Q: How can the Michaelis constant (Km) be identified and used to represent the affinity between the enz...
A: The correct answer is given below
Q: A 0.050 M monoprotic weak acid solution has a pH of 2.40. Calculate the pKa of the acid. Express you...
A:
Q: Given that H2X has pKa1 = 7.30 and pKa2 = 10.60, calculate the pH of 0.25 M NaHX. Express your answe...
A:
Q: Calculate the concentration of sulfide ions in a solution that is 0.050M in hydrogen sulfide and con...
A:
Q: R2. Ph H Br, Ph Ph H. R3. Br2, H2O
A:
Q: 1. Mother adds 5 ml of distilled water to 10.00 g of a powdered drug. The final volume of the soluti...
A:
Q: Describe the preparation of the following solutions. show all calculations. a.) 100mL of 0.5N NaOH ...
A:
Q: Acetylsalicylic acid (C9H8O4) is a monoprotic acid commonly known as "aspirin." A typical aspirin ta...
A: Find out moles of NaOH and then use mol to mol ratio to find moles of aspirin. Convert that moles in...
Q: Calculate the value of the EMF of galvanic cells and write the electrode reactions occurring in them...
A: a) Zn/Zn2+(0.001)∥Cd2+(0.1)/Cd Anode : Zn = Zn2+ + 2e- ...
Q: 2 - Explain the bonding, hybridization and geometry of carbon nanotubes that are used to help store ...
A: For storage of energy, the electronic properties of graphene is the mostly used. However the physic...
Q: What is the weaker acid, NH3 (ammonia) or [NH4]Cl (ammonium chloride)?
A: between NH3 (ammonia) and [NH4]Cl (ammonium chloride), ammonia is a base and ammonium chloride is i...
Q: Which of the following statements about the sensitivity of an analytical method is true? O The sensi...
A: Two questions based on tools in analytical chemistry that is to be accomplished.
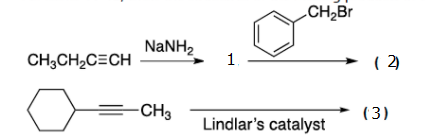
Q: Which of the following bases would you not use to deprotonate 1- pentyne? B H3C H3C CH2 lithium n-bu...
A: Terminal Alkyne has acidic hydrogen which can be removed by using strong base.
Q: Does Lucas Test show positive/negative reaction and color changes to: 1-butanol 2-propanol Tert-buty...
A: 1. Lucas Test : A positive test is indicated by a change from clear and colorless to turbid, signali...
Q: What atomic terms due to spin-orbit interactions are possible for following configurations? For each...
A:
Q: mixture of NaOH, NaCL and MgCl2 contains 34.6% by mass chloride and 20.0% by mass hydroxide. What ar...
A: From the hydroxide mass we can calculate the moles of Hydroxide and hence the moles of NaOH. And mas...
Q: A mixture of krypton and argon gas is compressed from a volume of 73.0 L to a volume of 30.0 L, whil...
A:
Q: The electrons in the outermost orbital determine the physical and chemical properties of the element...
A: Valence electrons are electrons in the outermost cell of an atom . For example oxygen has six v...
Q: aler completng the lab. Answer in the space provided. 1. A student reacted 0.500 g of Pb(NO1)2 with ...
A:
Q: Write the molecular, Total Ionic and Net Ionic equations Sulfuric acid and sodium hydroxide a. M: b...
A:
Q: Balance the following equations and write the corresponding ionic and net ionic equations (if approp...
A: Write the balanced molecular equation Split strong electrolytes to their corresponding ions to achi...
Q: CH;OH НО OH What is the name of this structure? OH OH alpha-D-gulopryranose beta-L-gulofuransone alp...
A:
Q: Calculate the concentration of sulfide ions in a solution that is 0.080M in hydrogen sulfide and con...
A: Here we have to determine concentration of sulfide ions in a solution that is 0.080M in H2S and cont...
Q: Calculate delta G° for the reaction below at 298 K. PCl3(g) + Cl2(g) -> PCl5(g...
A:
Q: step-s H3 O+ or anya Na oci codium hypochlocite acia protanated hypochlogous a cid step-2 (acia) HE ...
A: Answer: In this reaction we are preparing cyclohexanone by the hypochlorite oxidation of cyclohexano...
Q: A mixture of NaOH, NaCl, and MgCl2 contains 34.6% by mass chloride and 20.0% by mass hydroxide. What...
A:
Q: NaBr Ca(C2H3O2) , P:Os Ti(SO4)2 FEPO4
A: Ionic compounds have very high melting and Boiling points . The cation and anions held together by ...
Q: Find the molarity of Ba(OH)2 used by a laboratory analysts for standardization. It took 23.28 mL of ...
A:
Q: What is the element indicated by the residue formed in the charring test? 2. How do the conductivit...
A: Q.1 What is the element indicated by the residue formed in the charring test? Ans: Charring is a be...
Q: When dinitrogen pentoxide, N2O5, a white solid, is heated, it decomposes to nitrogen dioxide and oxy...
A:
Q: Differentiate the colligative properties of an electrolyte from a nonelectrolyte solution. Limit you...
A: Colligative properties: The colligative properties are those properties of solutions that depend on ...
Q: 40
A: Since you have posted multiple questions, the answer for first question is given below. Kindly repos...
