Q: Calculate the wavelength (in m) associated with a 20Ne atom moving at a velocity of 2.0 105 m/s. MM…
A:
Q: What is the pH of neutral water at 40 °C? [H3O+] = [OH-] = 1.71 x 10-7 M pH = -log[H3O+] pH = [?]
A:
Q: 4.) Explain what happens to the H* and OH concentrations of a solution when the pH goes from 7 to 4…
A:
Q: A galvanic cell is made using 0.25M of Zinc Sulfate and an unknown amount of copper (II) sulfate.…
A:
Q: QUESTION 9 What concentration would you get if you diluted 0.600L of 1.000 M copper(II) sulfate…
A:
Q: True or False Arsenic sulfide is basic in nature.
A: To answer: Arsenic sulfide is basic in nature.True or False
Q: 11) Which is more highly regioselective the addition of HCl to methylenecyclohexane or to 1-…
A: Answer : Regioselectivity: Markonikov's addition reaction is a type of reaction that involves…
Q: Which does not belong to the group?* Pb⁺² + 2Cl⁻¹ ⇌ PbCl₂ Ag⁺¹ + Cl⁻¹ ⇌ AgCl₂ Hg₂ + 2Cl⁻¹ ⇌…
A:
Q: 5. Which one of the following statements about atomic structure is false? A An atom is mostly empty…
A: Atom is made up of three fundamental particles that is electron proton and neutrons .
Q: 20. According to the VSEPR theory, the geometry of the SO, molecule is A. pyramidal. B. distorted…
A:
Q: 4. The following IR spectrum is produced by a compound with C9H10O2 as its empirical formula. What…
A:
Q: 4. How many molecules (or formula units) are in 4.59 x 10³ g Na³PO4 sample? a) 1.69 x 1023 Na₂PO4…
A:
Q: 27.Formaldehyde has the formula CH₂O. How many molecules are there in 0.11 g of formaldehyde? A) 6.1…
A: Given : Mass of formaldehyde = 0.11 g
Q: A chemist is studying the following equilibirum, which has the given equilibrium constant at a…
A: The given equilibrium reaction is: 2CH4(g) ⇌ 3H2(g) + C2H2(g) ; Kp = 9.*10-10
Q: What is precipitimetry?
A:
Q: How are electron configuration and periodic trends related?
A: From the electronic configuration of an element we can know group number and period number of that…
Q: What is the expected product from the addition reaction of 1-pentene (CH₂=CHCH₂CH₂CH₂) with bromine…
A:
Q: 0.10 M hydrofluoric acid + 0.10 M sodium fluoride 0.23 M hydrobromic acid + 0.22 M sodium bromide…
A: Buffer solution is a solution which resist the change in its pH on addition of small amount of acid…
Q: What are the possible values for the missing quantum number in the following quantum number…
A: The state of an electron in an atom is determined by a set of four quantum numbers. They are,…
Q: Describe the trend in atomic size: Describe the trend in atomic radius:
A:
Q: 4NH3(g) +50₂(g) = 4NO(g) + 6H₂O(g) ΔΗ° = -1000. kJ K = 1.35 When the reaction is altered as below,…
A: If the reaction is reversed, change the sign of ΔH. If the reaction is halfed,ΔH should also be…
Q: 1. 2. 3. Pb(NO3)2 _Cl₂ + C3H6+ + ______K₂CrO4 --> KCI + KI --> _0₂ --> PbCrO4 + ____I₂ _CO₂ + H₂O…
A:
Q: 4. What is the expected product of the following reaction? A) OCH 3 B) OH CH 3 C) CH3OH H+ OCH 3 D)…
A: Here protonation occur at 3 position because the generated carbocation is stabilized by 2p-2p…
Q: The diagrams 1 to 3 below can be arranged to show the mechanism involved in a familiar reaction. The…
A:
Q: True or False Cyanide ion forms an unstable colorless complex with copper ions.
