Q: 10. The internuclear distance of HF is 0.92 Å. The molecular mass of H is 1.00794m, and F is…
A: Step 1:Step 2:
Q: None
A: Step 1:Ozonolysis (O3,H2O2), The first step is ozonolysis,where the alkene (dimethyl cyclopentene)…
Q: The alkaline hydrolysis of the following compound yields: CH3CON(CH2CH3)2
A: The objective of the question is to determine the product of the alkaline hydrolysis of the given…
Q: Draw the Michael adduct and the final product of the Robinson annulation reaction. Ignore inorganic…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Select the major product of the following reaction sequence. NH2 NH 1. CH3l (xs) 2. Ago2 3. heat
A: Step 1: Explanation: Amine reacts with an excess of alkyl halide to form a quaternary ammonium ion,…
Q: H₂N NH2 + HCl
A: The reaction is an acid-catalyzed amide hydrolysis. Amide typically is resistant to hydrolysis even…
Q: CaCO3(s)CaO(s) + CO2(g)Using standard thermodynamic data at 298K, calculate the entropy change for…
A: The objective of the question is to calculate the entropy change for the surroundings when 1.89…
Q: 7 1/1 point Find the solubility of calcium oxalate in pure water and in a solution containing 0.20 M…
A: Solubility refers to the maximum amount of a solute that dissolves in a solvent, usually expressed…
Q: Please don't provide handwritten solution ...
A: Given: Trial123[A],M0.3900.3900.507[B],M0.3500.9100.350Rate,M/s0.01850.1250.0241Step 1: Write…
Q: Imagine the main chain of a protein bends back on itself, so that two amino acid residues R₁ and R₂…
A: Sure, let's delve into each interaction:1. Valine - Alanine (Hydrophobic Interaction): Both valine…
Q: 13. Which of the following are most likely the conjugate acid and base in the reaction below? OH OH…
A: Thank you.
Q: 2. Provide a mechanism for the following transformation: OH H3O + CO2
A: Protonation occurs in the first step then via tautomerism a beta-keto acid forms which on heating…
Q: 12. Collect a sample of lake water; a) 100 mL titrate with 0.05N HCl to HCO3 eq.pt. and we need to…
A: Volume of titrate = 100 mLa) Titrate is titrated with 0.05 N HCl to HCO3-Volume of acid required =…
Q: 5. Devise concise syntheses for the following transformations. Clearly show the reagent and product…
A: Step 1: (5)a. In the first step excess NaNH2 generates the alkyne. Then in the next step NaNH2…
Q: If a container holds 2.75 gallons of a substance that is 50 percent solution/50 percent water, and…
A: The objective of the question is to find out the percentage of the solution within the container…
Q: what is the major product
A: Step 1: Step 2: Step 3: Step 4:
Q: All structure and mechanism I need computer answer expert solutions please
A: Step 1:To determine the configuration of the stereogenic centers in the amino acids shown in the…
Q: Lets say I want to seperate compunds A,B through Column Chromotography. In the image attched 6…
A: Among the given plates, the plate with 10:1 solvent system showed close separation between the 2…
Q: 6) During the Wittig reaction, you perform the reaction as follows:4-chlorobenzaldehyde (136 mg) and…
A:
Q: Draw the product of this Robinson annulation reaction. Ignore inorganic byproducts. O + 0 D KOH,…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Service Departments Production Departments 1 2 A B Total $234 $310 $1,140 $850 $2,534 Service…
A: Explanation:Step 1: Allocate Service Department Costs to Production DepartmentsAllocate Service…
Q: Show work, thank you!
A: To calculate the equilibrium constant (Keq), we can use the relationship between Keq and ΔG°: ΔG° =…
Q: 12. In the hydrogenation reaction shown, which of the alkenes below would yield the observed…
A: In a hydrogenation reaction involving H2 Pd/C, both the hydrogen atoms attach on the same side. This…
Q: 12. Propose a mechanism that synthesizes a-acoradiene from the isoprene units below in aqueous acid.…
A: Biosynthesis means organic synthesis that occurs in nature. The given reaction is the synthesis of…
Q: Provide the systematic name for each of the following isomeric esters with the chemical formula…
A: Naming esters follows a systematic approach based on the constituent parts of the molecule. Here's…
Q: Table 1 Selected standard reduction potentials for half-reactions red measured in Reduction…
A: explained above
Q: Name the following compound. CI
A: In the given question we have to write the IUPAC name of the compound. we have to follow these…
Q: Decide if each of the following molecules is chiral or achiral. CH3 CH3COCH CH3 OH CH CHCCH3 CH3 CH3…
A: Step 1: Step 2: Step 3: Step 4:
Q: We said that organometallic compounds are often used to make carbon-carbon bonds. Specifically, with…
A: Step 1:Determine the possible functional groups present in the compound. The compound has the…
Q: Arrange the following radicals in order of increasing stability. A B C D E A<E < C <D <B □<口 ☑
A: Step 1:How to Check Stability:The stability of free radicals can be analyzed based on a few…
