.Assume the solubility product of Mg(OH)2 is 1.3 x 10 at a certain temperature. What minimum OH concentration must be attained (for example, by adding NaOH) to decrease the Mg2+ concentration in a М. solution of Mg(NO,), to less than 1.2 x 10-10 x 10 M (Enter your answer in scientific notation.)
.Assume the solubility product of Mg(OH)2 is 1.3 x 10 at a certain temperature. What minimum OH concentration must be attained (for example, by adding NaOH) to decrease the Mg2+ concentration in a М. solution of Mg(NO,), to less than 1.2 x 10-10 x 10 M (Enter your answer in scientific notation.)
Chapter16: Solubility And Complex Ion Equilibria
Section: Chapter Questions
Problem 100CP
Related questions
Question
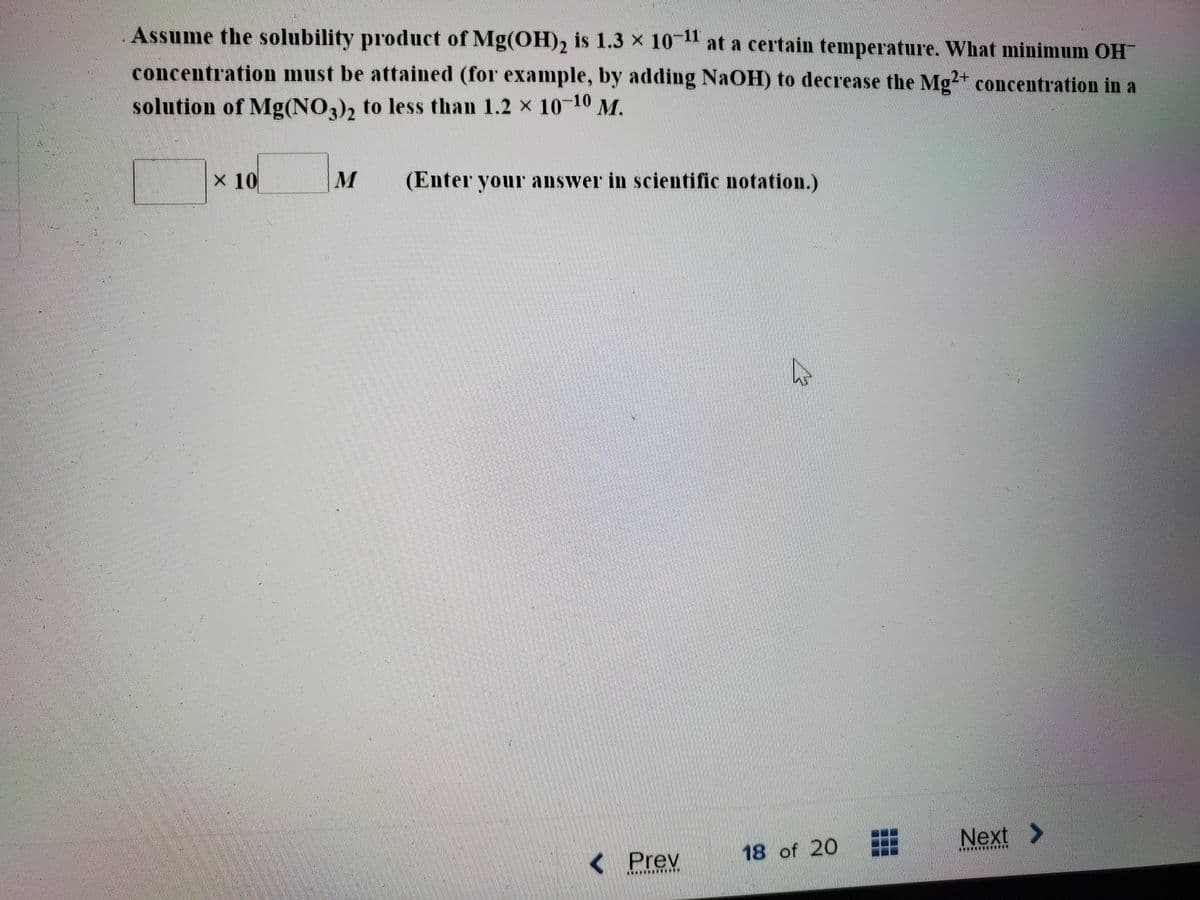
Transcribed Image Text:Assume the solubility product of Mg(OH)2 is 1.3 x 10 at a certain temperature. What minimum OH
concentration must be attained (for example, by adding NaOH) to decrease the Mg2+ concentration in a
solution of Mg(NO3), to less than 1.2 x 10-10 M.
3/2
x 10
M
(Enter your answer in scientific notation.)
Next >
< Prev
18 of 20
****
.*****
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax