Q: Calculate the value of f and the uncertainty of f (i.e. Af) given the input values for a b, and c.…
A: Given,a= b=c=To find the value of f and .
Q: Calculate the volume in milliliters of a 2.43M iron (II) bromide solution that contains 75.0 g of…
A: The objective of the question is to find the volume of a 2.43M iron (II) bromide solution that…
Q: (19) What is the major product of the following reaction sequence ? Br Br HBC ta 8-14 он atay LOH…
A: Alkyne is electron rich so it can act as a nucleophile .Alky halide and Mg metal form Grignard…
Q: HO 2.0 == (11) I2, PPh 3 imidazole, CH₂Cla 7 OIC. H
A: The given chemical reaction involves the replacement of hydroxyl group with the alkene group in the…
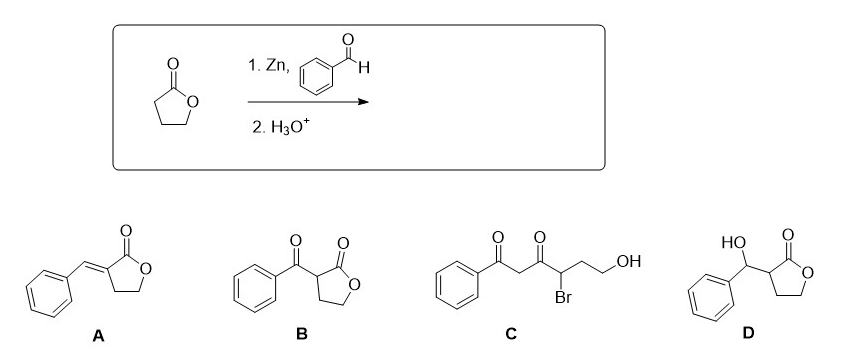
Q: NaOH Ph EtOH, heat
A: The reactant is an aldehyde. It reacts with a base sodium hydroxide and subsequently with the…
Q: Draw an equivalent resonance structure that minimizes charge. Include all lone pairs in your…
A: Resonance structures are formed as a result of the delocalization of pi or lone pair electrons…
Q: 8. Select the substance that is not in its standard state. a) NaCl(s) b) H₂O(1) c) C(s; graphite) d)…
A: The objective of the question is to identify the substance that is not in its standard state.
Q: 10.19 For each reaction, draw the complete detailed mechanism and the major product. NaCl HCI ? ?
A: Given:The reaction of 2-phenyloxirane with nucleophiles is being considered.Epoxide ring opening is…
Q: 8.50 g of a certain compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have…
A: The objective of the question is to determine the molecular formula of a compound X, which is known…
Q: A chemist adds a chemical to pure water and there is a 100-fold incre approximation of the new pH…
A: The objective of the question is to calculate the new .
Q: 4.- Provide the products of the following reactions. As well as the mechanism from which they form +…
A: Given that, the reaction is:
Q: хан NaOEt EtOH
A:
Q: 001 TRANSHETTANCETT 50 IR: CCI solution пр lent
A: The question is based on the concept of organic spectroscopy. We need to analyse the spectral data…
Q: Each row of the table below describes an aqueous solution at about 25 °C. Complete the table. That…
A: Three numerical problems based on pH calculation, which are to be accomplished.Formula pH =…
Q: 11.- According to geometric isomerism, provide what isomer each molecule is: a) |||| b) FA FA
A: Constitutional isomers : Functional isomers are the constitutional isomers which share the same…
Q: What is the product of the following reaction ? NO₂ temp. ?
A: The Diels-Alder reaction is a powerful and widely used method in organic chemistry for constructing…
Q: H3C Draw the alkene product of this Wittig reaction. Ph3P CN ?
