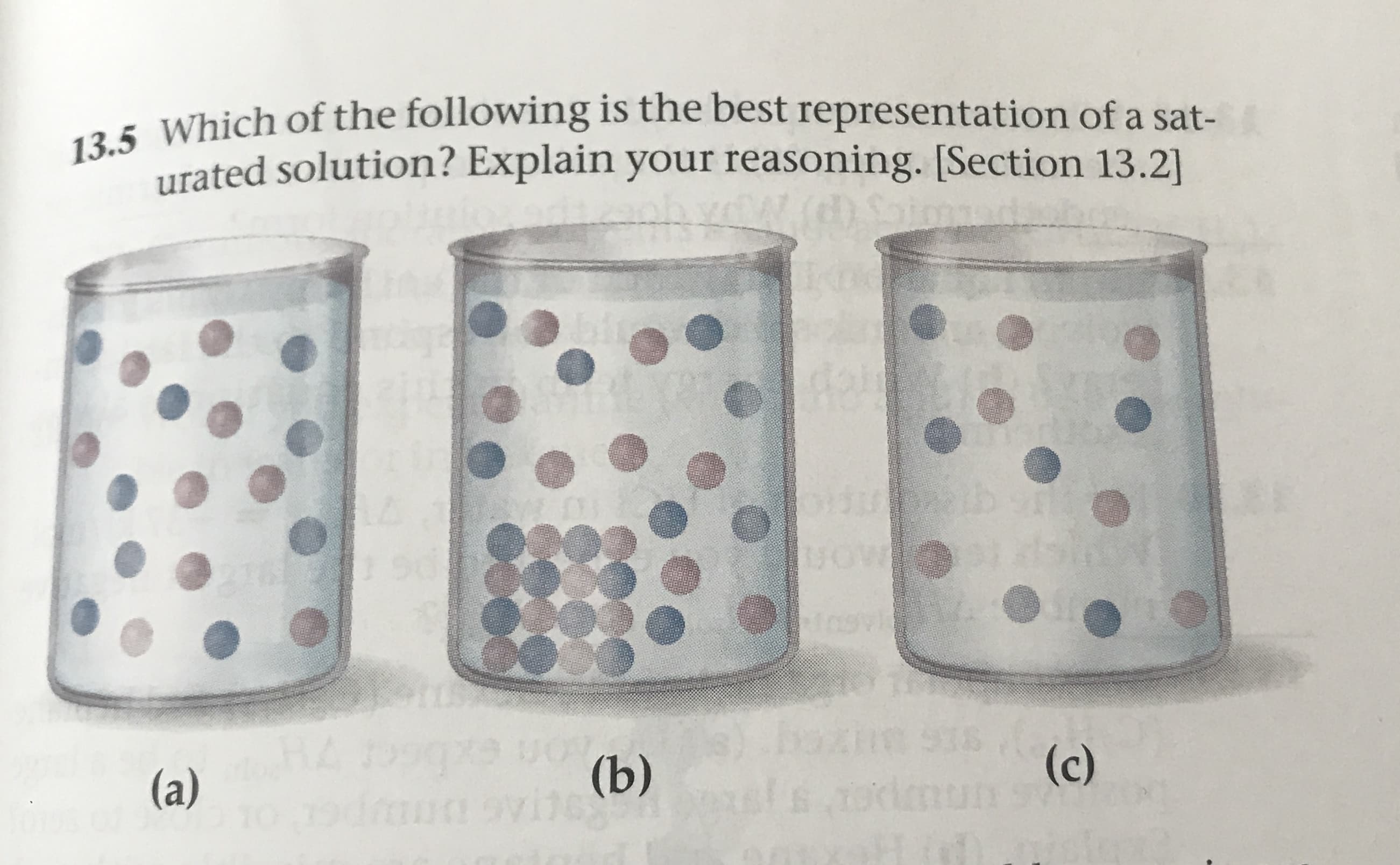
13.5 Which of the following is the best representation of a sat- urated solution? Explain your reasoning. [Section 13.2 -h yW. s) 279 VOV (b) 0: (c) ur 938 (a) RCTAG DOIUD
Q: WHAT IS The VAPOR PRessuRe at 20°C of A solution of 3.50g. NATA NAphthalene · Ciotly in 28.75g of…
A:
Q: Calculate the molarity and mole fraction of acetone in a 1.70-m solution of acetone (CH3 COCH,) in…
A: Given data: molality of solution = 1.70 m density of (acetone) solute = 0.788 g/cm3 density of…
Q: of the so
A: In this question, we have to determine the Molarity of the solution. The molarity of a solution is…
Q: Determine the boiling point elevation of a solution of 110.0 mg of carvone (a.k.a. oil of spearmint,…
A:
Q: 12.61 The molar mass of benzoic acid (C,H,COOH) determined by measuring the freezing-point…
A:
Q: What does Parry mean when he says that Solutions must be "intersectional in nature"?
