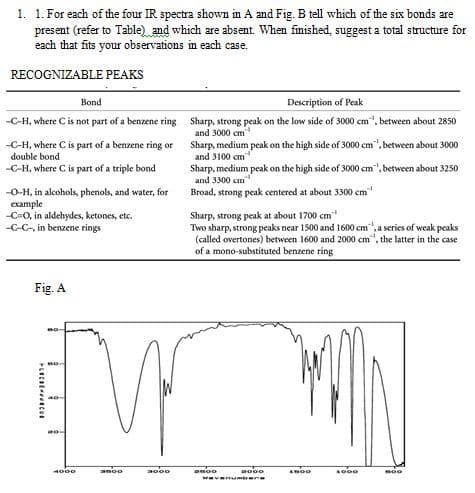
1. 1. For each of the four IR spectra shown in A and Fig. B tell which of the six bonds are present (refer to Table) and which are absent. When finished, suggest a total structure for each that fits your observations in each case. RECOGNIZABLE PEAKS Bond Description of Peak C-H, where C is not part of a benzene ring Sharp, strong peak on the low side of 3000 cm", between about 2850 and 3000 cm -C-H, where C is part of a benzene ring or Sharp, medium peak on the high side of 3000 cm", between about 3000 and 3100 cm double bond -C-H, where C is part of a triple bond Sharp, medium peak on the high side of 3000 cm", between about 3250 and 3300 cm Broad, strong peak centered at about 3300 cm -O-H, in alcohols, phenols, and water, for example Sharp, strong peak at about 1700 cm -CO, in aldehydes, ketones, etc. -C-C-, in benzene rings Two sharp, strong peaks near 1500 and 1600 cm", a series of weak peaks (called overtones) between 1600 and 2000 cm", the latter in the case of a mono-substituted benzene ring Fig. A BO- 1800 1000 BOO 2600 2000 wavenumbere PLECHECO 4000 2000 2000
Carbohydrates
Carbohydrates are the organic compounds that are obtained in foods and living matters in the shape of sugars, cellulose, and starch. The general formula of carbohydrates is Cn(H2O)2. The ratio of H and O present in carbohydrates is identical to water.
Starch
Starch is a polysaccharide carbohydrate that belongs to the category of polysaccharide carbohydrates.
Mutarotation
The rotation of a particular structure of the chiral compound because of the epimerization is called mutarotation. It is the repercussion of the ring chain tautomerism. In terms of glucose, this can be defined as the modification in the equilibrium of the α- and β- glucose anomers upon its dissolution in the solvent water. This process is usually seen in the chemistry of carbohydrates.
L Sugar
A chemical compound that is represented with a molecular formula C6H12O6 is called L-(-) sugar. At the carbon’s 5th position, the hydroxyl group is placed to the compound’s left and therefore the sugar is represented as L(-)-sugar. It is capable of rotating the polarized light’s plane in the direction anticlockwise. L isomers are one of the 2 isomers formed by the configurational stereochemistry of the carbohydrates.
1
Step by step
Solved in 2 steps with 2 images




