Q: Draw the six alkenes of molecular formula C5H10. Label one pair of diastereomers.
A: Alkenes are the hydrocarbon-containing carbon-carbon double bond. For example: CH3-CH=CH2propene ;…
Q: Draw the structure of all compounds that t the following descriptions.a. ve constitutional isomers…
A: (a)All the possible structures with the molecular formula C4H8 can be drawn by arranging the four…
Q: stereoisomerism of the compound a
A:
Q: Draw the eight constitutional isomers having molecular formula C3H;,CI. Label any stereogenic…
A:
Q: Drawn are four isomeric dimethylcyclopropanes. Which of the compounds are meso compounds?
A: The meso compounds are the optical inactive isomer of the compound although they have multiple…
Q: a) How many degrees of unsaturation (double bond equivalents) are represented by the molecular…
A:
Q: Draw the structure of all compounds that fit the following descriptions. twelve constitutional…
A: Isomers are compounds having same molecular formula but different structures. Constitutional isomers…
Q: Santalbic acid, a fatty acid isolated from the seeds of the sandalwood tree, is an unusuai fatty…
A: Given structure,
Q: Q1: The shrub ma huang contains two biologically active stereoisomers ephedrine and…
A: R/S is the Absolute configuration +/- tells about the direction in which light is rotated by the…
Q: Q2: Drawn are four isomeric dimethylcyclopropanes. a. How are the compounds in each pair related…
A:
Q: (a) Draw all stereoisomers of molecular formula C5H10Cl2 formed when (R)-2-chloropentane is heated…
A: a) Alkyl halides on reaction with Cl2 in presence of light undergoes chlorination through free…
Q: two are: ule punds t isomers isomers FH,CH, H- -CH, CH- H- CH, CH, CH,CH,
A: Isomers having same molecular formula but different molecular structure and spatial arrangement…
Q: Which compound in each pair is more basic: (a) (CH3)2NH and NH3; (b) CH3CH2NH2 and ClCH2CH2NH2?
A:
Q: Five isomeric alkanes (A–E) having the molecular formula C6H14 are each treated with Cl2 + hv to…
A: The isomers of C6H14 are as follows: A. Consider the treatment of 2-methylpentane with Cl2 + hv.…
Q: Drawn are four isomeric dimethylcyclopropanes. a.How are the compounds in each pair related…
A: Since you have posted a question with multiple subparts, we will solve first three subparts for you.…
Q: How many rings and π bonds are contained in compounds A–C? Draw one possible structure for each…
A: Note: The molecular formula of compound A in first option is C5H8.
Q: An optically active unknown compound B, whose molecular formula is C6H10, reacts with H2/Ni produce…
A:
Q: (a) Which double bonds in (E)-ocimene, a major component of the odor of lilac flowers, can exhibit…
A: (a) The double bond in (E)-ocimene that could exhibit stereoisomerism has to be given.
Q: Betamethasone is a synthetic anti-inflammatory steroid used as a topical cream for itching.…
A: Betamethasone it is a highly potent steroid that prevents the release of the inflammation causing…
Q: (a) (b) (c) B 1 + enantiomer
A:
Q: 5.57 The shrub ma huang (Section 5.4A) contains two biologically active stereoisomers-ephedrine and…
A:
Q: C6H12 , and has the following; 2 secondary carbons 2 primary carbons A plane of symmetry…
A:
Q: Q1: The shrub ma huang contains two biologically active stereoisomers ephedrine and…
A:
Q: How many rings and π bonds are contained in compounds A–C? Draw one possible structure for each…
A: a. Before hydrogenationMaximum number of hydrogen = 2n + 2 = 12Number of Hydrogen less than 12 =…
Q: How many rings and π bonds are contained in compounds A—C? Draw one possible structure for each…
A:
Q: ur iBomeric dimethylcyclopropanes. How are the compounds in each pair related (enantiomers,…
A: Enantiomers : Enantiomers are the pair of stereoisomers which are non-super imposable mirror image…
Q: 3.1 Draw both chair conformations of compound M on the templates (X and Y) provided. Indicate which…
A: Most stable chair conformer
Q: Drawn are four isomeric dimethylcyclopropanes. Which compounds have a plane of symmetry?
A: The given compounds are:
Q: Draw the structure of all compounds that f t the following descriptions. a. Five constitutional…
A:
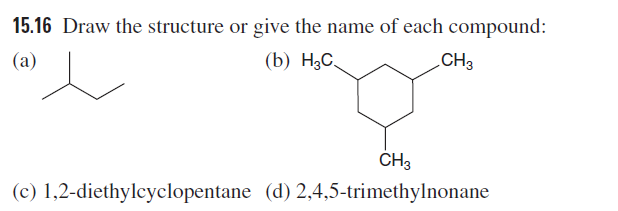
Q: Give the IUPAC name for each polyfunctional compound.
A: A systematic name is given to an organic compound by IUPAC nomenclature. The set of rules which are…
Q: 3.1 Draw both chair conformations of compound M on the templates (X and Y) provided. Indicate which…
A: The solution is as follows:
Q: The cis ketone A is isomerized to a trans ketone B with aqueous NaOH. Asimilar isomerization does…
A: (a) Tautomers are isomers which differs only in the position of the protons and electrons of the…
Q: Drow the most electron-rich carbon atom in he compound below:
A: By + R resonance effect electron density will increase at particular carbon atom.