Q: Why use plastic reagent bottle for the storage of the standard NaOH solution instead of typical glas...
A: NaOH is a base. It is hygroscopic in nature. Hygroscopic means tends to absorb moisture from air. I...
Q: 3. Which of the following is NOT considered to be a factor for effective collisions? Select one: ...
A: for effective collisions molecule should have molecules must be under go collision with proper orie...
Q: There are solutions of Ba(NO3)2 and A12(SO4)3 with the same molar concentration. Which of the soluti...
A:
Q: Iodination of Acetone (Experiment): https://youtu.be/KkpeiuelvTI 1. Why were the reagents equilibra...
A: 1. The reagents were equilibrated in water bath before mixing, to bring them all close to the same ...
Q: Provide a structure for the given compound. UV spectrum: Amax = 272 nm (e = 39500); El mass spectrum...
A:
Q: For each of the following precipitation reactions, calculate how many grams of the first reactant ar...
A:
Q: Calculate the pH of a HF/F- buffer solution after the addition of 32.65 mL of 0.100 M NaOH to 25.00 ...
A: A buffer solution is formed by the mixing of a weak acid and its conjugate base, or vice versa. The ...
Q: The Murchison meteorite that was found in Australia in 1969 is important to scientists because it co...
A: The Murchison shooting star is a shooting star that fell in Australia in 1969 close to Murchison, Vi...
Q: How many peaks will show up in the H NMR spectrum of the given compound below. Choose one: A. 9...
A: The proton-NMR spectra of a compound is used to determine the presence of different types of hydroge...
Q: Determine the molarity for each of the following solutions using the given data v 0.734 mol of CoClz...
A:
Q: (a) Many gasoous reactions occur in car engine and exhaust systems One of these is NOla)+ COlg)- NO ...
A: Since both questions are not interrelated to each other, we solve only first question according to t...
Q: Calculate the pH during the titration of 20.00 mL of 0.1000 M HCN(aq) with 0.2000 M RbOH(aq) after 7...
A: We have to find the pH of resulting solution.
Q: . Calculate the mass of hydrogen bromide once bromine combines with 24.3 L of hydrogen. a) Write and...
A: When hydrogen reacts with bromine ,it gives hydrogen bromide. At STP volume of 1 mol of gas is 22.4 ...
Q: Molar concentration of Fe(NO), Q.02m Molar concentration of NaSCN CN_Q.D01M Standard Solutions Blank...
A: Thus 1 mol of Fe(NO3)3 reacts with 1 mol of SCN- to form 1 mol of Fe(SCN)2+ Note: As per our company...
Q: Calculate the theoretical percentage of water in CoCl3·4H2O (Molar mass of the hydrate is 237.37 g/m...
A: Given :- chemical formula of hydrate = CoCl3.4H2O Molar mass of CoCl3.4H2O = 237.37 g/mol To calcu...
Q: Calculate the KHC8H4O4 titer of 0.125 M KOH. Give the chemical reactions involved.
A:
Q: In which direction will the following equilibrium shift when solid sodium chloride is added to an aq...
A: Answer: This question is based on the Le-chatalier's principle which suggests on changing any parame...
Q: Calculate the pH during the titration of 30.00 mL of 0.1000 M ammonia with 0.1000 M HCl(aq) after 12...
A:
Draw the product of the following compounds with the given image.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Draw the organic products formed when cyclopentene is treated withfollowing reagent. (CH3)3COOH, Ti[OCH(CH3)2]4, (−)-DETWould 7b show stereochemistry? It looks like it has 2 chiral centers.DHA is a fatty acid derived from sh oil and an abundant fatty acid in vertebrate brains. Hydrogenation of DHA forms docosanoic acid [CH3(CH2)20CO2H] and ozonolysis forms CH3CH2CHO, CH2(CHO)2 (ve equivalents), and HCOCH2CH2CO2H. What is the structure of DHA if all double bonds have the Z conguration?
- Which double bonds in the attached natural products can exhibitstereoisomerism? Nerolidol is isolated from the angel's trumpet plant,caryophyllene is present in hemp, and humulene comes from hops.Ignoring stereoisomers, draw the two possible enols for butan-2-one (CH3COCH2CH3), and predict which one is more stable.(a) Why does p-dichlorobenzene have a higher m.p. than its o- and m-isomers?(b) Why is (±)-Butan-2-ol optically inactive?
- Draw the organic products formed when cyclopentene is treated withfollowing reagent. [1] CH3CO3H; [2] H2O, HO−Draw the structure of the unbranched isomer of C5H11Br that is most reactive in an SN1 reaction.What are the reagents used to convert 2-butyne to C4H8? What are the products when that C4H8 is mixed with OsO4, and H2O (the formula will be C4H10O2 racemic)? What is the product when C4H8 is mixed with RCO3H, and H2O (the formula will be C4H10O2 meso)?
- 4-chloro-2-pentene has a double bond that can have either the E or The Z configuration and a sterogenic center that can have either the R or The S configuration. How many steroisomers are possible ? Draw the structure of each?Draw the products formed when both cis- and trans-but-2-ene are treated with OsO4, followed by hydrolysis with NaHSO3 + H2O. Explain how these reactions illustrate that syn dihydroxylation is stereospecic.Name the alkene below.Use only E/Z designators to indicate stereochemistry.

