Q: Determine the precision of the graduated cylinders, and get the volume with the right significant…
A: Determine the precision of the graduated cylinders, and get the volume with the right significant…
Q: a. Estimate the reading from the following ruler to the appropriate precision a. b. Round…
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for…
Q: Need to see work on how to know how many decimal places to go after doing conversions
A: Conversion:
Q: conversion factor
A:
Q: An empty graduated cylinder has a mass of 30.760 g. 50.0mL of an unknown liquid are added and the…
A: Given: Mass of empty graduated cylinder=30.760 g Mass of liquid + Mass of graduated cylinder=72.810…
Q: 5.00Mm to m
A: given: 5.00 mm we have to convert milimeter to meter. now, 1000 mm = 1 m now we have to divide the…
Q: Based on the measurement uncertainty of the below instruments, which instruments would yield the…
A: The apparatus that gives more accurate values, would yield the lowest percentage error.
Q: a. (1.70) (7.87 x 10-3) (7.986 × 10³)-(4.86 b. x × 10²) X C. (7.986 × 10³)+(4.86 × 10²) X 8.306 d.…
A:
Q: O MEASUREMENT Interconverting compoun... Convert uITC iollowing measurement. 3 kg g 4.9 × 10 3 %3D m…
A:
Q: cubic angstroms (1 angstrom = 10−¹⁰) Express your answer in cubic angstroms to two significant…
A:
Q: determine the precision of each measurement. 23.9m 14050ft 5.0 x 10^4s
A: The word Precision is defined by how closely a measurement matches the same measurement when…
Q: The radius of a dust speck is 4.5 * 10-3 mm. What is the correct value of thisnumber in decimal…
A: Given The radius of a dust speck = 4.5 x 10-3 mm
Q: 3.52
A: Dear student I have given answer to your question in the image format.
Q: Make an introduction about Precision and Accuracy. Cite your references
A:
Q: Every hour a quality control manager records the temperature ( in degree centigrade) in the…
A: Data recorded after some constant interval of time is a data of interval level. It is given that a…
Q: Measurements have shown that the concentration of salt in seawater is 35.5 g·L 1 '. Calculate the…
A: Concentration is basically the availability of the solute per unit volume of solution. It is usually…
Q: Determine the precision of the graduated cylinders, and get the volume with the right significant…
A: Significant figures are the digits of numbers that are the accurate and more reliable value of data.…
Q: Convert the density 2.54 x 10-2 dag/ L to cg/kL. Express all answers in the proper scientific…
A:
Q: Gravimetric means (volume measurement jor: the component to be analyzed .A True .B False .C .D
A:
Q: A 25 mL graduated cylinder has incremental marks every 1 mL. How many significant figures should be…
A: The burette can be read upto accuracy of 0.1 ml only . And the observation is expressed by as many…
Q: Convert the following measurement. 4 1.7 x 10 2 cm %3D ms Help
A:
Q: Limitation of accuracy
A: A graduated cylinder can measure upto 1 mL for 100 mL cylinder and upto 0.1 mL for a 10 mL cylinder.…
Q: [1.35 mm x (13.2 mm - 12)] =
A:
Q: Convert the following measurement. cm -6 6.9 x 10 m ms
A:
Q: The 10-mL graduated cylindar has markings at 1 ml increments and 0.1 ml increments. If the bottom of…
A: The given appearatus is 10 mL graduated cylinder. Graduated increments 0.1 mL.