A: The electronic configuration of Cu(29 e-) is [Ar] 3d10 4s1 The electronic configuration of Cu2+(27…
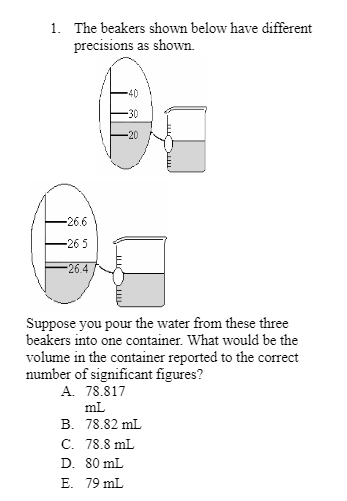
Q: Give the result of the following calculation to the correct number of significant figures. Express…
A: The significant rules are described as follows: 1. In addition and subtraction, the significant…
Q: How many energy (in kj/mol) is released in the fusion reaction of 4-6 with H-2 nuclei to prepare two…
A: In fusion reaction nucleolus fuse to give heavier nucleolus with release of energy
Q: A compound contains only carbon, hydrogen, nitrogen, and sulphur. A combustion analyzer collects…
A: Given, A compound contains only carbon hydrogen, nitrogen and sulpur. The combustion analyzer…
Q: a) • Name the compounds represented by the following line structures b) CH₂CH₂CH3 I CH3
A:
Q: Phosphorus pentachloride gas partially decomposes to phosphorus trichloride gas and chlorine gas. A…
A: Phosphorus pentachloride g →Decomposition Phosphorus trichloride g + Chlorine g1.20 mole of…
Q: 15. Which of the following pairs of elements would be most likely to form an ionic compound? A) CI…
A: Ionic compounds are the compounds which are formed by ions i.e cations and anions. Cation have…
Q: In the generic chemical reaction at equilibrium and given below, the value of the equilibrium…
A: Given : K = 0.43 Concentration of Z = 0.033 M Initial concentration of A & B = 0
Q: give a specific example of gravimetry analysis by enumerating the steps of the analysis
A:
Q: 11) Enterobactin is a molecule produced by bacteria to bind with Fe" ions in solution so that it can…
A: The three-dimensional structure of the molecule is stabilized by intermolecular H-bonding…
Q: What is the cell potential (in V) of a galvanic cell that is described by the following notation? Cd…
A:
Q: 3. How many molecules of ammonia, NH3, are present in 0.522 moles of ammonia? A) 3.14 x 1023 B) 8.67…
A:
Q: Mg(s)-> Wire (V) Voltmeter Salt bridge Wire Mg2+(aq) Ni2+(aq) Mg(s) + Ni2+(aq) →→→ Mg2+(aq) + Ni(s)…
A:
Q: 3.1 Propose a detailed mechanism for the reaction below: AICI 3 CI
A:
Q: Maps ! 1 F1 O A 1 tion N HO H HO HO 11 2 * F2 W S CHO command X )( # 3 CH₂OH H LU 80 F3 E OH H D H…
A:
Q: Calculate the ΔG° (in kJ/mol) for the following reaction at 25.0℃. K^+1(aq) + Li(s) → K(s) +…
A:
Q: Chemical formulas and reactions for water treatment using Chlorine, chlorine dioxide, sodium…
A:
Q: 2. Dip-coating process is one of the methods for electrode surface modification.
A: Dip coating method It is used for coating small sized materials. Dip coating method is a liquid…
Q: The diagrams 1 to 3 below can be arranged to show the mechanism involved in a familiar reaction. The…
A: Here we have to write the correct sequence of steps involved in hydration of epoxide in acidic…
Q: Starting with the active catalyst [HCo(CO)4], sketch the catalytic cycle for the production of…
A: The formation of aldehyde from alkene by HCo(CO)4 catalyst involves a number of steps. These steps…
Q: 16. Which one of the following is most likely to be a covalent compound? E) CaSO4 A)KF B) CaCl₂ C)…
A: The compound forms between metal and nonmetal is ionic compound. and the compound formed between…
Q: 12.An aluminum ion, Al³, has the following charged particles: A) 13 p, 13 e B) 27 p, 24 e C) 13 p,…
A: This can be solve from having idea of atomic no of aluminium as this is positively charge aluminium…
Q: Given : 1CH4 (g) + 2O2 (g) —> 1 CO2 (g) + 2 H2O (g) Q. calculate the mass of c02 gas formed when…
A:
Q: Which of the following is true about the structure below CH, 1 CH₂ - CH₂ - C - CH₂ I CH3 - It is a…
A:
Q: Find the [H+] concentration when it has a Ph of 12.2
A:
Q: What are the constituents which attribute to the alkalinity of a water sample? What are the possible…
A: A question based on environmental pollution that is to be accomplished.
