(1) The three (3) component mixture of fluorenone, ferrocene, and 2-acetyloxybenzoic acid (aka aspirin) shown below was loaded on the column and eluted with 95:5 petroleum ether: diethyl ether. Using the Molecular View of the column provided: (a) draw one (1) molecule of each analyte in the correct relative position in the column, and (b) clearly show the relevant intermolecular forces that explain the column elution order. You do not need to include solvent molecules. (8 points) buret wall AI top of column H Al- AI- H H IIIII -0-H -Ö-H bottom of column -‒‒‒‒‒‒‒. buret wall OH
(1) The three (3) component mixture of fluorenone, ferrocene, and 2-acetyloxybenzoic acid (aka aspirin) shown below was loaded on the column and eluted with 95:5 petroleum ether: diethyl ether. Using the Molecular View of the column provided: (a) draw one (1) molecule of each analyte in the correct relative position in the column, and (b) clearly show the relevant intermolecular forces that explain the column elution order. You do not need to include solvent molecules. (8 points) buret wall AI top of column H Al- AI- H H IIIII -0-H -Ö-H bottom of column -‒‒‒‒‒‒‒. buret wall OH
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.18QAP
Related questions
Question
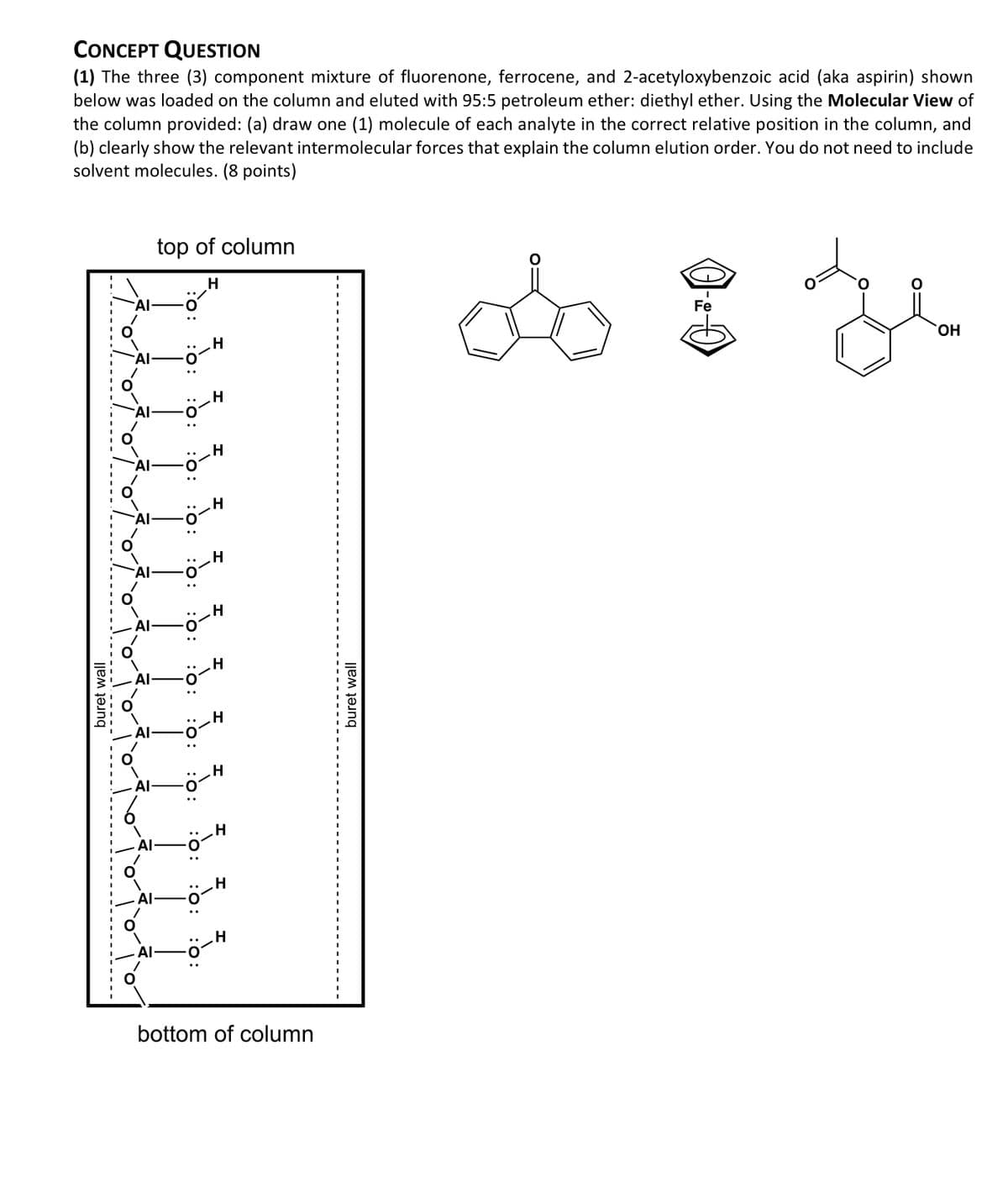
Transcribed Image Text:CONCEPT QUESTION
(1) The three (3) component mixture of fluorenone, ferrocene, and 2-acetyloxybenzoic acid (aka aspirin) shown
below was loaded on the column and eluted with 95:5 petroleum ether: diethyl ether. Using the Molecular View of
the column provided: (a) draw one (1) molecule of each analyte in the correct relative position in the column, and
(b) clearly show the relevant intermolecular forces that explain the column elution order. You do not need to include
solvent molecules. (8 points)
buret wall
Al-
UUUUU
top of column
H
Al-
Al-
Al-
AI
Al-
O
Al-
Al
bottom of column
buret wall
2-0)
Fe
OH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

