Q: Which of these is NOT true of a good recrystallization solvent? a) The compound is insoluble in the…
A: The process of purification of compounds by crystallizing it again with the help of a solvent is…
Q: pH = 3.4 What are the equilibrium concentrations of H₂A and A²- in this soluti [H₂A] = [A²-] =…
A:
Q: In the body, during glycolysis the following conversion occurs in a 2-step process. OPO. OH 0₂PO OH…
A: The reactant has both a secondary alcohol(-OH) and an aldehyde group(-CHO) that can be oxidized to…
Q: Consider the following reaction: 2HI(g) H₂(g) + 1₂ (9) An equilibrium mixture of the three gases in…
A: Given equilibrium reaction is 2HI(g) ⇌ H2(g) + I2(g) Given that , 13.0 L container has an…
Q: For the following and antibonding orbitals, show how bonding MO can be generated based on the choice…
A:
Q: In the body, during the citric acid cycle the following reaction occurs as the first part of an…
A: Given :
Q: Problem # 2: Determine the enthalpy of the following reaction: 3Fe₂O3(s) + CO(g) 2Fe₂O4(s) + CO₂(g)…
A: Given, Determine the enthalpy of the following reaction, 3Fe2O3(s) + CO(g) ----> 2Fe3O4(s) +…
Q: O CHEMICAL REACTIONS Theoretical yield of chemical reactions Liquid octane (CH, (CH₂), CH,) reacts…
A: Given, mass of Octane reacts = 10.3 g mass of oxygen gas react = 68.8 g Theoretical yield of carbon…
Q: A 5.00 mol sample of an ideal gas is compressed irreversibly and isothermally la
A:
Q: 2. Br CI AICI,
A: The given reaction is an example of Friedel–Crafts acylation which involves introduction of acyl…
Q: Choose all of the processes from below which describe changes which are independent of the path by…
A: A question based on chemical thermodynamics that is to be accomplished.
Q: What is the concentration of A after 12.5 minutes for the reaction A → Products when the initial…
A:
Q: Explain how the stoichiometry between cation-to-anion (or vice-versa) affects whether a given ionic…
A:
Q: ● Question 5: Label each stereogenic center as R or S.
A: GIVEN : Structures of molecules
Q: NICKEL(II) SALT EXP. UNKNOWN # Mass of unknown, (g) Mass of crucible + beaker + precipitate, (g)…
A:
Q: Using the Ideal Gas Law, PV = nRT, where R = 0.0821 L atm/mol K, calculate the volume in liters of…
A:
Q: Insert the four quantities a, b, c and d respectively that are required to balance the equation…
A: Balanced equation is the equation in which equal number of each and every atom is present on both…
Q: 15.0 grams of table sugar (C12H22O11) is added to 200.0 mL of water. Assuming that environmental…
A:
Q: 1. Describe the general principle of spectrophotometry 2. What is Beer's law? What is the…
A: As per the guidelines we are supposed to answer the first three questions to solve remaining…
Q: a. Use strain energy increments in the OWL Table Reference (see References button, Strain Energy…
A: For the first molecule
Q: Sildenafil (ViagraTM) Secondary amide Tertiary amine Primary amide. Secondary amine Primary amine HN…
A: we have to identify the functional group that is circled in red
Q: (d) Consider the three potential reactions shown below. A B с O₂N NO₂ NO₂ Eto Na Eto Na* Eto Na*…
A:
Q: What type of ion is Na+?
A:
Q: 1- Determine the mass of 6.46 mol of Pb, H₂SO4? 2- Determine the moles in 125.0 grams of nickel,…
A:
Q: 6) You are making 100 mL of 1 M phosphate buffer [pKa,1 = 2.12; pKa,2 = 7.21; pka,3 12.33] at pH 7.…
A: Required pH of the buffer solution, pH = 7 A buffer solution is made of a weak acid and its…
Q: Identify the absolute configuration of the chirality centers in each of the following compounds as R…
A:
Q: Identify the absolute configuration of the chirality centers in each of the following compounds as R…
A: Since, Chiral molecule are those whose all four group or atoms are different and that atom which is…
Q: Н. H H H2 Н H₂ CH3 Н CH3 'H
A: For IUPAC naming following steps are required - First of all identify the longest carbon chain…
Q: Draw the structure of a 1°, 2°, and 3° carbocation, each having molecular formula C4H, (only use…
A:
Q: Choose the correct product of the following reaction: H3C О Br CH H3C H3C H3C H3C H₂ Br CH ОН CH Br…
A:
Q: What quantity of atoms of carbon are produced by the complete reaction of 7.09 grams of magnesium…
A:
Q: 1a) 2. ه تھک HD cl - H NC ه H
A: To circle which compound would be more reactive in a reaction with HCN at pH of 10.
Q: The value of K, for acetylsalicylic acid (aspirin), HC,H704, is 3.00×10-4 Write the equation for the…
A: The equilibrium constant of a chemical reaction is the value of its reaction quotient at chemical…
Q: What is the ΔTB of a 1.59 m solution of acetone (C3H6O) in water (Kb = 0.512 °C/m)
A: Given, Molality of the solution = 1.59 m Boiling point elevation constant of water (Kb) = 0.512 °C/m…
Q: (a) What is the molar heat capacity of liquid water? J/mol °C 75 (b) What is the heat capacity of…
A:
Q: Differentiate between crystalline and amorphous solids
A: Crystalline Solids : Crystalline solids consist of particles that are arranged in a…
Q: Does a reaction occur when aqueous solutions of chromium(III) nitrate and copper(II) sulfate are…
A:
Q: HCI C:Ⓒ
A: Reaction-1: The terminal alkyne anion acts as a base(accepts proton). Hence, the alkyl anion takes…
Q: • Question 7: Considering each of the following values and neglecting entropy, tell whether the…
A:
Q: Which of the following description(s) are improperly used related to the process of filtration?…
A: Since you have asked multiple question, we will solve the first question for you.If you want any…
Q: The enthalpy change that is observed during a chemical reaction is denoted byAH which equals:…
A:
Q: your ice cream of choice has 330 mg of sodium chloride per pint. one pint of ice cream has 15 ounces…
A: Given that we have 1 pint of icecream. I ounce = 29.5735 ml of water. Since 1 pint = 15 ounce, =…
Q: Consider the Haber-Bosch process for the synthesis of ammonia from its elements. Calculate the…
A: Given, N2(g) +3 H2(g) →2 NH3(g) Mass of N2(g) : 59.8 g Excess reagent : H2
Q: Atenolol (Tenormin ™M) HO IZ Ether Ketone Carboxylic acid Ester Alcohol "NH₂
A:
Q: Having read the COSH and risk assessment, what is the most dangerous chemical being used as part of…
A: #1: Most dangerous chemicals are those that cause severe skin, eye, and lungs damage and are highly…
Q: For the following reaction, choose the major E2 product. CI CH3 CH3 NaOCH3 CH3OH CH3 Product
A:
Q: If the heat of combustion for a specific compound is -1090.0 kJ/mol and its molar mass is 49.47…
A: Heat of combustion of compound is - 1090.0 kJ /mol. Negative sign indicates that the heat is…
Q: The trigonal-bipyramidal type of molecular geometry involves a central atom and at least: six bonded…
A: Given Trigonal-bipyramidal geometry
Q: The density of water at 4.00°C is 0.967 g/mL. How many molecules of water are present in a 499.8 mL…
A: given : density = 0.967 g/mL volume = 499.8 mL
Q: 2 KMnO4 + H₂SO4 -K₂SO4 + Mn₂07 + H₂O a. How many moles of KMnO4 is needed to produce 6.80 moles of…
A:
Draw the major product of this reaction. Ignore inorganic byproducts
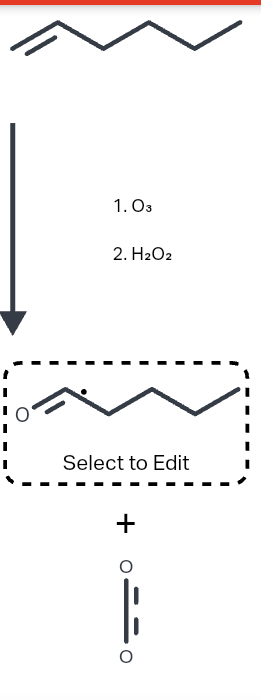
Step by step
Solved in 3 steps with 2 images

- A volumetric calcium analysis on triplicate samples of the blood serum of a patient believed to be suffering from a hyperparathyroid condition produced the following data: mmol Ca/L = 3.15, 3.25, 3.26. What is the 95% confidence interval for the mean of the data, assuming no prior information about the precision of the analysis?IUPAC naminclature2pt/min=___ pt/h using dimensional analysis
- Chemistry (i) For T = 298K ΔG° = -8.314 x 298 x ln (2.83x10-3) = 14.524 KJ/mol (ii) For T = 308.15K ΔG° = -8.314 x 308.15 x ln (9.619x10-3) = 11.891 KJ/mol (iii) For T = 318.15K ΔG° = -8.314 x 318.15 x ln (3.14x10-2) = 9.147 KJ/mol (iv) For T = 328.15K ΔG° = -8.314 x 328.15 x ln (8.82x10-2) = 6.621 KJ/mol (v) For T = 338.15K ΔG° = -8.314 x 338.15 x ln (2.42x10-1) = 3.990 KJ/mol Based on the measured ΔG° values, is this equilibrium spontaneous at room temperature? Which factor, entropy, or enthalpy, has the greater impact on spontaneity in this case? Explain your answers.Ch.2 Q.)3 USE IMAGE AS REFERENCEWhat volume of 95.0% alcohol by weight (density: 0.809g/cm^3) must be used to prepare150 cm^3 of 30.0% alcohol by weight (density: 0.957g/cm^3)



