Q: The ratio of C-14 to C-12 in a piece of wood from an archeological site was found to be only 0.354…
A: we have to calculate the age of wood
Q: What is the effect of temperature and volume of a gas on its entropy? Entropy of one mole of an…
A: Entropy is a measure of disorder of a system or energy available to do work . As the temperature is…
Q: Which of the following are Brønsted acids? (Select all that apply.) NH4Cl KHSO3 HClO CH3CO2H CaH2
A: Answer: Any specie that gives H3O+ ion in the solution are called as Bronsted acids. H3O+ ion in the…
Q: What is the stereochemistry of chiral carbons in D-Mannose? 2R, 3R, 4S, 5S 2R, 35, 4S, 5S O 2S, 3S,…
A:
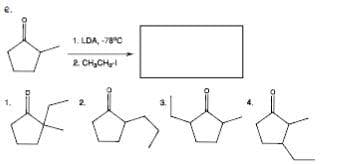
Q: Given the starting material below, design a synthesis to prepare the diketone shown. سال
A: 1) Br2/hv 2) NaOH 3) Na2Cr2O7/H2SO4 4) LDA 5) butanoyl chloride
Q: 3. Identify the reagents in the following scheme. Place your answers in the lines provided HO. HO
A:
Q: 1. Balance the following redox equation by the half-reaction method. This reaction takes place in an…
A:
Q: Part A What is the vapor pressure of SiCl, in mmHg at 23.0 °C? The vapor pressure of SiCL, is 100…
A: Information
Q: Find out what mass of CaSO3 must have been present initially in order for a factory to produce…
A: Since on decomposition of CaSO3, CaO and SO2 gas will be generated. So the following image consists…
Q: A sample of methane gas at a pressure of 0.652 atm and a temperature of 216 °C, occupies a volume of…
A: By concept of Charles’ law- “At constant pressure, the volume (V) of a gas is directly proportional…
Q: A voltaic cell is constructed in which the following cell reaction occurs. The half-cell…
A: Anode is the electrode where oxidation takes place. The cathode is the electrode where reduction…
Q: Q22. You make a standard curve of BSA using the same method as in the first class and obtain the…
A: a) Slope of the line is y=0.055x b) 51uL of acid precipitation fraction c) Absorbance = 0.972 Find…
Q: Transmittance 40 VE 2858; 58.787 8331; 54.315 2925; 40.809 LY 1447; 53.602 1138: 47.353 978; 35.244…
A: The question is based on IR spectroscopy. we need to assign possible functional groups based on…
Q: A chemist adds 0.10 L of a 5.02 mol/L silver nitrate (AgNO3) solution to a reaction flask. Calculate…
A:
Q: The activation energy, Ea, for a particular reaction is 37.8 kJ/mol. If the rate constant at 280 K…
A:
Q: 10) Complete the following reaction by filling in the necessary reagents.
A: Sodium salt is formed on reaction of amine with NaNH2
Q: 1. Give the IUPAC name of the following compounds HO h
A: A question based on nomenclature of organic compounds. Structural formula of 3 organic compounds are…
Q: QUESTION 10 Determine the electron geometry (eg) and molecular geometry (mg) of KrF4. eg = trigonal…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: Can you explain to me about the chiral carbon because I don't get it at all? Also can you give me an…
A: The atom of the molecule which is connected with four different groups or atoms is called chiral…
Q: At the maximum volume of the airbag in the simulation, how many moles of gas are contained? Use all…
A: 1 mole of gas contains 22.4 L volume at STP ideally.
Q: How many mL of 0.100 M NaCl would be required to make a 0.0140 M solution of NaCl when diluted to…
A: The equation to find the unknown value from known values during dilution is given by, M1V=M2V2 where…
Q: Consider the reaction: KCIO4(s)→→KCI(s) +202(g) and the table of values given on the right. Do you…
A:
Q: In a reversible, adiabatic expansion, compressed nitrogen gas (1 atm in 10.0 L, 170 °C, molar Cv,m =…
A: From, the first law of thermodynamics dq = dU + dw where dU is the change in internal energy, dq is…
Q: Trial 1: Atmospheric pressure (torr) Report the atmospheric pressure in units of torr. 29.47
A: Atmospheric pressure is the measurement of pressure exerted by air at normal temperature.
Q: A solution is prepared at 25 °C that is initially 0.21 M in benzoic acid (HC H₂CO₂), a weak acid…
A: Concentration of benzoic acid = [HC6H5CO2] = 0.21 M Concentration of potassium benzoate = [KC6H5CO2]…
Q: What is the predicted product of the reaction shown? ον ο ο ο ο ο >= 2 O IV Oll Br 1. NaN3 2. H₂, Pd…
A:
Q: How many ml of 0.643 Molar HCl are needed to react completely with 72.3 ml. of 0.302 M NaOH…
A: Sodium hydroxide reacts with hydrochloric acid, to form sodium chloride and water. The equation for…
Q: Calculate the change in standard entropy (AS, J K-¹) for the following reaction: 2 NH3(g) N₂ (g) + 3…
A: Change in entropy is the measure of degree of randomness or disorder in the system. It is an…
Q: À voltaic cell is constructed from a standard Co²+ | Co half cell Eºred = -0.280V) and a standard…
A:
Q: What type of orbital is designated n = 4, l= 2, m= 2?