Q: Which pair of structures are constitutional isomers? (a) A and B (b) A and C (c) C and D (d) all are…
A: Note: According to our guidelines we are supposed to answer only one question. Kindly repost other…
Q: Label compounds B–D as stereoisomers, conformations, or constitutional isomers of A
A: Stereoisomers–these isomers have same molecular formula and same sequence of bonds but differ in…
Q: Compound A has a molecular formula C6H12 , and has the following; 2 secondary carbons 2…
A: Compound A is treated with H2 and Pd to give compound B. Compound A when treated with ozone gave 2…
Q: Label compounds B–D as stereoisomers, conformations, or constitutional isomers of A.
A: Stereoisomers–these isomers have same molecular formula and same sequence of bonds but differ in…
Q: When two six-membered rings share a C—C bond, this bicyclic system is called a decalin. There are…
A: a. The structure of trans-decalin and cis-decalin are,
Q: Answer the following questions about compounds A–D.a.How are the compounds in each pair related?…
A: a) compound A and B : In the compound A and B both same formula but the methyl group connectivity…
Q: Does methylcyclopentane have a stereogenic center?
A: Methyl cyclopentane has no stereogenic center, because no any carbon atom is connected to four…
Q: Draw all 8 constitutional isomers of compounds with the molecular formula CaH11N.
A: Degree of unsaturation : C4 H11N DOU = (2C + 2 - H + N - X ) / 2 where C =…
Q: Which compound has conjugated C=C bonds? А. В. С. D.
A: Conjugated C=C bond: If the organic compound bears double bonds and single bonds between the carbon…
Q: Compound X has the formula C35H54BR2. When X is treated with excess H2 over Pd/C, the product has…
A:
Q: #544 of Isomers (chiral) Which of the following 2-chloro-1-butanol (A), 3-chloro-1-butanol (B), and…
A:
Q: If the compound A:B undergoes reaction with C to form C2B + A, what conclusion can you make?
A: IF a compound A2B is reacting with C, it is forming C2B and displacing the A from its compound. It…
Q: Draw 1,2,3,4,5,6-hexachlorocyclohexane with a. all the chloro groups in axial positions. b. all the…
A: a. The hexachlorocyclohexane molecule with chloro groups in axial and equatorial places are shown…
Q: Drawn are four isomeric dimethylcyclopropane. a. How are the compounds in each pair related…
A: A pair of compound is said to be constitutional isomers if it has the same molecular formula with…
Q: Answer the following question about compounds A–D (See in attachment) How are the compounds in each…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- The cis ketone A is isomerized to a trans ketone B with aqueous NaOH. A similar isomerization does not occur with ketone C. (a) Draw the structure of B using a chair cyclohexane. (b) Label the substituents in C as cis or trans, and explain the difference in reactivity.Butylated hydroxytoluene (BHT) is a common preservativeadded to cereals and other dry foods. Its systematic name is 1-hydroxy-2,6-di-tert-butyl-4-methylbenzene (where “tert-butyl”is 1,1-dimethylethyl). Draw the structure of BHT.Answer the following questions about compounds A–D.a.How are the compounds in each pair related? Choose from constitutional isomers, stereoisomers, or identical molecules: A and B; A and C; B and D. b.Label each compound as a cis or trans isomer. c.Draw B as a hexagon with wedges and dashed wedges to show the stereochemistry of substituents. d.Draw a stereoisomer of A as a hexagon using wedges and dashed wedges to show the orientation of substituents.
- An optically active unknown compound B, whose molecular formula is C6H10, reacts with H2/Ni produce compound C, whose molecular formula is C6H14. Compound C is optically inactive. What are the structures of compound B and C ?Drawn are four isomeric dimethylcyclopropane. a. How are the compounds in each pair related (enantiomers, diastereomers,constitutional isomers): A and B; A and C; B and C; C and D?b. Label each compound as chiral or achiral.c. Which compounds, alone, would be optically active?d. Which compounds have a plane of symmetry?e. Which of the compounds are meso compounds?f. Would an equal mixture of compounds C and D be optically active? Whatabout an equal mixture of B and C?g. How many stereogenic centers are there for each compound?Drawn are four isomeric dimethylcyclopropanes. a.How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and B; A and C; B and C; C and D? b.Label each compound as chiral or achiral. c.Which compounds alone would be optically active? d.Which compounds have a plane of symmetry? e.How do the boiling points of the compounds in each pair compare: A and B; B and C; C and D? f.Which of the compounds are meso compounds? g.Would an equal mixture of compounds C and D be optically active? What about an equal mixture of B and C?
- (a) Draw C in its more stable chair conformation. (b) Convert D to a hexagon with substituents on wedges and dashed wedges.Treatment of cyclohexene with C6H5CHI2 and Zn(Cu) forms two stereoisomers of molecular formula C13H16. Draw their structures and explain why two compounds are formed.The cis ketone A is isomerized to a trans ketone B with aqueous NaOH. Asimilar isomerization does not occur with ketone C. (a) Draw thestructure of B using a chair cyclohexane. (b) Label the substituents in Cas cis or trans, and explain the difference in reactivity.
- How many rings and π(pi) bonds are contained in compound A and draw one possible structure for this compound A. Compound A has molecular formula C6H10 and is hydrogenated to a compound having molecular formula C6H12How many rings and π bonds are contained in compounds A–C? Draw one possible structure for each compound. a. Compound A has molecular formula C5H18 and is hydrogenated to a compound having molecular formula C510H10. b. Compound B has molecular formula C10H16 and is hydrogenated to a compound having molecular formula C10H18. c. Compound C has molecular formula C8H8 and is hydrogenated to a compound having molecular formula C8H16.Draw the structure of all compounds that fit the following descriptions. twelve constitutional isomers having the molecular formula C6H12 andcontaining one ring