A: Interpretation- To tell about here that what does parry means when he says that solution must be…
Q: What would be the saturation concentration (mole/L) of oxygen (O2) in a river in winter when the air…
A:
Q: Butane, C4H10, has been suggested as the refrigerant in household compressors such as those found in…
A:
Q: freezing point
A:
Q: calculate the amount of sodium chloride required to prepare 40mL of a concentrated solutionso that…
A:
Q: A concentrated phosphoric acid solution is 85.5%H 3 PO 4 by mass and has a density of 1.69g / m * L…
A:
Q: 5. The addition of 5.00 g of a compound to 250 g of naphthalene lowered the freezing point of…
A:
Q: I have converted the NaCl (ppm) into NaCL (M) but am uncertain what the unquenched concentration…
A: Unquenched concentration fraction is denoted as: I0IQ
Q: Identify the principal type of solute-solvent interaction responsible for forming the following…
A: A solution is a homogeneous mixture of two or more components. The component which is in less amount…
Q: gHow many grams of sodium hydroxide (M.wt-58.5) can add to Iliter of solution mixture of 0.2M…
A: Ammonia, NH3 is a weak base and ammonium chloride or ammonium ion, NH4+(aq) is a conjugate acid of…
Q: 3. The freezing point of biphenyl (molar mass 154 g/mole) is 71.0°C. When 1.240 g of benzophenone…
A: Freesing point is a colligative property meaning it depends upon the amount of solute or ions…
Q: Express concentration of 1.55 mol% caffeine (CSH10N4O2) in wt solute/ 100 wt solvent. Solvent is…
A:
Q: At a certain temperature where the density of water is 1 00 gmi 2 moles of NaCi are dissolved in 500…
A:
Q: 1. What is the mole fraction of 40% phenol(C6H6O) in benzene (C6H6)? 2. In a solution of naphthalene…
A: Answer: Mole fraction of a substance is the ratio of moles of that substance and total number of…
Q: Methanol (CH₃OH) and ethanol (C₂H₅OH) are miscible be-cause the major intermolecular force for each…
A:
Q: Calculate the molarity and mole fraction of acetone in 1.00-m solution of acetone (CH3COCH3) in…
A: Given- The concentration of the solution of acetone in ethanol = 1.00 m 1 mol of acetone in 1 kg of…
Q: The boilim a human h Determine You may
A: NOTE : according to the policies, we can only answer one question at one time. you have to post…
Q: From the following list of aqueous solutions, select th point. A 1.0 m CuCl2 B 1.0 m NaCl C 1.0 m…
A:
Q: A drop of BaCl2 was added to 0.5 F Na2SO4. If the solubility of BaSO4 is surpassed and assuming that…
A: When a drop of Barium chloride is added to 0.5 F Sodium sulfate, they both will react and form a…
Q: 4) Calculate the freezing point depression constant for chloroform (solvent) if 25.6 g naphthalene…
A: When a non volatile solute is added to a pure solvent it's freezing point decreases. Experimentally…
Q: The addition of 10.00 g of a compound to 250 g of naphthalene lowered the freezing point of the…
A: Introduction The lowering in freezing point can be derived by applying the expression shown below.…
Q: 3 An Aqueous sounon is repored by dissom ng 20-0g of Ha Br in 240.0mL solunon' ot 25.0c°. The…
A:
Q: When 1.50 g of a nonpolar solute was dissolved in 55.0g of phenol, the latter's freezing point was…
A: Given: mass of nonpolar solute=1.50 gmass of phenol=55.0 g=0.055 kgchange in temperature,…
Q: 3) A student weighed 1.75 g of an unknown molecular compound and dissolved it in 15 ml of water. The…
A:
Q: Express as % solution: 0.07 mg of maltose in 300 µL of water
A: (m/v)% or (mass/volume) % is defined as the mass of solute in grams present in 100ml of the…
Q: Which of the following solvent pairs cannot form a miscible solution? O a. and H₂C-OH O b. O c. O d.…
A:
Q: Describe the preparation ofthe following solutions: 600mL of a solution that is 0.0550 M in Cl2,…
A: Anhydrous ferric chloride can be prepared by the reaction of Fe(III) and Cl2. Hence, in the…
Q: Henry’s law is important in environmental chemistry,where it predicts the distribution of pollutants…
A: According to Henry,s law, the distribution of pollutants in water and atmosphere can be predicted…
Q: Which of the following solutes would raise the boiling point of 1.00 kg of water more one mole of…
A: The elevation in boiling point can be given as follows: where ΔTb: Elevation in boiling point…
Q: A 1.065 sample of an unknown substance is dissolved in 30g of benzene (Kf = 5.12 and Tf = 5.5ºC);…
A:
Q: Q.3 The elevation of boiling point and depression of freezing point of a certain solution of a…
A: Boiling point elevation is the phenomenon where the boiling point of the solution has a higher…
Q: The boiling point of benzene is 80.1 °C and its molal boiling point elevation constant (Kb) is 2.53…
A:
Q: 1. Calculate the normal freezing point of a 0.7439 mol aqueous solution of C12H22O11 that has a…
A: According to Q&A guidelines of Bartleby, we are supposed to answer only one question out of…
Q: Potassium Dichromate molar mass-294.185g/mol c at saturation-0.510mol/L solubility-576.833…
A: The given solubility is must be in 576.833 g/L and the molar solubility (s) is calculated as shown…
Q: b) Determine the freezing point of 500 g antifreeze, C2H6O2 in 2.5 kg of water. Given Kf for water…
A:
Q: If the solubility of barium fluoride at a temperature of 20 degrees Celsius, 0.16gm / 100ml of…
A:
Q: How momy grame of amimmium oxalate 'must be added to 500g of water to 7owver. the freezing point of…
A:
Q: molar mass When Oi4199 of the solkite of 252.4 gmall is dissolved 'en 7Sa of the Solvact , the…
A: We are authorized to answer three subparts at a time, since you have not mentioned which part you…
Q: What is the freezing point of a 0.15 m organic compound solution in benzene?
