Q: Draw a structural formula for the major organic product of the reaction shown below. ether…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: The correct product formed from the reaction below is 0 [0] No reaction OH OH OH
A: Oxidation is a process in which either the hydrogen atoms are eliminated or the oxygen atoms are…
Q: Draw the product of the following reaction sequence. CI 1. Mg(s), THF 2. CO₂(s) 3. H₂O+
A: In the above reaction 2-chloropronane is first treated with solid magnesium metal in the present of…
Q: What is the product of the following reaction? 1. LiAlH 2. H₂O OH HO OH OH IV
A:
Q: Question 4. Methylvinyl ketone (MVK) can undergo a polymerization reaction in the presence of AIBN…
A: The objective of the question is to understand the mechanism of the polymerization reaction of…
Q: A volume of 500.0 mL of 0.170 M NaOH is added to 625 mL of 0.200 M weak acid (Ka=2.88×10−5). What is…
A: pKa = - log Ka = - log (2.88 * 10-5) = 4.541
Q: 2. What dicarbonyl compounds forms the molecules below upon treatment with catalytic NaOCH¸ in HOCH¸…
A: Given reaction is an example of nucleophilic addition-elimination reaction of dicarbonyl…
Q: For each pair of compounds listed, check the box next to the one with the higher boiling point.…
A: Boiling point of hydrides increases as we move down the group because size increases as we move down…
Q: Write detailed reaction mechanism for synthesis of 1 - hexanol from 1 - hexene in presence of acid…
A: This is hydration reaction of alkene, Here addition of water molecule occurs following…
Q: Consider the following reactants: CI OH Would elimination take place at a significant rate between…
A: An elimination reaction is a type of organic reaction in which two substituents are removed from a…
Q: A local AM radio station broadcasts at a frequency of 621 kHz. (1 kHz= 1000 s¹) Calculate the energy…
A: Understanding the energy associated with electromagnetic radiation is fundamental in various fields,…
Q: Calculate how many grams of silver chloride will dissolve in 1.0 L of a 1.0 M NH3 solution through…
A: We will use stoichiometry of the reaction to get the answer.Balanced reaction:AgCl + 2NH3. -->…
Q: Which statement is false? The following reaction would help you to decide the answer: CO32- + HSO4…
A: -> In acid-base reaction there is formation of conjugate base and conjugate acid.-> When acid…
Q: Please don't provide handwriting solutions....
A: The objective of the question is to calculate the Gibbs free energy change (ΔG) for the given…
Q: A voltaic cell consists of a Zn/Zn2+ half-cell and a Ni/Ni2+ half-cell at 25 °C. The initial…
A: Given:Initial concentration of Ni+2 = 1.60 MInitial concentration of Zn+2 =0.110 M
Q: A chemistry graduate student is studying the rate of this reaction: - H2CO3(aq) H₂O (aq) + CO2 (aq)…
A: We are given a reaction, and given the concentration of reactant H2CO3 , let say it A at different…
Q: (b) H H CH2CH3 ·C. -CH2CH3 CH3 CH3OH (H)
A: The methanolysis of a tertiary alkyl halide involves the substitution of the halogen atom (usually a…
Q: Aqueous sulfuric acid (H2SO4) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium…
A:
Q: An antibacterial substance obtained from the bark of a flowering shrub (plumeria) has been…
A: Due to the high percentage of covalency in C-Cu bond in dialkylcuprates these compounds act as a…
Q: Calculate AG for the following reaction at 25 °C. C2H4(g) + 302(g) →>> 2CO2(g) + 2H2O (1) Round your…
A: The objective of this question is to calculate the Gibbs free energy change (ΔG) for the given…
Q: Provide the correct IUPAC name for the compound shown here. OH
A:
Q: A student determines the density of an object to be 5.9 g/mL. The actual density is 5.78 g/mL. The…
A: Answer:
Q: Given the standard enthalpy changes for the following two reactions: (1) 4C(s) + 5H2(g)-CH₁0(g) AH°…
A:
Q: hoose which of the following sets of quantum numbers is/are valid based on the quantum number rules.…
A: The objective of this question is to determine which sets of quantum numbers are valid based on the…
Q: O₂N. O2N ow. Ignore morgan byproducts. LL F OH
A: In the given reaction the presence of the electron-withdrawing group (nitro group) increases the…
Q: Identify product Z in the following reaction sequence: HOCH2-C=C-CH₂CH₂OH H₂ H₂SO4 CH3COOH X Y Z…
A:
Q: What Grignard reagent is needed to convert 2-pentanone O ethylmagnesium bromide Omethylmagnesium…
A: Grignard reagents add to carbonyl compounds to give primary, secondary, and tertiary alcohol
Q: a. Using the graph paper below, to plot the enzyme velocity versus the substrate concentration in…
A: Plotting the substrate concentration ([S]) on the x-axis and the enzyme velocity (V) on the y-axis…
Q: Please don't provide hnad writtin solution.....
