Q: Which bond in each of the following pairs of bonds is the strongest? H-F of H-
A: Solution- A chemical covalent bond is formed by equal sharing of electrons between two bonded atoms.…
Q: 1.71 Answer the following questions about compound A. a. Label the shortest C–C single bond. b.…
A: Hybridisation is defined as concept of intermixing of orbitals. There are three different types of…
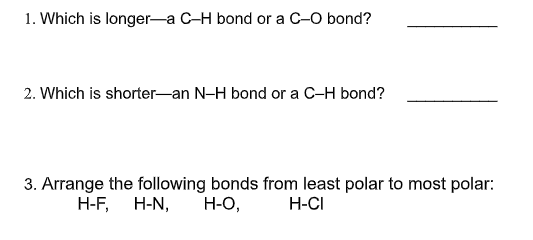
Q: 16. Among N-H, H-H, O-H, C-H. is the least polar bond, and is the most polar bond. A. H-H and 0-H B.…
A: The polarity is observed by the difference in electronegativities. Here, the respective…
Q: Rank the following bonds in order of strength from weakest to strongest: C-C C=C C-H C-O C=O C-F…
A: Bond strengh :- The strengh of a covalent bond is measured by its bond dissociation energy, that is…
Q: 20. Which is the most polar bond? (а) С-С (b) С-Н (c) N-H (d) О-н (e) Se-H
A: Relationship between polarity of bond and electronegativity difference (∆EN) Polarity of bond can be…
Q: Which bond would be the strongest? ON=N ON=N ON-N OH
A: We have to find the strongest bond .
Q: . Identify the a. shortest C-H bond b. longest C-H bond 2. Predict the C-C bond that has the…
A: A question based on chemical bonding that is to be accomplished.
Q: Rank the bonds listed from most to least polar (most polar = 1, least polar = 3) a. H-Br b. Ag-Cl…
A: We have to rank the bond from the most polar to least polar.
Q: A C H H H H-c=C-c-CEC-H H
A: We are to discuss which carbon-hydrogen bond is easier to break in the given structure.
Q: In which bond does the H atom have the highest electron density? Select one: а. Н-Li b. H-N с. Н-СI…
A: Solving only first question in accordance with guidelines. Greater the electronegativity of the…
Q: Use the observed bond lengths to answer each question. (a) Why is bond [1] longer than bond [2] (143…
A: The C-OH bond in carboxylic acid shows resonance stabilization due to which the bond length of C-OH…
Q: Which bond is most polar? O A. O–H O B. F-H O C. N-H O D. CI-H
A: Which bond is more polar ? A. O-H B. F-H C. N-H D. Cl-H
Q: Choose the bond below that is MOST polar. O H-F O H-I O H-Br O H-CI O C-H
A: Polarity of bond : The bond polarity mainly depends on the electronegativity difference of the two…
Q: ich bond in the compound below is considered to be polar covalent? H. Н-о-с-С-о-н H H O The O-H…
A: The compound given is,
Q: Rank the bonds listed from most to least polar. а. Н-Br b. Ag-CI C. S-I H-Br Ag-CI S-I
A:
Q: See the Attached compound A. Ques: Label the shortest C–C single bond.
A: Given:
Q: Which bond is most polar? O A. O-H O B. F-H O C.N-H
A: With the increase in the electronegativity difference between the two bonded atoms, the polarity of…
Q: ing have the SHORTEST C-C bond?
A: Bond order is inversely proportional to bond length. As the bond order increases, bond length…
Q: c. How many sigmas -bonds and how many -bonds are present in the Acetic Acid molecule? sigma-bonds :…
A: The solution for part c is as follows :
Q: Rank the following bonds in order of increasing strength:
A: As we move down the group the electronegativity( tendency to attract shared pair of electrons)…
Q: Which of the following bonds is the most polar? a. H-Cl B. C-CL C. H-H D. N-F
A: We can easily predict the polarity of any bond by considering the electronegative difference between…
Q: Which bond would you expect to be the most polar? O C-0 O N-C O C-F O N-O O P-C
A: Please find your solution below : Polarity depends on the electronegativity difference. More is the…
Q: Which of the following bonds is the most polar? a) C-O b) C-H c) C-S d) C-N
A: Larger the difference in electronegativity between the two atoms, more the polarity of the bond.
Q: 4) How many sigma bonds are in 2-butyne (H3C-C=C-CH3)? H H H C-C=C-C-H H H а. 3 С. 9 b. 6 d. 11
A: Given, The number of sigma bonds are in 2-butyne (H3C-CoC-CH3) is:
Q: Which has a greater dipole moment? CH2Cl2 or CH2F2
A: CH2F2 has a greater dipole moment. CH2F2 is a polar molecule. And yes considering Fluorine’s…
Q: Of the bonds below, which is the least polar. C-O N-O C-F S-O K-Br
A:
Q: How many o and à bonds are in this molecule? H H H number of o bonds: ━H number of bonds:
A:
Q: Which of the following bonds is the most polar? O N--H O P---H O C-H O B--H
A: Given Bond N----H P----H C -----H B -----H Most polar bond = To be determined
Q: In the following set, indicate the most polar bond. H-H H-Si H-P H-S
A: Relationship between Ionic character and electronegativity difference (∆EN) The polarity of the bond…
Q: 4) Choose the bond below that is the strongest. A) C-Cl В) С -О С) С-Br D) I-I E) C=C
A: The bond strength of any bond is directly proportional to the bond length. And bond length of any…
Q: Choose the bond below that is most polar. O C-H O H-Br H-I H-CI H-F
A: Polarity is the measure of electronegativity difference of the two atoms. Higher the…
Q: Which bond is most polar? O H-S H-H O H-P O H-CI
A: Given :- H-S H-H H-P H-Cl To determine :- The most polar bond
Q: Which bond is more polar? N--H or Cl-O
A: Which bond is more polar? N--H or Cl-O
Q: Which of the labeled C-H bonds is the WEAKEST? На- Hc. He A C-Ha B C-Hp C-Hc D C-Hd
A: When the conjugate anion gets stability then the C-H bond will be weak.