1 answer pls
Step by step
Solved in 2 steps

- At 25% 10.0181g of an unknown liquid was found to have a volume of 6.75 mL.A student dissolves 12.4g of ammonium nitrate (NH4NO3) in 200.g of water in a well-insulated open cup. She then observes the temperature of the water fall from 21.0°C to 16.9°C over the course of 7 minutes. NH4NO3(s) → NH+4(aq) + NO−3(aq) You can make any reasonable assumptions about the physical properties of the solution. Be sure answers you calculate using measured data are rounded to 2 significant digits. Note for advanced students: it's possible the student did not do the experiment carefully, and the values you calculate may not be the same as the known and published values for this reaction. 1. Is this reaction exothermic, endothermic, or neither? 2.If you said the reaction was exothermic or endothermic, calculate the amount of heat that was released or absorbed by the reaction in this case. = kJ 3.Calculate the reaction enthalpy ΔHrxn per mole of NH4NO3. = kJ/molCalculate the volume of liquid in the tank sketched below. Give your answer in liters, and round to the nearest 0.1L
- A student obtains three liquids (A, B & C) in three seperate test tubes. The densities for liquids A, B and C are found to be 2.3 g/ml, 0.95 g/ml, and 0.67 g/ml respectively. Liquid A is poured into Liquid B. Two layers appear. What liquid is likely to be in the top layer?a. Convert 52.6 mM (millimolar) to μM (micromolar) and to M (molar). b. Convert 200.0 μM to mM (millimolar) and to M (molar). c. The new U.S. Public Health Service optimal fluoride concentration in water is 0.700 mg/L. Convert this value to M (molar) and to ppm. Upload your responses here._____1. Which of the ff you can NOT use to measure liquids? a) beaker b) serological pipet c) graduated cylinder d) volumetric pipette _____2. It can be used to contain samples for heating with flames >400 °C a) test tube b) O-ring c) Bunsen burner d) crucible with cover _____3. The ff can be used for boiling a solution of salt, EXCEPT a) beaker b) Bunsen burner c) Buchner funnel d) Wire gauze ____4. Which of the should be used when you want to separate OIL and WATER? a) buret b) separatory funnel c) buchner funnel d) Pasteur pipette
- Based on the charles' law, the following errors in procedure will affect the experimental result. Explain how wach action causes an incorrect result. Procedure(part A) 1) Student A was in a hurry and stopped heating, even though bubbles were still emerging from the rubber tubing. 2) Student B used the measurement of Flask no. 1 to be 250mL, without measuring the volume directed. 3) Student C recorded the temperature in degrees Celcius and used these values in calculations. Procedure(part B) 4) Water remained on the outside of the flask and on the foil when the final mass was determined.Given Benzene - https://fscimage.fishersci.com/msds/02610.htm What is its boiling point (____grams per 100 mL)? Final answer must be rounded off to 2 decimal places and shall NOT have any unit.When the buret in the example delivers from 0.06 mL to 40.00 mL. What is the real volume delivered?
- To control epilepsy, a patient is prescribed 125 mg of phenytoin to be taken three times a day for a week. The volume of 2.5% w/v phenytoin suspension to be dispensed for the prescription is _____ mL. Note: Express answer with two decimal places.kindly help me with this problem solve as neatly as possible and show completesolution. Round your final answer to 4 decimal places and box / highlight all final answers. all values must include proper units with properconversion if needed in your solution. Please follow the format Given,Required,Solution thank you! Additional-the given temperature is 0 degrees 2. The densities of Methane at 0C were measured at several pressures with the followingresults: Pressure (atm) Density(g/L) 1⁄4 0.17793 1⁄2 0.35700 3⁄4 0.53740 1 0.71707 Find the Molecular Weight of MethaneConvert 0.30 mg L-1 to ppb. Put value without unit.