Q: 3. (15 points) In the Applied Organometallic Chemistry article the authors compare the relative…
A: Step 1:Higher Efficiency of Pd-NHC2@M Catalyst at Lower Loading The article in Applied…
Q: None
A: Let HA represent the monoprotic weak acid. By definition, a monoprotic weak acid is a weak acid that…
Q: complete the following condensation reaction C. S+ LOCH3 H3CO CH₂ONa CH3OH
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: × Incorrect. Which of the four mechanistic steps is happening? Add curved arrow(s) to draw step 4 of…
A: Step 1: Step 2: Step 3: Step 4:
Q: Modify the given copy of the starting material to draw the major radical bromination product(s),…
A: This is free radical addition of HBr to an alkene. Generally, Br adds to the less substituted…
Q: Draw the resonance structure for CH2NO2
A:
Q: 4. Propose a synthesis of each of the following compounds using the indicated starting material. You…
A:
Q: When the following equation is balanced properly under basic conditions, what are the coefficients…
A: Given: Br2+SO32−→Br−+SO42−Step 1: Write the half-reaction.Br2→Br−SO32−→SO42− Step 2: Balance…
Q: 24. Which compound has a conjugated system? Yo 2 3 a. only 1 b. only 2 c. 1 and 2 d. 1, 2 and 3 25.…
A: Step 1: Step 2: Step 3: Step 4:
Q: A Lewis base is also known as an electrophile. A)True B)False When water evaporates, the entropy of…
A: 1:A Lewis base is an electron donor. Lewis base has lone pair of electrons. Lewis base is known as…
Q: None
A: Hc: This hydrogen is relatively acidic due to the electronegativity of oxygen, which can stabilize…
Q: A chemistry graduate student is given 500. mL of a 0.30M chlorous acid (HC102) solution. Chlorous…
A: To solve this problem, we need to use the Henderson-Hasselbalch equation, which relates the pH of a…
Q: Solid cobalt(II) nitrate is slowly added to 50.0 mL of a 0.0541 M sodium hydroxide solution. The…
A: The objective of the question is to find the concentration of cobalt(II) ion required to just…
Q: 4. Proteases are enzymes that catalyze the hydrolysis of peptide bonds in proteins. The enzyme…
A: Approach to solving the question:Identify the enzyme involved, trypsin, and its substrate…
Q: 1. A balloon was filled to a volume of 2.50 L when the temperature was 30.0∘C. What would the volume…
A: The objective of this question is to find out the new volume of the balloon when the temperature…
Q: 12) Predict the structure of the products resulting from the reaction between limonene, a natural…
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: Find the energy produced per gram of U-235 (atomic mass = 043922 amu) for the neutron-induced…
A: The objective of the question is to calculate the energy produced per gram of U-235 in a…
Q: Of the following, which represents the intermediate that is most likely formed in the first step of…
A: The question is not clear whether the addition to the alkene is using Br2 or HBr. If it is…
Q: None
A:
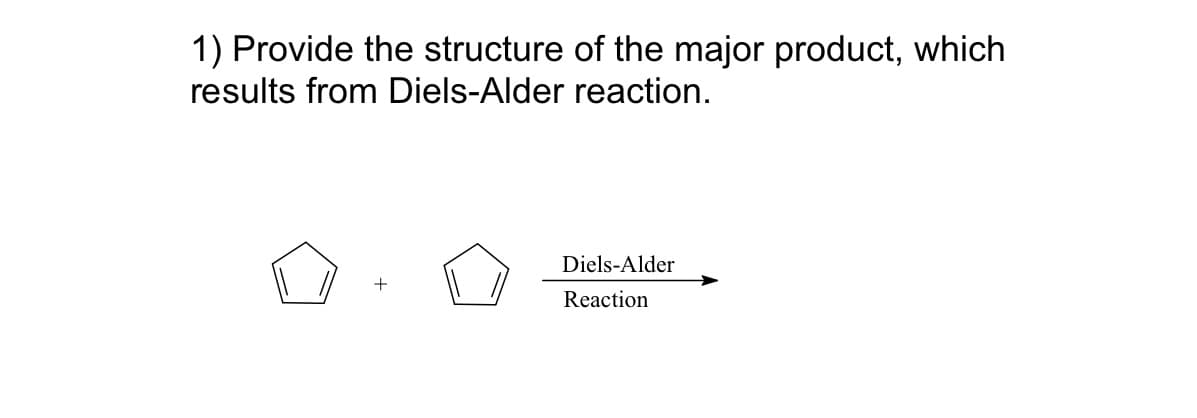
Step by step
Solved in 2 steps with 1 images

- For the following structure, propose the starting materials necessary to form it through a Diels- Alder reaction. Hint: You may need to pay attention to the configuration of the dene and or dienophileThis is a Diels-Alder reaction between cyclopentadiene and maleic anhydride (endo and exo products). For each cycloaddition product, draw in all hydrogen atoms, and write the molecular formula below.Give the structure of a, b, c, d and e in clear handwritten?
- This is a Diels-Alder reaction between furan + maleic anhydride (endo and exo products). For each cycloaddition product, draw in all hydrogen atoms, and write the molecular formula below.What is the product E and how is it formed? Can this be explained using frontier molecular orbital analysis? What would the regiochemistry and stereochemistry be?Rank the following in order from slowest to fastest rate of reaction in a Diels–Alder reaction with buta-1,3-diene
- Which of the following compounds can arise directly from a Diels-Alder reaction? Circle any structures that can. Then, identify the appropriate diene(s) and dienophile(s) that would lead to the compound. Pay attention to E-/Z- stereochemistry in all of the reactants. For those that cannot undergo a Diels-Alder reaction, please explain why.Give the structure of the main product of each of the following Diels-Alder reactions. Make sureto show the stereochemistry of the products when unambiguous.What diene and dienophile are needed to prepare the compound shown below? Please explain
- Draw a detailed mechanism and give the products for the following Diels-Alder reaction. Label the Endo and Exo diastereomer.Diels Adler reaction-- predict the product--- provide mechanism of formation.For each of the following products, draw the structures of the diene and dienophile necessary to synthesize the compound in a Diels-Alder reaction.