A: Principal quantum number, n = 4 Azimuthal quantum number , l= 2 Magnetic quantum number, m= 2
Q: How many orbitals have the following set of quantum numbers: n=5, l=3, mi=-1
A: Given, Principal quantum number , n = 5 Azimuthal quantum number , l = 3 Magnetic quantum number, mi…
Q: in electrophilic aromatic substitution at the meta position? O I, III, and IV OV ● III 01 00 H E IV…
A: The given reaction is an electrophilic substitution of nitrobenzene. Nitro group is a meta directing…
Q: A sample of limestone and other soil materials was heated, and the limestone decomposed to give…
A: 1) mass of CO2 = 2.45 g = total mass of limestone sample = 6.671 g mass percent of CaCO3 = ?…
Q: A chemistry graduate student is studying the rate of this reaction: H₂CO3(aq) → H₂O (aq) + CO₂(aq)…
A: Answer: This question will be solved using trial and error method where we will put this data in…
Q: When heated to a temperature of 311 "C, formic acid, which has the molecular formula of HCOOH, will…
A: The integral rate equation for a first-order reaction is: [A] =[A]o e-kt At t1/2, [A] = [A]o /2. e-k…
Q: What is the predicted major product of the reaction shown?
A: The amine gets methylated in excess to form quaternary ammonium halide salt.
Q: Hydrazoic acid (HN₂) is a weak acid with a K, of 2.5 x 105. What will be the equilibrium pH of a…
A: HN3 is a weak acid, hence, HN3 exhibits an equilibrium with its ions as…
Q: Which of the following would lead to an increased rate of reaction for gaseous reactants? (A)…
A: Rate of reaction for gaseous reactant depends on temperature because as the temperature increase…
Q: What is the pOH of a solution that has a hydronium ion concentration of 1.2 x 10-8 M? 6.08 7.92 1.2…
A: Concentration of the hydronium ion = [H3O] = 1.2 × 10-8 M pOH of the solution = ?
Q: If a gas experiences 2.351 atm of pressure within a container at 285.6 K, then what pressure would…
A: Initially, Pressure (P1) = 2.351 atm Temperature (T1) = 285.6 K Later, Pressure (P2) = ? Temperature…
Q: pka of butanoic acid is 4.82. Round your answer to 2 decimal places. Note for advanced students: you…
A:
Q: Suppose an equimolar ideal liquid mixture of A and B initially at 1000C and 200 kPa is isothermally…
A: Initial pressure of the mixture = 200 kPa Saturation pressure of A = 120 kPa Saturation pressure of…
Q: Shown below is the ¹H-NMR spectrum of a compound with the formula C5H10O2. 2H Peak Splitting quartet…
A: NMR spectroscopy is a very important tool for determine the structure of the organic compound. The…
Q: Choose the correct order of reagents for performing the following transformation. 1) OEt A) 1) NaOEt…
A:
Q: Mass of salt (g) Volume of DI water (mL) Mass of DI water (g) Temperature of DI water (°C)…
A:
Q: Under normal conditions, the human adrenal medulla secretes epinephrine (C9H13NO3) at a rate…
A: Molarity is number of mole present in 1L of a solution.
Q: For each of these molecules, identify the proper MO diagram and the number of valence electrons. The…
A: The molecules given are B2, C2, N2 and O2.
Q: The hydronium ion concentration of a solution increases. Select all of the following that will occur…
A: Given that The hydronium ion concentration of a solution increases. pH = -log [H3O+] pH + pOH = 14…
Q: Which one of the following is least likely to undergo SN1 reaction with CH3OH ? Br d) a. CH3 Br H₂C.…
A: In SN1 reaction rate of reaction depends on concentration of alkyl halide. But it is independent…
Q: It can be shown that a 1.0-mg sample of 232 Th decays at the rate of 4.1 a emissions per second. Set…
A:
Step by step
Solved in 4 steps with 4 images

- why would you write 13.90 when only two sig figs were given? Where does the extra "precision" come from?https://www.youtube.com/watch?v=wS872X6_ZwwTo determine the mass of Fe2O3 from the given mass of K3Fe(CN)6, the gravimetric factor to be used is a. Fe2O3 / K3Fe(CN)6 b. Fe2O3 / 2K3Fe(CN)6 c. 2Fe2O3 / K3Fe(CN)6 d. 3Fe2O3 / 2K3Fe(CN)6
- 789mK/s to uK/ms. Express your answer in three significant figures.EXPLAIN ME LIKE I M 5. PLEASE IN DETAIL WITH HANDWRITTENA chemist obtained the following data for the percent compound Z in triplicates (n=3) of an insecticide preparation: 7.47, 6.98, and 7.27. Calculate the 90% confidence limit for the mean of the data assuming that only information about the precision of the method is the precision for the three data points. 7.24 ± 0.74% 7.24 ± 0.13% 7.24 ± 0.42% 7.24 ± 0.27%
- A chemist obtained the following data for the percent compound Z in triplicates (n=3) of an insecticide preparation: 7.47, 6.98, and 7.27. Calculate the 90% confidence limit for the mean of the data assuming that only information about the precision of the method is the precision for the three data points.You work in a research organization that is looking for markers of various diseases that can be used as a diagnostic for the disease. It has been reported in the past that high levels of Cu are found in the sweat of people with cystic fibrosis. One of the research projects is focused on looking for high levels of Cu in samples that can be obtained non-invasively such as saliva, sweat, hair, nails, etc. The lab will analyze large samples for Cu. What instrument would you recommend purchasing to support this work, Atomic absorption spectrophotometer or an inductively coupled plasma atomic spectrophotometer? Explain the basis for your decision.How is (1/T_2) - (1/302.6K) Calculated? Thanks.
- What is the simplest CGS unit for "poise"?Explain why Fe3+ is in large excess in the standardization procedure. Fe3+ + SCN- → FeNCS2+A. B. and C. already solved (https://www.bartleby.com/questions-and-answers/chemistry-question/a89691d6-2162-4677-b0cc-dc84fcceda08). Please answer E, F and GCircle in pencil in the image on th eirght cide of the diagram is a "2"( Pb(NO3)2 ).