A: The concentration of Ag+ that remains is approximately 2.69 x 10-22 M.Explanation:Given that:Volume…
Q: For the reaction below, the concentration of HI is 0.210 M and the concentrations of H2, and 12 are…
A: Given:Reaction is concentration of Hl = 0.210M concentration of H2= 0.564 Mconcentration of I2=…
Q: Draw the structures of two different alkyl bromides that could yield the alkene product shown as a…
A: The given structure of compound is a product of dehydrohalogenation of two different alkyl bromides…
Q: Nickel and carbon monoxide react to form nickel carbonyl, like this: Ni(s)+4CO(g)-Ni(CO)4(9) At a…
A:
Q: A 125.0-mL sample of a solution that is 3.0 × 10-3 M in AgNO3 is mixed with a 230.0-mL sample of a…
A: In the first step, we calculate the total volume of the solution obtained by mixing both the…
Q: Should a G-M detector be used to measure the exposure rate in the vicinity of a storage safe for…
A: A Geiger-Muller (G-M) detector is a common type of radiation detector that detects the presence and…
Q: c) Ph ONa 1 Equiv OH solvent
A: Given reaction is an example of nucleophilic addition reaction of carbonyl compounds.In this…
Q: Draw the skeletal (line-bond) structure of (S)-ethyl-2-methylheptanoate. Use a dash or wedge bond to…
A:
Q: 9. Provide a mechanism for the following reaction and rationalise the reactivity in terms of the 3…
A: The objective of the question is to provide a mechanism for the given reaction and explain the…
Q: How to determine the percentage of CaCO3
A:
Q: What is the IUPAC name? A
A: At first the longest carbon chain which contains more numbers of functional groups or…
Q: References The cation He'(g) has an excited electron that drops from it's excited energy to the…
A:
Q: Based on the activity series, which one of the reactions below will occur? Select one: O a. 3Hg (1)…
A: The objective of the question is to determine which of the given reactions will occur based on the…
Q: Nitrogen dioxide is one of the many oxides of nitrogen (often collectively called "NOx") that are of…
A: Given,Nitrogen dioxide reacts itself to form dinitrogen tetroxide.Initial,moles of NO2 = 36.…
Q: Choose the major product for the following reaction. NHEL NH₂ H₂O+ O a a b d NHET NH 2 NE NEt
A:
Q: 7. (8 pts) Calculate the solubility product, Ksp, for SrF2, given that its solubility is 0.0080 g…
A: The answer is-Explanation:Step 1: Step 2: Step 3: Step 4:
Q: Identify the functional groups present in the following molec
A: We are given an organic molecule, we have to tell about the functional groups.
Q: The standard enthalpy change for the following reaction is -722 kJ at 298 K. 2 Na(s) + Br2(l) — 2…
A: The standard enthalpy of formation occurs when 1 mole of a substance is formed from its elements in…
Q: Please don't provide handwritten solution ...
A: ✓ TrueExplanation:Step 1: Step 2: Step 3: Step 4:
Q: Consider the equilibrium reaction. H2(g) +12(9) 2 HI(9) In this case, 1.000 M H₂ reacts with 2.000 M…
A: General Chemistry question Explanation:Approach to solving the question: General approach Detailed…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. Assume that the water side…
A: Carbonyl compounds react with a secondary amine to form an enamine. Enamines are nitrogen analogous…
Q: Determine whether the following solution is acidic, basic, neutral, or more information is required.…
A: Given:Determine the type of soltion.
Write notes on the most important absorptions and fragmentations found in the following spectra and use the information to suggest the best possible structure. (Molecular formula: C8H1602, M =144)...