Q: If a beaker is weighed on a plus or minus 0.01 gram precision balance to be 152.63g and the beaker…
A: Given : Weight of beaker = 152.63 g Precision of beaker weight = 0.01 g Weight of beaker with test…
Q: Rewrite this measurement with a simpler unit, if possible. g cm 6.5 3. cm Note: If you can simplify…
A:
Q: Convert the following values and write answers in scientific notation, where appropriate. 67 L to qt
A: Given:Conversion of 67 L to qt
Q: What is the correct reading for the graduated cylinder in the close up? Make sure to report your…
A: The correct reading of a graduated cylinder is always measured from the lower meniscus. So, the…
Q: 28. 29. 30. -30| -75 100 20 65 90 10 55 80 Graduated cylinders #28-30 were mL. measuring increments…
A: The reading given are,
Q: L00 50 4000 3000 2000 1500 NAVENUN ERI 1000 500
A: The question is based on concept of IR Spectroscopy. It involves molecular structure determination…
Q: You decide that it is too tedious to kneel or adjust your eye level when trying to read the volume…
A: Random error causes one measurement to differ slightly from the next. it comes from unpredictable…
Q: Determine the density of an object that has a mass of 149.8 g and displaces 12.1 mL of water when…
A: Given mass of object = 149.8 g Volume of object = 12.1 mL To find : density of an object
Q: Calculate the relative error, in percentage, of two numerical values i.e., 5.8 and 10. 8
A:
Q: 3) 4) ml. ml 10 2. 8.
A:
Q: 1 E2
A: Precision in graduated cylinder means the to what decimal place the small markings between the big…
Q: Part A Measure the diameter of the circle using Ruler A and Ruler B.Given that the actual diameter…
A: Given : Ruler A and Ruler B . To Find : Some statements are given, put them correctly in the two…
Q: Discuss the accuracy of a graduated cylinder and buret. State the advantages and disadvantages of…
A: Burettes are more accurate than graduated cylinders. A 50 mL burette is more accurate than a 50 mL…
Q: Which of the following are acceptable readings for the volume of fluid in the graduated cylinder…
A: Since 10 spaces makes 5 mL So 1 space makes = (5/10) mL = 0.5 mL…
Q: The mass of substance X is 730 pounds and its density is 20.3 g/mL. Substance Y is 1.45 kg and its…
A: Given: mass of substance X is 730 pounds and its density is 20.3 g/mL. Substance Y is 1.45 kg and…
Q: . A metallic cube measures 15.6 millime (mm) on a side and has a mass of 10.25 g. Find density in…
A:
Q: Convert the following measurement. cm 0.00803 %3D 2. ms
A: The given measurement is: 0.00803 cm / ms2 We have to convert this measurement to m/s2.
Q: e) 12.01 mL +35.0 mL+6 mL f) 0.021 cm * 3.2 cm * 100.1 cm g) 505 kg - 450.25 kg
A: Significant figures rule for addition and subtraction final answer must contain significant number…
Q: Determine the graduated cylinder's precision and report the volume in right significant figures
A: The graduated cylinder is of 5 mL volume . So we have to measure all the reading in the form of 3…
Q: (c) 5.28 mm to cm
A: 5.28 millimeter (mm) is to be converted to centimeter (cm)The conversion can be done as follows:
Q: If it appears that your 50 ml capacity burette is exactly at ten milliliters, how should this be…
A: A general thumb rule states that in a measuring device the measured values can be estimated as one…
Q: An analyst weighs the same tablet three times on a well calibrated analytical balance and obtains…
A: Environmental changes, such as in temperature or humidity. Exposure to harsh conditions, such as…
Q: Perform the following multiple operation problem using the current scientifically established…
A: Applying the BODMAS rule to solve the above question: Where, B=bracket O=of D=division…
Q: Compute (4.29 x 105) · (1.89×10-4). Express your answer to three digits.
A: For this calculation, first of all, we determine the significant figure in the starting numbers of…
Q: Correct! Now, determine the uncertainty value for the measurement. 7.48 ± A mL 7 SUBMIT UNCERTAINTY…
A: Uncertainty is the half of the smallest possible measurment that can be made with that glassware.
Determine the graduated cylinder's precision and report the volume in right significant figures.
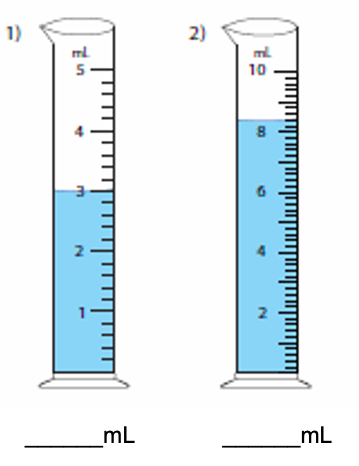
Step by step
Solved in 2 steps


