _______ concentrated to an adjacent region...
Related questions
Question
Why does water enter a cell that is placed in a hypotonic solution?
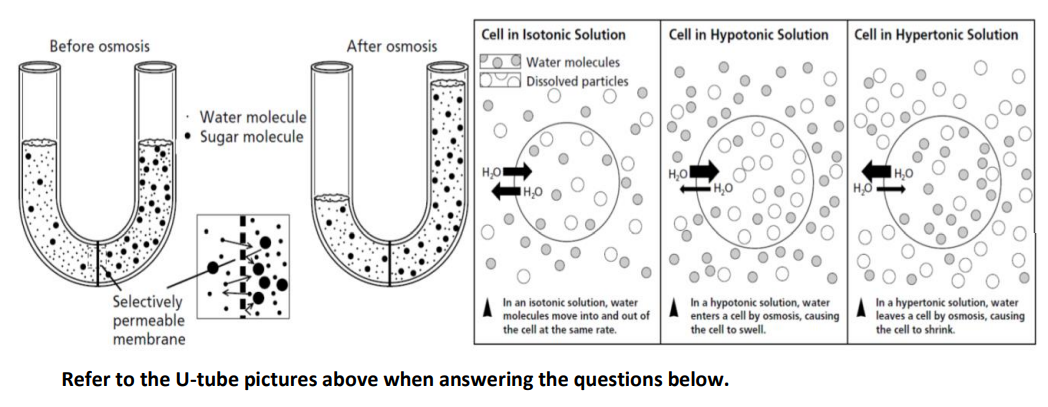
Transcribed Image Text:Cell in Isotonic Solution
Cell in Hypotonic Solution
Cell in Hypertonic Solution
Before osmosis
After osmosis
Pog Water molecules
DA Dissolved particles
Water molecule
• Sugar molecule
00
H,O
H,O
0,00
H,O
HO O
Selectively
permeable
membrane
In an isotonic solution, water
molecules move into and out of
the cell at the same rate.
In a hypotonic solution, water
enters a cell by osmosis, causing
the cell to swell.
In a hypertonic solution, water
leaves a cell by osmosis, causing
the cell to shrink.
Refer to the U-tube pictures above when answering the questions below.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781337408332
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781337408332
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning