Q: Draw the product formed when the compound shown below undergoes a reaction with HBr in CH₂Cl₂.…
A: The alkene undergoes Markovnikov’s addition. The double bond attacks the hydrogen ion from the…
Q: Part E. Label the following molecule as R- or S- configuration H H3C CH3 C Br Н CH₂OH H НО II. CH3 Н…
A: For the chiral carbon i.e. the carbon atom having four different groups attached to itself.If the…
Q: Consider the three-dimensional structure shown. Note that only the bonding groups (outer atoms) are…
A: Bond pairslone payersBond angleshape of the molecule10not…
Q: Why is better to describe oxygen using MO theory vs. VB theory?
A: In valence bond (VB) theory bond formation is assumed through the spin-pairing of electrons between…
Q: Draw a structural formula for methanoic acid.
A: To draw the structural formula, we can follow these steps -At first we need to determine the number…
Q: which of the following gas samples would be most likely to behave ideally under the stated…
A: We have to calculate the option in which the gas at a particular condition behaves ideally.
Q: 9. What is the single oxidation number of N in NO₂ (aq)? a. +2 b. -2 C. +3 d. -3 e. -6 10. Balance…
A:
Q: Complete the table. Number of electron groups Bond angle Outer atoms/Lone pairs 2 ? 2/0 3/0 2/1 3 4…
A: According to VSEPR Theory, Steric no. : 2 Electron groups = 2Bond angle= 180°Shape - linear
Q: The solubility of SrCO3 in water at 25 °C is measured to be 0.0045 Round your answer to 2…
A:
Q: Question 12 What is the molar mass of the compound below? 0 131.11 g/mol 133.13 g/mol 136.16 g/mol…
A:
Q: 애
A: The question is based on the concept of IUPAC naming. we need to write name of given compound…
Q: What is the relationship between this pair of structures? CI CI CI pair 1 identical compounds O…
A:
Q: Which pair of ions will form a compound of formula MX2? Sodium and chloride Calcium and bicarbonate…
A: Ionic compounds formed from combination of cation and anions. Charge of cation is neutralized by…
Q: Would you expect a solution of ammonium chloride (NH CI) to be acidic or basic when added to water?…
A: When NH4Cl is hydrolysis with H20 it gives NH4OH and H+Here actually products of hydrolysis are…
Q: Draw a structural formula for the organic anion (i.e., do not include NH4+) formed when ethanal is…
A: Ethanal reacts with Tollens reagent to form which product
Q: 1 Determine the position of equilibrium for each acid-base reaction below. + OH ه OH + میں ۔ + H2O H
A: The question is based on the concept of chemical equilibrium. We need to identify the direction of…
Q: If you were trying to prepare a buffer with ammonia, NH3, what would be the substance that you would…
A: 1. Buffer solution are those whose pH value doesn't chage appricableby addition of small of acid…
Q: A buffer solution is made from 0.650 mol/L ammonia and 0.350 mol/L ammonium chloride (to total 1L of…
A: [base]=[ammonia]=0.650mol/L[conjugate acid]=[ammonium chloride]=0.350M
Q: What reagents are needed to obtain cyclohexyl acetate from 1-cyclohexylethan-1-one
A: To obtain cyclohexyl acetate from 1-cyclohexylethan-1-one, you would typically need the following…
Q: Indicate the stereochemical configuration for the tetrahedral center shown at site 1. R not a…
A: -> Chiral center is center which attached with four different groups.->For R, S following…
Q: Escoja la reacción que no funcionaría como está escrita: O a. O b. 0 с. O d. НО. H₂CO EtO OCH3 O 'Н…
A: choose the reaction which reaction is not possible from the given reactions
Q: Question 15 Which of the following compounds has the lowest boiling point? 2-chloro-2-methyl propane…
A:
Q: (1) (a) and (b) are elements that belong to period 5 and 6 in the periodic table, respectively. Both…
A: (1)The position of silver (Ag) is in the 5th period of the periodic table and lead (Pb) is in the…
Q: Part A. For each pair of isomers A and B, choose the one molecule that is chiral. * Isomer A…
A:
Q: Calculate the AG for the vaporisation of mercury at 24°C using AH = 61.0 kJ mol-1 and AS = 89.0 J…
A:
Q: The most stable carbocation from the following is: O a. IV O b. I O c. 11 Od. III B IV 11 III
A: A carbocation is a species in which carbon is carrying positive charge. In the given question it has…
Q: Draw a structural formula for 2-methylbutanoic acid. • You do not have to consider stereochemistry.…
A:
Q: 8. For the reaction 2H₂O(1) + 2e → H₂(g) + 2OH(aq), calculate the volume of "dry" hydrogen gas…
A:
Q: half-reactions in the reaction
A: Zn-MnO2 battery is a widely used primary battery, in which zinc act as the anode(oxidation occurs)…
Q: For a reversible first-order reaction AB, the rate constant for the forward reaction is 10-2 s-1 and…
A: From integrated rate equation for reversible first order reaction we can determine the the…
Q: 2 molecules of A come together to form an AA dimer. The rate of binding is slightly less than…
A: Dimerization of molecule ARate of forward reaction, i.e Binding or dimerization A-A= 2X106…
Q: ered swer According to MO theory, overlap of two p atomic orbitals produces two л MOs, two л* MOs,…
A:
Q: Consider carbon allotropes diamond and graphite. Unlike diamond, graphite conducts electricity. Why?…
A: In Graphite , carbon atom is bonded with other three carbon atoms and there is one electron left .…
Q: An electric power station annually burns 3.5 × 107 kg of coal containing 2.4 percent sulfur by mass.…
A: Ideal gas equation is the equation which obeys laws of gas under all conditions of temperature and…
Q: What type(s) of intermolecular forces are expected between triethylamine molecules? CH₂CH3…
A: Amines are the compounds which contain badic nitrogen atomswith lone
Q: Part B. Mark each chirality center in each molecule with an asterisk (*). Name Biotin Colchicine HN…
A: A question based on introduction to organic chemistry. Structures of two organic compounds are given…
Q: For the following anion, select (an) allowable resonance structure(s), and (an) allowable…
A: Resonance structures are the collection of Lewis structure which represent the delocalisation of…
Q: Which of the following are representations of the same molecule? (i) CH3CH2CH2CH2CH2CH2CH3 (ii)see…
A:
Q: Methyl acetate is hydrolyzed in approximately 1M HCI at 25°C. Aliquots of equal volume are removed…
A: As we know the rate constant is given by The average rate constant is given by taking the…
Q: 4. In the water and glycerol mixture, the relationship between molar volume and mol fraction of…
A:
Q: How many structures are possible for a tetrahedral molecule with a formula of AX3 Y? structures: 1…
A: Possible structure means in how many ways you represent a molecule,in every way molecule has…
Q: State the time-dependent Schrödinger equation. Subsequently derive the time-independent equation and…
A: The time-dependent Schrödinger equation describes the evolution of a quantum system in time. It is…
Q: The dehydrogenation of gaseous alcohols, RCH₂CH₂OH, to give an olefin, RCH=CH2, plus water on many…
A: Unit of rate constant= (Concentration)1-n time-1 Where, n = order of reactionFor zero order…
Q: How many arenes are present in vancomycin? a) 1 b) 2 c) 3 d) 4 e) 5
A: The simplest arene is benzene (C6H6). Arenes are substituted benzene rings.
Q: A chemical engineer is studying the two reactions shown in the table below. In each case, he fills a…
A:
Q: 35.0 mL of 0.255 M HNO3 is added to 45.0 mL of 0.660 M Mg(NO3)2. What is the concentration of…
A: Volume of HNO3 = V1 = 35.0 mlMolarity of HNO3 = M1 = 0.255 MVolume of Mg(NO3)2 = V2 = 45.0…
Q: Part C. The constitution of ectocarpene a volatile, sperm-cell-attracting material released by the…
A: Condensed formula of ectocarpene Structural formula of ectocarpene ?
Q: Calculate the pH for each of the cases in the titration of 25.0 mL of 0.110 M pyridine, C-H₂N(aq)…
A: Weak base reacts with strong acid to form basic buffer solution. The pOH of this solution is…
Q: NEED presentation any topic u own choose but topic is good and with reactions and awesome ok i have…
A: Let's proceed step by step. For the topic of your presentation, I suggest "Nuclear Chemistry and its…
Q: Consider the following equilibrium: Hind + H₂O H3O+¹ + Ind-¹ At the transition point for an…
A: At transition point, solution changes colour. At transition point, [HIn] = [In-] HIn = indicator ,…
Step by step
Solved in 6 steps with 4 images

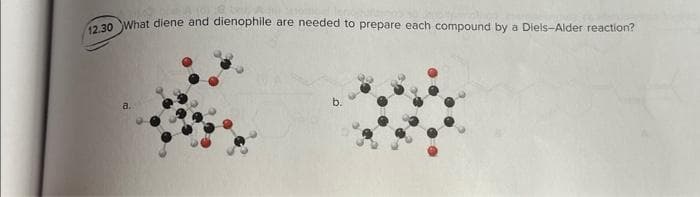
- (a) Explain how pyrrole is isoelectronic with the cyclopentadienyl anion.(b) Specifically, what is the difference between the cyclopentadienyl anion and pyrrole?(c) Draw resonance forms to show the charge distribution on the pyrrole structure.What diene and dienophile are needed to prepare each compound by a Diels–Alder reaction?Devise a stepwise synthesis of attached compound from dicyclopentadieneusing a Diels–Alder reaction as one step. You may also use organiccompounds having ≤ 4 C's, and any required organic or inorganicreagents.
- Zolpidem (trade name Ambien) promotes the rapid onset of sleep, making it a widely prescribed drug for treating insomnia. a.In what type of orbital does the lone pair on each N atom in the heterocycle reside? b. Explain why the bicyclic ring system that contains both N atoms is aromatic. c.Draw all reasonable resonance structures for the bicyclic ring system.Sulfur ylides, like the phosphorus ylides, are usefulintermediates in organic synthesis. Methyl trans-chrysanthemate, anintermediate in the synthesis of the insecticide pyrethrin I,can be prepared from diene A and a sulfur ylide. Draw a stepwisemechanism for this reaction.The Grignard reagent is reacted with oxethane (four-ring cyclic ether) to giveprimary alcohol, but the reaction is slower than the reaction between ethyleneoxides. Write down the reasons for the reaction and explain the reasons why there was a differencethe reactivity between oxethane and ethylene oxide?
- (−)-Hyoscyamine, an optically active drug used to treat gastrointestinal disorders, is isolated from Atropa belladonna, the deadly nightshade plant, by a basic aqueous extraction procedure. If too much base is used during isolation, optically inactive material is isolated. (a) Explain this result by drawing a stepwise mechanism. (b) Explain why littorine, an isomer isolated from the tailflower plant in Australia, can be obtained optically pure regardless of the amount of base used during isolation.Draw the product (A) of the following Diels–Alder reaction. A was a key intermediate in the synthesis of the addicting pain reliever morphine, the chapter-opening molecule.Why is the structure of compound X? - how do I know??
- Addition of HCl to alkene X forms two alkyl halides Y and Z. (A) Label Y and Z as a kinetic or thermodynamic product and explain why. (B) Explain why the addition of HCl occurs at the exocyclic C=C, rather than the other C=CExplain why compound A is much more stable than compound B.a. How many vinylic hydrogens does cyclopentene have? b. How many allylic hydrogens does it have?