A: We know that ∆Tf= molality×kf Given is molality=0.15m Kf for benzene is =-5.12°C/mol/kg So ∆Tf=…
Q: What is the freezing point (in oC) of a solution prepared by dissolving 11.3 g of Ca(NO3)2 (formula…
A: ∆Tf = Kf x m⇒∆Tf = Kf x W1 x 1000M1X W2Where ∆Tf = Depressing in freezing pointKf = molar freesing…
Q: How many grams of sodium acetate (MM=105.99) would you add to 500.0 g water to produce a freezing…
A: Using the freezing point depression formula ∆Tf = i×Kf×mi = van't hoff factor∆Tf = freezing point…
Q: At -11.5 °C, a common temperature for household freezers, what is the maximum mass of aspartame…
A: Mass of pure water = 2 kg Freezing point of Solution = -11.5oC
Q: An osmotic pressure of 954mmHg is measured for a 1.0L solution of 1.50g MCl(aq) at 25°C. What is the…
A:
Q: *5.47. A nonideal solution contains na of substance A and nB of substance B and the mole fractions…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- The freezing point of 0.20 m HF is -0.38C. Is HF primarily nonionized in this solution (HF molecules), or is it dissociated to H+ and F- ions?What is the freezing point (in oC) of a solution prepared by dissolving 11.3 g of Ca(NO3)2 (formula weight = 164 g/mol) in 115 g of water? The molal freezing-point depression constant for water is 1.86oC/m.Methanol (CH₃OH) and ethanol (C₂H₅OH) are miscible be-cause the major intermolecular force for each is hydrogen bond-ing. In some methanol-ethanol solutions, the mole fraction ofmethanol is higher, but the mass percent of ethanol is higher. Whatis the range of mole fraction of methanol for these solutions?
- What mass of glucose (molar mass 180.16 g/mol, i=1) must be dissolved in 2.50 kg of solvent to lower the freezing point by 0.500 oC if the freezing point depression constant of the solvent is 9.51 oC/m?The partial pressure of O2 in air at sea level is 0.21 atm. Usingthe data in Table 13.1, together with Henry’s law, calculatethe molar concentration of O2 in the surface water ofa mountain lake saturated with air at 20 °C and an atmosphericpressure of 650 torr.When 13.66g of Lactic Acid (C3H6O3) are mixed with 115g of stearic acid (C18H36O2), the mixture freezes at 62.7oC. The freezing point of pure stearic acid is 69.4oC. What is the freezing depression constant of lactic acid?
- 1. What is the mole fraction of 40% phenol(C6H6O) in benzene (C6H6)? 2. In a solution of naphthalene (C10H8) in toluene (C7H8), the mole fraction of naphthalene is 0.2. What is the % by mass toluene and naphthalene in the solution?If 528 mg of an unknown sample was dissolved in 10.00 grams of t-butanol(C4H9OH) and the freezing point decreased by 3.46oC the apparent molar massfrom this data is6. An aqueous solution of hydrofluoric acid is 12.5 molal with respect to HF and has adensity of 1.070 g/mL at 25oC. Calculatea. mole fraction of HFb. %w/w of HFc. molarityd. molality
- 6 Caffeine, C8H10N4O2, is a stimulant found in tea andcoffee. A sample of the substance was dissolved in 45.0 gof chloroform, CHCl3, to give a 0.0946 m solution. Howmany grams of caffeine were in the sample10.0 grams of an unknown non-electrolyte is dissolved in 2.5 moles of water (kf=1.86 °C/m) and the change in freezing point is determined to be 4.13° C water, what is the molar mass of the unknown?What mass of glucose (molar mass 180.16 g/moles i=1) must be dissolved in 2.50 kg of solvent to lower the freezing point by .500° c if the freezing point depression constant of the solvent is 3.43°C/m?