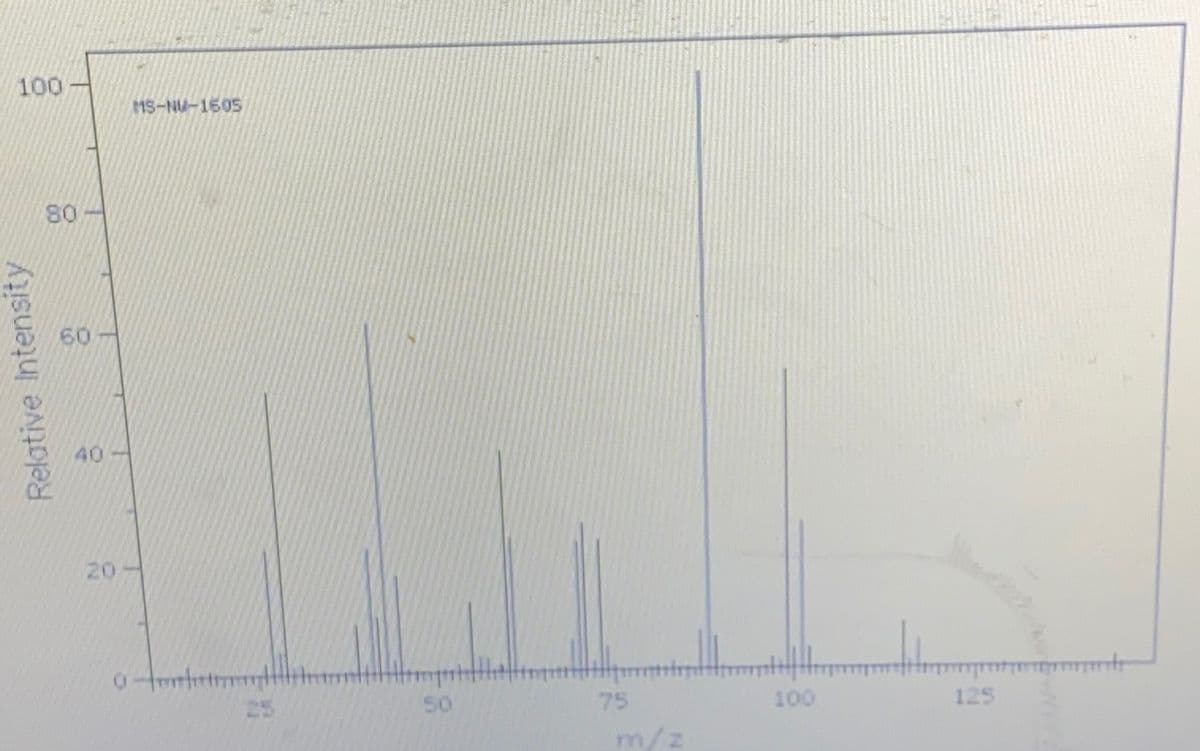
Step by step
Solved in 4 steps with 1 images

- 5.4.5 Chemical microanalysis5.4.5.1 Exploitation of characteristic X-raysElectron probe microanalysis (EPMA) of bulk samples is now a routine technique for obtaining rapid, accurate analysis of alloys. A small electron probe (100 nm diameter) is used to generate X-rays from a defined area of a polished specimen and the intensity of the various characteristic X-rays measured usingeither wavelength-dispersive spectrometers (WDS) or energy-dispersive spectrometers (EDS). Typically the accuracy of the analysis is š0.1%. One of the limitations of EPMA of bulk samples is that the volume of the sample which contributes to the X-ray signal is relatively independent of the size of theelectron probe, because high-angle elastic scattering of electrons within the sample generates X-rays. The consequence of this is that the spatial resolution of EPMA is no better than ¾2 µm. In the last few years EDX detectors have been interfaced totransmission electron microscopes which are capable of operating…Calculate the resolution required to resolve the following peaks; i) CH3N+ (MW = 28.0187) and N2+ (MW = 28.0061) ii) C2H4+ (MW = 28.0313) and CO+ (MW = 27.9949) iii) C3H7N3+ (MW = 85.0641) and C5H9O+ (MW = 85.0653) iv) 116Sn+ (At W = 115.90219) and 232Th2+ (At W = 232.03800)Below is given the FT-IR (Fourier Transform Infra Red) data of a 1:1 (by weight) mixture of ethylene carbonate (EC) and dimethyl carbonate (DMC). Absorbance Pis given as a function of wavenumber, m. Regress the above data to a second order polynomial P = a, + a, m+ az m? Find the absorbance at m = 1000 cm-1 Wavenumber, m Absorbance, P (arbitrary unit) ( cm-1) 804.184 0.1591 827.326 0.0439 846.611 0.0050 869.753 0.0073 889.038 0.0448 892.895 0.0649 900.609 0.1204
- How to get coupling constants from this spectra. Values at the top are in ppm. The instrument is 300 MHz.Calculate the resolution required to resolve peaks for N2+ (MW = 28.0061) and CO+ (MW = 27.9949)Peaks of scattered intensity are observed when the path difference would becomes equal to __________ λ/2 λ 2λ 3λ/2
- Calculate the (a) Linearity constant (r), (b) y-intercept, (c) Slope, and (d) Protein content of an unknown sample having an absorbance of 0.325.Mass spectral measurements were made on 10 replicate low concentration Cd-EDTA samples: 181.5, 148.5, 158.5, 173.5, 142.5, 211.9, 163.5, 168.1, 157.5, and 175.1 counts. Ten measurements of a blank had a mean measurement of 42.1 counts. A sample containing 2.00 µM Cd-EDTA gave a measurement of 1827 counts. Estimate the signal and concentration detection limits for Cd-EDTA. signal detection limit: counts concentration detection limit: µMA 19.8474 gram vial is used to calculate ethe partition coefficient of benzoic acid in a 50/50 DCM/water mixture using 59.9 mg of benzoic acid. If the vial + benzoic acid from the DCM layer weighs 19.8819 grams, what is the partition coefficient (DCM/water)?
- Describe the origin of shot noise in a spectrophotometer. How does the relativeuncertainty vary with concentration if shot noise is the major noise source?The resolution product of Ag2SO3 is 1.5 x 10-14. Calculate E0 for Ag2SO3 (k) + 2e- ↔ 2 Ag(k) + SO32-. Ag+ + e- ↔ Ag , E0 = + 0.799 VCalculate the Ksp for Ni(OH)2 Ni(OH)2(s) + 2 e- ⟶ Ni(s) + 2OH-(aq); Eo = -0.720 VNi2+(aq) + 2 e- ⟶ Ni(s); Eo = -0.250 V 7.56x1015 1.15x10-8 1.70x10-33 1.32x10-16