Q: Which bond in attached pair has the higher bond dissociation energy?
A: As the atomic size increases on moving down in a group of periodic table. Hence the size of S >…
Q: 7) Re-draw the following molecules showing the full Lewis structure at the indicated location. NO2…
A:
Q: 46. Which of the following bonds is least polar? a) C-O b) H-C c) P-Cl d) Na-Cl
A: There are different type of bonding present between two atoms in a Molecule. The bond polarity of…
Q: Which bond is most polar? C-F C-O C-I C-Cl C-Br
A: In chemistry, a polar bond is a type of covalent bond between two or more dissimilar atoms, in which…
Q: Rank these bonds in order of increasing polarity: (a) F-H, (b) S-H, (c) P-H.
A: Polarity of bond can be determined by the electronegativity difference between the atoms. When…
Q: Which of the structures shown below has two sigma (o) bonds? H-Ċ-H H-Ñ=ö: :Ci-C-Ċi: :CEO: O CCI20 O…
A: Single bond has only one sigma bond Double bond have one sigma and one pi-bond. Triple bond have…
Q: Which of the structures shown below has three sigma (o) bonds? H-C-H H-N=ö: Ci-C-Ci: :CEO: O CH4 O…
A:
Q: d) An assignment of polar or nonpolar molecule. (Draw in the bond dipolos to ahow how you arrived at…
A: Polar molecule -----> Has net dipole moment Non-polar molecule -----> Zero dipole moment
Q: Which is the most polar bond? a) C-C b) C-H c) N-H d) O-H e) Se-H
A: Polar bond is generally, formed between atoms in which electrons present in the bond are unequally…
Q: Which bond is more polar? C--S or S-O
A: The polarity of a bond: It is the dispersion of electric charge along with a bond, resulting in an…
Q: 16. How many central atoms does the molecule H2NCH3 have, and what is the shape about each? H. H- N-…
A: The shape around any atom is given by the sum of total bond paired and total non-bond paired…
Q: a. Which bond would be longer? b. Which bond would be stronger? 1. C - Cl or C - I 2 C - C or C - Cl…
A: a. Bond length 1. C - Cl < C - I 2 C - C < C - Cl 3 H - Cl > H - F
Q: which of the follo whs is the bond with the Strongest dipole? a.) H-0 b.) H-S d.)c-H PrJ o-0
A: Ans is A
Q: In which bond does the H atom have the highest electron density? Select one: O a. H-Li O b. H-N c.…
A: Since you have posted a multiple question so we have solved first for you. If you want any specific…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- Which of the following are nonpolar bonds? A) I - I B) Na - F C) C - O D) K - Cl E) O - O F) N - O G) Cu - O H) C - N I) I - Cl J) All of the above K) None of the aboveThe bond length of NaF is 231.00 pm. What would be the dipole moment, in D, of this compound by assuming a completely ionic bond? Remember e = 1.60x10-19 C; 1 D = 3.34x10-30 C*mArrange the following bonds from the least polar to the most polar bond: N-O, Ca-O, Si-O, O-O What information do I need to arrange the bonds from the least polar to the most polar bond?
- Arrange the following bonds in order of increasing polarity:O-Cl, C-Cl, H-Cl, and F-Cl O-Cl < C-Cl < F-Cl < H-Cl C-Cl < O-Cl < F-Cl < H-Cl H-Cl < F-Cl < C-Cl < O-Cl O-Cl < C-Cl < H-Cl < F-Cl C-Cl < O-Cl < H-Cl < F-ClCan you explain to me from part A through part E, please? a) H-H nonpolar bond b) H-F polar bond c) N - - - N nonpolar bond d) O-H polar bande) 0=0 nonpolar bondplace the following bonds in order of polarity highest to lowest. Is this the correct order? O-K<O-H,<Cl-As< N-N
- Use Paul ing electronegativit ies to predict the polarities of the bonds (a) P-H. (b) B-H.M I-Br F-I F-Br The most polar bond is25.Which of the following does have H- bond? electronegativities: ) = 3.5, H = 2.1, C = 2.5, Cl = 3.0, S = 2.8 O || I. H- C- O-H II. SCl2 III . H | H - C - S -H | H IV. H H - C - O - H | H a.IIIb.IIc.I, II, IVd.I and IV
- a. Which bond would be longer? b. Which bond would be stronger? 1. C - Cl or C - I 2 C - C or C - Cl 3 H - Cl or H - FPart 1: Which of the following are ionic bonds: A) I - I B) C - O C) I - Cl D) N - O E) C - N F) Cu - O G) O - O H) Na - F I) K - Cl J) All of the above K) None of the aboveArrange the following bonds in order of decreasing polarity: H-F, H-O, H-N, H-C, H-B, H-Be