A: The conversion of carbonyl compounds into alkene by the reaction with the triphenyl phosphonium…
Q: The bond angles in antimony trifluoride are 87°. Describe the bonding in SbF3, including a picture…
A: The bonding in a molecule can be explained as a result of overlapping interactions between the…
Q: Starting with ammonia as the nitrogen source, the compound shown can be practically synthesized…
A: We have to prepare the given compound from ammonia.To solve this question we should know the basics…
Q: * Incorrect. The conjugate base of diethyl malonate can serve as a nucleophile to attack a wide…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: 3 - Which statement BEST describes the distinction between a hydration and a dehydration reaction?…
A: Here we have to choose the correct statement out of 5 statements about hydration and…
Q: Which one is the major product for the shown reaction? OH H₂C-C-CH3 CH3 O А OB C OE Na2Cr2O7 H2SO4 A…
A: In the question asked we need to find the major product out of the given options.To solve this we…
Q: The lone pairs on the oxygen atom in O both localized O both delocalized O one localized and one…
A: The compound belongs to a class of organic compounds called heterocyclic compounds. In this…
Q: 11. The pH of the water is measured as 8.5 and the total concentrations of carbonate species is…
A: Given,The total concentration of carbonate species is: 0.0018 MTo the find concentration of the…
Q: a) to c) show the formulas of rubidium oxides and these are repeated in d) to f). From the options…
A: Answer:
Q: 1 Which reaction sequence cen most efficiently bring about the following transformation? * a) DC1₂,…
A: Reactions are processes by which a reactant is converted to a product. During the processes, a…
Q: c. In reaction B, a second isomeric product is expected to be formed in roughly equal amounts to the…
A: SN1 reaction mechanism: This reaction path proceeds through formation of a carbocation…
Q: Scheme 6. Alkynyl Aza-Prins Cyclization of Phthalimido Enyne Hemiaminals 16 N OH BiX3 (1.2 equiv)…
A: The given reaction involves the conversion of alkyne into alkene along with the cyclization.The…
Q: Predict the products of the following reaction. If no reaction will occur, use the NO REACTION…
A:
Q: Which of the following bond-line structures are of the same compound? Oland II OII and III O III and…
A: It is based on the numbering of the parent chain based on the substituents attached.The numbering of…
Q: 1. 2. Submitted Br NaOEt NaOH a = Proton transfer 1. b = Lewis acid/base c = Radical chain…
A: The question is based on organic reactions.We need to explain product formation & identify…
Q: Methyl salicylate (C₂H8O3) (density = 1.18 g/mL) is dissolved in 100 mL of 6.0 M NaOH, and heated…
A: Theoretical is defined as the maximum amount of the product that can be formed in a given chemical…
Q: The following product is classified as a (an) only need to write ONE option. YO product (Michael,…
A: The question is based on the concept of organic retrosynthesis.We need to identify the type of…
Q: What strategy X was used to develop the new drug Y? Select one answer. H₂CO H₂CO CH3 X HO H₂CO H₂C…
A: The given reaction of [4+2] cycloaddition reaction to form the product drug Y. The reactant has two…
Q: On the paper provided, draw the chemical structure of a peptide with a sequence YIQV at pH 14. The…
A: When two amino acids combine, they involve loss of water molecule. This is a type of condensation…
Q: The following retrosynthesis is correct. O True O False Eto OEt
A:
Q: When 15.0 mL of a 1.67 * 10^-4M copper(II) acetate solution is combined with 25.0mL of a 5.93 *…
A: Solubility product quotient, Qsp is the product of the concentrations of the products, each raised…
Q: X Incorrect. Predict the major product obtained when the following compound undergoes a Claisen…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: 12) Write a detailed stepwise mechanism for the free radical halogenation of the molecule below. H3C…
A: The given compound is shown below.We have to draw the radical halogenation mechanism of the given…
Q: Imagine that a chemist puts 5.88 mol each of CH, and O, in a 100 container at constant temperature…
A: The objective of the question is to calculate the equilibrium constant.
Q: Identify a systematic (IUPAC) name for the following compound: CI CI O…
A: Basic rules for IUPAC nomenclature:1) Select longest chain or block of carbon atoms as parent. 2)…
Q: Would a precipitate from if a solution contained 5.0 * 10^-4 M silver nitrate and 2.8 * 10^-4 M…
A: Given,Molarity of silver nitrate = 5.0 x 10-4 MMolarity of potassium chromate = 2.8 x 10-4 M
Q: Write the relative reaction rates for the following reaction: A + 2B C
A:
Q: 3. Please give a mechanism using curved arrows for the following reaction. AIC13 CI
A:
Q: If 5.08 g of copper(II) oxide is reacted with excess hydrogen gas and 2.88 g of copper is collected,…
A: The mass of copper (II) oxide (CuO) is 5.08 g.The mass of copper (Cu) collected is 2.88 g.The…
Q: A chemist adds 0.80 L of a 0.134M iron (11) bromide (FeBr.) solution to a reaction flask. Calculate…
A: Concentration of iron(II) bromide = 0.134 M = 0.134 mol/LVolume of the solution = 0.80 Lmillimoles…
Q: Given the following rate law, what is the order of the reaction with respect to Mel? Rate =…
A:
Q: At 45.0 °C, a 14.0 L vessel is filled with 7.25 moles of Gas A and 8.45 moles of Gas B. What is the…
A: From ideal gas law equation P•V = n•R•T P = pressure of gas V = volume R = gad constant T =…
Q: The following product is classified as a(an) you only need to write ONE option. & product (Michael,…
A: The question is based on the concept of organic retrosynthesis.We need to identify the type of…
Major product?
Step by step
Solved in 3 steps with 1 images


